GYKI 52895
This is an old revision of this page, as edited by DMacks (talk | contribs) at 04:21, 22 June 2020 (Remove malformatted |molecular_weight= when infobox can autocalculate it, per Wikipedia talk:WikiProject Pharmacology#Molecular weights in drugboxes (via WP:JWB)). The present address (URL) is a permanent link to this revision, which may differ significantly from the current revision.
 | |
 | |
| Clinical data | |
|---|---|
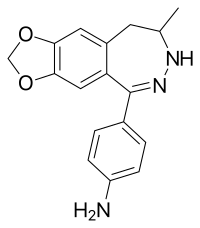
| Other names | 4-(8,9-Dihydro-8-methyl-7H-1,3-dioxolo[4,5-h][2,3]benzodiazepin-5-yl)benzenamine |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider |
|
| KEGG |
|
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C17H17N3O2 |
| Molar mass | 295.342 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
GYKI-52895 is a drug which is a 2,3-benzodiazepine derivative that also shares the 3,4-Methylenedioxyphenethylamine pharmacophore. Unlike other similar drugs, GYKI-52895 is a selective dopamine reuptake inhibitor (DARI),[1][2] which appears to have an atypical mode of action compared to other DARIs.[3] Its DRI activity is shared by numerous addictive drugs including amphetamine and its derivatives (e.g. dextromethamphetamine), cocaine, and methylphenidate and its derivatives (e.g. ethylphenidate). However, dopaminergic drugs are also prone to producing emetic effects such as in the case of apomorphine.
See also
- GYKI-52,466, another 2,3-benzodiazepine with other than GABAergic function
- Tifluadom
- Lufuradom
- Benzodiazepine
- Substituted methylenedioxyphenethylamine
References
- ^ Horváth K, Szabó H, Pátfalusi M, Berzsenyi P, Andrási F (1990). "Pharmacological Effects of GYKI 52895, a New Selective Dopamine Uptake Inhibitor". European Journal of Pharmacology. 183 (4): 1416–1417.
- ^ Huang CL, Chen HC, Huang NK, Yang DM, Kao LS, Chen JC, et al. (June 1999). "Modulation of dopamine transporter activity by nicotinic acetylcholine receptors and membrane depolarization in rat pheochromocytoma PC12 cells". Journal of Neurochemistry. 72 (6): 2437–44. doi:10.1046/j.1471-4159.1999.0722437.x. PMID 10349853.
- ^ Vaarmann A, Gandhi S, Gourine AV, Abramov AY (2010). "Novel pathway for an old neurotransmitter: dopamine-induced neuronal calcium signalling via receptor-independent mechanisms". Cell Calcium. 48 (2–3): 176–82. doi:10.1016/j.ceca.2010.08.008. PMID 20846720.
| 1,4-Benzodiazepines |
|
|---|---|
| 1,5-Benzodiazepines | |
| 2,3-Benzodiazepines* | |
| Triazolobenzodiazepines | |
| Imidazobenzodiazepines | |
| Oxazolobenzodiazepines | |
| Thienodiazepines | |
| Thienotriazolodiazepines | |
| Thienobenzodiazepines* | |
| Pyridodiazepines | |
| Pyridotriazolodiazepines | |
| Pyrazolodiazepines | |
| Pyrrolodiazepines | |
| Tetrahydroisoquinobenzodiazepines | |
| Pyrrolobenzodiazepines* | |
| Benzodiazepine prodrugs | |
* atypical activity profile (not GABAA receptor ligands) | |
This drug article relating to the nervous system is a stub. You can help Wikipedia by expanding it. |
