From Wikipedia, the free encyclopedia
Chemical compound
Azaperone is a pyridinylpiperazine and butyrophenone neuroleptic drug with sedative and antiemetic effects, which is used mainly as a tranquilizer in veterinary medicine.[ 1] antipsychotic drug.
Azaperone acts primarily as a dopamine antagonist but also has some antihistaminic and anticholinergic properties as seen with similar drugs such as haloperidol . Azaperone may cause hypotension and while it has minimal effects on respiration in pigs, high doses in humans can cause respiratory depression .
Veterinary use
The most common use for azaperone is in relatively small doses as a "serenic " (to reduce aggression) in farmed pigs, either to stop them fighting or to encourage sows to accept piglets. Higher doses are used for anesthesia in combination with other drugs such as xylazine , tiletamine and zolazepam . Azaperone is also used in combination with strong narcotics such as etorphine or carfentanil for tranquilizing large animals such as elephants.[ 2]
The European Medicines Agency has established a maximum residue limit for azaperone when administered to pigs.[ 3]
Azaperone (under the brand name Stresnil) was approved for use in pigs in the USA in 1983, under NADA 115-732.[ 4]
Synthesis
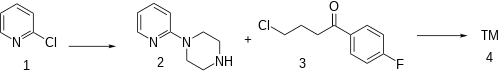
52%:[ 5] [ 6] [ 7] [ 8] The alkylation of 2-chloropyridine (1 ) with piperazine gives 1-(pyridin-2-yl)piperazine [67980-77-2] (2 ). The attachement of the sidechain by reaction with 4-chloro-4'-fluorobutyrophenone [3874-54-2] (3 ) completed the synthesis of azaperone (4 ).
See also
References
^ Posner LP, Burns P (2013). "Sedative agents: tranquilizers, alpha-2 agonists, and related agents. Veterinary pharmacology and therapeutics. 2009:." . In Riviere JE, Papich MG (eds.). Veterinary Pharmacology and Therapeutics (9th ed.). Somerset: Wiley. pp. 337-380 (366). ISBN 978-1-118-68590-7 ^ "The Elephant Formulary" . Archived from the original on 2012-02-05. Retrieved 2007-04-30 .^ "Azaperone Summary Report (2)" (PDF) . European Medicines Agency . Committee for Veterinary Medicinal Products. November 1997. Retrieved 2017-01-15 .^ "Rules and Regulations" (PDF) . Federal Register . 48 (202): 48229. 18 October 1983. Retrieved 2017-01-15 .^ Jaen, Juan C.; Caprathe, Bradley W.; Pugsley, Thomas A.; Wise, Lawrence D.; Akunne, Hyacinth (1993). "Evaluation of the effects of the enantiomers of reduced haloperidol, azaperol, and related 4-amino-1-arylbutanols on dopamine and .sigma. receptors". Journal of Medicinal Chemistry. 36 (24): 3929–3936. doi:10.1021/jm00076a022.
^ Janssen Paul Adriaan Jan, U.S. patent 2,958,694
^ Taghizadeh, M. J., Mohammadnia, M. S., Ghalkhani, M., Sohouli, E. (April 2022). "Improved Method for the Total Synthesis of Azaperone and Investigation of Its Electrochemical Behavior in Aqueous Solution". Chemical Research in Chinese Universities . 38 (2): 546–551. doi :10.1007/s40242-021-1061-2 . ISSN 2210-3171 1005-9040, 2210-3171 . ^ Soudijn, W.; van Wijngaarden, I. (1968). "A rapid and convenient method for the synthesis of labelled tertiary amines". Journal of Labelled Compounds. 4 (2): 159–163. doi:10.1002/jlcr.2590040209.
mAChRs Tooltip Muscarinic acetylcholine receptors
Agonists Antagonists
3-Quinuclidinyl benzilate 4-DAMP Aclidinium bromide (+formoterol )Abediterol AF-DX 250 AF-DX 384 Ambutonium bromide Anisodamine Anisodine Antihistamines (first-generation) (e.g., brompheniramine , buclizine , captodiame , chlorphenamine (chlorpheniramine) , cinnarizine , clemastine , cyproheptadine , dimenhydrinate , dimetindene , diphenhydramine , doxylamine , meclizine , mequitazine , perlapine , phenindamine , pheniramine , phenyltoloxamine , promethazine , propiomazine , triprolidine )AQ-RA 741 Atropine Atropine methonitrate Atypical antipsychotics (e.g., clozapine , fluperlapine , olanzapine (+fluoxetine ), rilapine , quetiapine , tenilapine , zotepine )Benactyzine Benzatropine (benztropine) Benzilone Benzilylcholine mustard Benzydamine Bevonium BIBN 99 Biperiden Bornaprine Camylofin CAR-226,086 CAR-301,060 CAR-302,196 CAR-302,282 CAR-302,368 CAR-302,537 CAR-302,668 Caramiphen Cimetropium bromide Clidinium bromide Cloperastine CS-27349 Cyclobenzaprine Cyclopentolate Darifenacin DAU-5884 Desfesoterodine Dexetimide DIBD Dicycloverine (dicyclomine) Dihexyverine Difemerine Diphemanil metilsulfate Ditran Drofenine EA-3167 EA-3443 EA-3580 EA-3834 Emepronium bromide Etanautine Etybenzatropine (ethybenztropine) Fenpiverinium Fentonium bromide Fesoterodine Flavoxate Glycopyrronium bromide (+beclometasone/formoterol , +indacaterol , +neostigmine )Hexahydrodifenidol Hexahydrosiladifenidol Hexbutinol Hexocyclium Himbacine HL-031,120 Homatropine Imidafenacin Ipratropium bromide (+salbutamol )Isopropamide J-104,129 Hyoscyamine Mamba toxin 3 Mamba toxin 7 Mazaticol Mebeverine Meladrazine Mepenzolate Methantheline Methoctramine Methylatropine Methylhomatropine Methylscopolamine Metixene Muscarinic toxin 7 N-Ethyl-3-piperidyl benzilate N-Methyl-3-piperidyl benzilate Nefopam Octatropine methylbromide (anisotropine methylbromide) Orphenadrine Otenzepad (AF-DX 116) Otilonium bromide Oxapium iodide Oxitropium bromide Oxybutynin Oxyphencyclimine Oxyphenonium bromide PBID PD-102,807 PD-0298029 Penthienate Pethidine pFHHSiD Phenglutarimide Phenyltoloxamine Pipenzolate bromide Piperidolate Pirenzepine Piroheptine Pizotifen Poldine Pridinol Prifinium bromide Procyclidine Profenamine (ethopropazine) Propantheline bromide Propiverine Quinidine 3-Quinuclidinyl thiochromane-4-carboxylate Revefenacin Rociverine RU-47,213 SCH-57,790 SCH-72,788 SCH-217,443 Scopolamine (hyoscine) Scopolamine butylbromide (hyoscine butylbromide) Silahexacyclium Sofpironium bromide Solifenacin SSRIs Tooltip Selective serotonin reuptake inhibitors (e.g., femoxetine , paroxetine )Telenzepine Terodiline Tetracyclic antidepressants (e.g., amoxapine , maprotiline , mianserin , mirtazapine )Tiemonium iodide Timepidium bromide Tiotropium bromide Tiquizium bromide Tofenacin Tolterodine Tricyclic antidepressants (e.g., amitriptyline (+perphenazine ), amitriptylinoxide , butriptyline , cidoxepin , clomipramine , desipramine , desmethyldesipramine , dibenzepin , dosulepin (dothiepin) , doxepin , imipramine , lofepramine , nitroxazepine , northiaden (desmethyldosulepin) , nortriptyline , protriptyline , quinupramine , trimipramine )Tridihexethyl Trihexyphenidyl Trimebutine Tripitamine (tripitramine) Tropacine Tropatepine Tropicamide Trospium chloride Typical antipsychotics (e.g., chlorpromazine , chlorprothixene , cyamemazine (cyamepromazine) , loxapine , mesoridazine , thioridazine )Umeclidinium bromide (+vilanterol )WIN-2299 Xanomeline Zamifenacin
Precursors (and prodrugs )
nAChRs Tooltip Nicotinic acetylcholine receptors
Agonists PAMs Tooltip positive allosteric modulators )
5-HIAA 6-Chloronicotine A-84,543 A-366,833 A-582,941 A-867,744 ABT-202 ABT-418 ABT-560 ABT-894 Acetylcholine Altinicline Anabasine Anatabine Anatoxin-a AR-R17779 Bephenium hydroxynaphthoate Butinoline Butyrylcholine Carbachol Choline Cotinine Cytisine Decamethonium Desformylflustrabromine Dianicline Dimethylphenylpiperazinium Epibatidine Epiboxidine Ethanol (alcohol) Ethoxysebacylcholine EVP-4473 EVP-6124 Galantamine GTS-21 Ispronicline Ivermectin JNJ-39393406 Levamisole Lobeline MEM-63,908 (RG-3487) Morantel Nicotine (tobacco )NS-1738 PHA-543,613 PHA-709,829 PNU-120,596 PNU-282,987 Pozanicline Pyrantel Rivanicline RJR-2429 Sazetidine A SB-206553 Sebacylcholine SIB-1508Y SIB-1553A SSR-180,711 Suberyldicholine Suxamethonium (succinylcholine) Suxethonium (succinyldicholine) TC-1698 TC-1734 TC-1827 TC-2216 TC-5214 TC-5619 TC-6683 Tebanicline Tribendimidine Tropisetron UB-165 Varenicline WAY-317,538 XY-4083 Antagonists NAMs Tooltip negative allosteric modulators )
Precursors (and prodrugs )
H1
Agonists Antagonists
Others: Atypical antipsychotics (e.g., aripiprazole , asenapine , brexpiprazole , brilaroxazine , clozapine , iloperidone , olanzapine , paliperidone , quetiapine , risperidone , ziprasidone , zotepine )Phenylpiperazine antidepressants (e.g., hydroxynefazodone , nefazodone , trazodone , triazoledione )Tetracyclic antidepressants (e.g., amoxapine , loxapine , maprotiline , mianserin , mirtazapine , oxaprotiline )Tricyclic antidepressants (e.g., amitriptyline , butriptyline , clomipramine , desipramine , dosulepin (dothiepin) , doxepin , imipramine , iprindole , lofepramine , nortriptyline , protriptyline , trimipramine )Typical antipsychotics (e.g., chlorpromazine , flupenthixol , fluphenazine , loxapine , perphenazine , prochlorperazine , thioridazine , thiothixene )
H2
H3
H4
Simple piperazines(no additional rings) Phenylpiperazines
2C-B-PP 3,4-CFP Acaprazine Antrafenine Aripiprazole Batoprazine Bifeprunox BRL-15,572 Ciprofloxacin CSP-2503 Dapiprazole DCPP DMPP Diphenylpiperazine Dropropizine EGIS-12,233 Elopiprazole Eltoprazine Enpiprazole Ensaculin Etoperidone Flesinoxan Fluanisone Flibanserin Fluprazine Itraconazole Ketoconazole Levodropropizine Lorpiprazole mCPP Mefway MeOPP Mepiprazole Naftopidil Naluzotan Naphthylpiperazine Nefazodone Niaprazine Oxypertine Pardoprunox pCPP pFPP Posaconazole S-14,506 S-14,671 S-15,535 SB-258,585 SB-271,046 SB-357,134 SB-399,885 Sonepiprazole TFMPP Tolpiprazole Trazodone Urapidil Vesnarinone Vilazodone Vortioxetine WAY-100,135 WAY-100,635 Benzylpiperazines Diphenylalkylpiperazines (benzhydrylalkylpiperazines) Pyrimidinylpiperazines Pyridinylpiperazines Benzo(iso)thiazolyl piperazines Tricyclics (piperazine attached via side chain) Others/Uncategorized