From Wikipedia, the free encyclopedia
Chemical compound
Arbaclofen placarbil Pregnancy ATC code Legal status
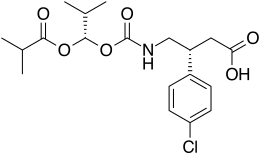
(3R )-3-(4-chlorophenyl)-4-[[[(1S )-2-methyl-1-[(2-methylpropanoyl)oxy]propoxy]carbonyl]amino]butanoic acid
CAS Number PubChem CID ChemSpider UNII KEGG ChEMBL CompTox Dashboard (EPA ) ECHA InfoCard 100.221.150 Formula C 19 H 26 Cl N O 6 Molar mass −1 3D model (JSmol )
CC(C)C(=O)O[C@@H](OC(=O)NC[C@H](CC(=O)O)c1ccc(Cl)cc1)C(C)C
InChI=1S/C19H26ClNO6/c1-11(2)17(24)26-18(12(3)4)27-19(25)21-10-14(9-16(22)23)13-5-7-15(20)8-6-13/h5-8,11-12,14,18H,9-10H2,1-4H3,(H,21,25)(H,22,23)/t14-,18-/m0/s1
Key:JXTAALBWJQJLGN-KSSFIOAISA-N
Arbaclofen placarbil ( ar-BAK -loh-fen plə-KAR -bil XP19986 ) is a prodrug of R -baclofen . Arbaclofen placarbil possesses more favorable pharmacokinetic profile than baclofen, with less fluctuations in plasma drug levels. It was being developed as a potential treatment for patients with GERD and spasticity due to multiple sclerosis ; however, in May 2013 XenoPort announced the termination of development because of unsuccessful results in phase III clinical trials.[1]
It is being developed as an addiction medicine to treat alcoholism. [2] [3]
See also [ edit ] References [ edit ]
Ionotropic
GABAA Tooltip γ-Aminobutyric acid A receptor
Positive modulators (abridged; see here for a full list): α-EMTBL Alcohols (e.g., drinking alcohol , 2M2B )Anabolic steroids Avermectins (e.g., ivermectin )Barbiturates (e.g., phenobarbital )Benzodiazepines (e.g., diazepam )Bromide compounds (e.g., potassium bromide )Carbamates (e.g., meprobamate )Carbamazepine Chloralose Chlormezanone Clomethiazole Dihydroergolines (e.g., ergoloid (dihydroergotoxine) )Etazepine Etifoxine Fenamates (e.g., mefenamic acid )Flavonoids (e.g., apigenin , hispidulin )Fluoxetine Flupirtine Imidazoles (e.g., etomidate )Kava constituents (e.g., kavain )Lanthanum Loreclezole Monastrol Neuroactive steroids (e.g., allopregnanolone , cholesterol , THDOC )Niacin Niacinamide Nonbenzodiazepines (e.g., β-carbolines (e.g., abecarnil ), cyclopyrrolones (e.g., zopiclone ), imidazopyridines (e.g., zolpidem ), pyrazolopyrimidines (e.g., zaleplon ))Norfluoxetine Petrichloral Phenols (e.g., propofol )Phenytoin Piperidinediones (e.g., glutethimide )Propanidid Pyrazolopyridines (e.g., etazolate )Quinazolinones (e.g., methaqualone )Retigabine (ezogabine) ROD-188 Skullcap constituents (e.g., baicalin )Stiripentol Sulfonylalkanes (e.g., sulfonmethane (sulfonal) )Topiramate Valerian constituents (e.g., valerenic acid )Volatiles /gases (e.g., chloral hydrate , chloroform , diethyl ether , paraldehyde , sevoflurane )Negative modulators: 1,3M1B 3M2B 11-Ketoprogesterone 17-Phenylandrostenol α3IA α5IA (LS-193,268) β-CCB β-CCE β-CCM β-CCP β-EMGBL Anabolic steroids Amiloride Anisatin β-Lactams (e.g., penicillins , cephalosporins , carbapenems )Basmisanil Bemegride Bicyclic phosphates (TBPS , TBPO , IPTBO )BIDN Bilobalide Bupropion CHEB Chlorophenylsilatrane Cicutoxin Cloflubicyne Cyclothiazide DHEA DHEA-S Dieldrin (+)-DMBB DMCM DMPC EBOB Etbicyphat FG-7142 (ZK-31906) Fiproles (e.g., fipronil )Flavonoids (e.g., amentoflavone , oroxylin A )Flumazenil Fluoroquinolones (e.g., ciprofloxacin )Flurothyl Furosemide Golexanolone Iomazenil (123 I) IPTBO Isopregnanolone (sepranolone) L-655,708 Laudanosine Lindane MaxiPost Morphine Morphine-3-glucuronide MRK-016 Naloxone Naltrexone Nicardipine Nonsteroidal antiandrogens (e.g., apalutamide , bicalutamide , enzalutamide , flutamide , nilutamide )Oenanthotoxin Pentylenetetrazol (pentetrazol) Phenylsilatrane Picrotoxin (i.e., picrotin , picrotoxinin and dihydropicrotoxinin )Pregnenolone sulfate Propybicyphat PWZ-029 Radequinil Ro 15-4513 Ro 19-4603 RO4882224 RO4938581 Sarmazenil SCS Suritozole TB-21007 TBOB TBPS TCS-1105 Terbequinil TETS Thujone U-93631 Zinc ZK-93426 GABAA -ρ Tooltip γ-Aminobutyric acid A-rho receptor
Metabotropic
GABAB Tooltip γ-Aminobutyric acid B receptor
Calcium
VDCCs Tooltip Voltage-dependent calcium channels
Potassium
VGKCs Tooltip Voltage-gated potassium channels
IRKs Tooltip Inwardly rectifying potassium channel
KCa Tooltip Calcium-activated potassium channel
K2Ps Tooltip Tandem pore domain potassium channel
Sodium
VGSCs Tooltip Voltage-gated sodium channels
ENaC Tooltip Epithelial sodium channel
ASICs Tooltip Acid-sensing ion channel
Chloride
CaCCs Tooltip Calcium-activated chloride channel
CFTR Tooltip Cystic fibrosis transmembrane conductance regulator
Unsorted
Others
TRPs Tooltip Transient receptor potential channels LGICs Tooltip Ligand gated ion channels