Bupropion: Difference between revisions
Delivery shell. |
Undid revision 529748900 by Walks on Water (talk) whoops! Self-revert. |
||
| Line 2: | Line 2: | ||
{{drugbox |
{{drugbox |
||
| Verifiedfields = changed |
| Verifiedfields = changed |
||
| Watchedfields = changed |
|||
| verifiedrevid = 443491992 |
| verifiedrevid = 443491992 |
||
| IUPAC_name = (±)-2-(''tert''-Butylamino)-1-(3-chlorophenyl)propan-1-one |
| IUPAC_name = (±)-2-(''tert''-Butylamino)-1-(3-chlorophenyl)propan-1-one |
||
| Line 32: | Line 31: | ||
<!--Identifiers--> |
<!--Identifiers--> |
||
| CASNo_Ref = {{cascite|correct|CAS}} |
| CASNo_Ref = {{cascite|correct|CAS}} |
||
| CAS_number_Ref = {{cascite|correct|??}} |
|||
| CAS_number = 34841-39-9 |
| CAS_number = 34841-39-9 |
||
| ATC_prefix = N06 |
| ATC_prefix = N06 |
||
| Line 60: | Line 60: | ||
}} |
}} |
||
'''Bupropion''' ({{IPAc-en|b|juː|ˈ|p|r|oʊ|p|i|.|ɒ|n}} {{respell|bew|PROH|pee-on}};<ref>entry "[http://www.ncbi.nlm.nih.gov/pubmedhealth/PMH0000970 Bupropion]" at [http://www.ncbi.nlm.nih.gov/pubmedhealth/PMH0000970/ "PubMed Health" (from the US government agency NIH, National Institutes of Health], retrieved 2011-04-07.</ref> marketed as '''Wellbutrin''', '''Zyban''', '''Voxra''', '''Budeprion''', or '''Aplenzin'''; and formerly known as '''amfebutamone'''<ref>The INN originally assigned in 1974 by the [[World Health Organization]] was "amfebutamone". In 2000, the INN was reassigned as ''bupropion''. See {{cite journal | author = [[World Health Organization]] | title = International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 83 | journal = WHO Drug Information | volume = 14 | issue = 2 | year = 2000 | url = http://82.77.46.154/gsdldata/collect/whodruginfo/index/assoc/h1463e/h1463e.pdf|format=PDF|accessdate=2009-06-22 |
'''Bupropion''' ({{IPAc-en|b|juː|ˈ|p|r|oʊ|p|i|.|ɒ|n}} {{respell|bew|PROH|pee-on}};<ref>entry "[http://www.ncbi.nlm.nih.gov/pubmedhealth/PMH0000970 Bupropion]" at [http://www.ncbi.nlm.nih.gov/pubmedhealth/PMH0000970/ "PubMed Health" (from the US government agency NIH, National Institutes of Health], retrieved 2011-04-07.</ref> marketed as '''Wellbutrin''', '''Zyban''', '''Voxra''', '''Budeprion''', '''Prexaton''', '''Elontril''' or '''Aplenzin'''; and formerly known as '''amfebutamone'''<ref>The INN originally assigned in 1974 by the [[World Health Organization]] was "amfebutamone". In 2000, the INN was reassigned as ''bupropion''. See {{cite journal | author = [[World Health Organization]] | title = International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 83 | journal = WHO Drug Information | volume = 14 | issue = 2 | year = 2000 | url = http://82.77.46.154/gsdldata/collect/whodruginfo/index/assoc/h1463e/h1463e.pdf|format=PDF|accessdate=2009-06-22 | archiveurl = http://web.archive.org/web/20110531073025/http://82.77.46.154/gsdldata/collect/whodruginfo/index/assoc/h1463e/h1463e.pdf | archivedate = 2011-05-31}}</ref>) is an atypical [[antidepressant]] and [[smoking cessation]] aid. Its chemical name is β-keto-3-chloro-''N''-tert-butylamphetamine, a [[substituted cathinone]] (β-ketoamphetamine), as well as [[substituted amphetamine]]. The drug therefore is a mild psychostimulant. Its primary pharmacological action is thought to be [[norepinephrine-dopamine reuptake inhibitor|norepinephrine-dopamine reuptake inhibition]]. It binds selectively to the [[dopamine transporter]], but its behavioural effects have often been attributed to its inhibition of [[norepinephrine]] [[reuptake inhibitor|reuptake]].<ref>{{cite journal | author = Terry P, Katz JL | title = Dopaminergic mediation of the discriminative stimulus effects of bupropion.. | journal = Psychopharmacology (Berl) | volume = 134| issue = 2 | pages = 201–12 | year = 1997 | url = http://www.biopsychiatry.com/wellbutrin.htm | pmid=9399385 | doi = 10.1007/s002130050443}}</ref><ref>{{cite journal | author = Learned-Coughlin SM, Bergstrom M, Savitcheva I, Ascher J, Schmith VD, Langstrom B | title = In vivo activity of bupropion at the human dopamine transporter.. | journal = Biol Psychiatry | volume = 54| issue = 8 | pages = 800–5 | year = 2003 | url = http://www.biopsychiatry.com/bupropion-dopamine.htm | pmid=14550679 | doi=10.1016/S0006-3223(02)01834-6}}</ref> It also acts as a [[nicotinic acetylcholine receptor]] [[receptor antagonist|antagonist]].<ref>{{cite journal | author = Slemmer J E, Martin R M, Damaj M I | title = Bupropion is a Nicotinic Antagonist | journal = J Pharmacol Exp Ther | volume = 295| issue = 1 |pages = 321–327 | year = 2000 | pmid = 10991997}}</ref><ref name="Fryer1999">{{cite journal | author = Fryer J D, Lukas R J | title = Noncompetitive functional inhibition at diverse, human nicotinic acetylcholine receptor subtypes by bupropion, phencyclidine, and ibogaine | journal = J Pharmacol Exp Ther | volume = 288|issue=6|pages = 88–92 | year = 1999 |pmid=9862757}}</ref> Bupropion belongs to the [[chemical class]] of [[aminoketone]]s and is similar in [[chemical structure|structure]] to stimulants [[cathinone]] and [[diethylpropion]], and to [[substituted phenethylamine|phenethylamine]]s in general. Medically, bupropion serves as a non-tricyclic antidepressant fundamentally different to most commonly prescribed antidepressants such as [[Selective serotonin reuptake inhibitors|SSRIs]]. |
||
Initially researched and marketed as an antidepressant, bupropion was subsequently found to be effective as a smoking cessation aid. With over 20 million retail prescriptions in 2007, it was the fourth-most prescribed antidepressant in the United States retail market after [[sertraline]], [[escitalopram]], and [[fluoxetine]].<ref>The bupropion prescriptions were calculated as a total of prescriptions for Wellbutrin XL, Budeprion XL, Budeprion SR, Bupropion SR and Bupropion ER using data from the charts for generic and brand-name drugs, see: {{cite web | url = http://www.drugtopics.com/drugtopics/article/articleDetail.jsp?id=491181 | title = Top 200 Generic Drugs by Units in 2007| accessdate = 2008-03-30 |date = 2008-02-18| format = PDF| publisher = Drug Topics}} and {{cite web | url = http://www.drugtopics.com/drugtopics/article/articleDetail.jsp?id=491207 | title = Top 200 Brand Drugs by Units in 2007| accessdate = 2008-03-30 | author = Verispan|date = 2008-02-18| format = PDF | publisher = Drug Topics}}</ref> |
Initially researched and marketed as an antidepressant, bupropion was subsequently found to be effective as a smoking cessation aid. With over 20 million retail prescriptions in 2007, it was the fourth-most prescribed antidepressant in the United States retail market after [[sertraline]], [[escitalopram]], and [[fluoxetine]].<ref>The bupropion prescriptions were calculated as a total of prescriptions for Wellbutrin XL, Budeprion XL, Budeprion SR, Bupropion SR and Bupropion ER using data from the charts for generic and brand-name drugs, see: {{cite web | url = http://www.drugtopics.com/drugtopics/article/articleDetail.jsp?id=491181 | title = Top 200 Generic Drugs by Units in 2007| accessdate = 2008-03-30 |date = 2008-02-18| format = PDF| publisher = Drug Topics}} and {{cite web | url = http://www.drugtopics.com/drugtopics/article/articleDetail.jsp?id=491207 | title = Top 200 Brand Drugs by Units in 2007| accessdate = 2008-03-30 | author = Verispan|date = 2008-02-18| format = PDF | publisher = Drug Topics}}</ref> |
||
Bupropion lowers [[seizure threshold]], and its potential to cause seizures has been widely publicized. However, at the recommended dose the risk of seizures is comparable to that observed for other antidepressants. Bupropion is an effective antidepressant on its own but it is particularly popular as an add-on medication in the cases of incomplete response to the first-line [[selective serotonin reuptake inhibitor]] (SSRI) antidepressant. In contrast to many other antidepressants, bupropion does not cause [[weight gain]] or [[sexual dysfunction]]; |
Bupropion lowers [[seizure threshold]], and its potential to cause seizures has been widely publicized. However, at the recommended dose, the risk of seizures is comparable to that observed for other antidepressants.<ref name=" Johnston " /> Bupropion is an effective antidepressant on its own, but it is particularly popular as an add-on medication in the cases of incomplete response to the first-line [[selective serotonin reuptake inhibitor]] (SSRI) antidepressant. In contrast to many other antidepressants, bupropion does not cause [[weight gain]] or [[sexual dysfunction]]; in most studies, groups placed on bupropion showed statistically significant increases in sexual functioning ([[libido]]), and mild to moderate [[weight loss]].<ref name="ReferenceA">For a short review, see: {{cite journal | author = Zimmerman M, Posternak MA, Attiullah N, Friedman M, Boland RJ, Baymiller S, Berlowitz SL, Rahman S, Uy KK, Singer S, Chelminski I, Thongy, T| title = Dr. Zimmerman and colleagues reply to MJ Menaster | journal = J Clin Psychiatry| volume = 66| issue=10 |pages = 1336–9 | year = 2005}}</ref> |
||
==Medical uses== |
==Medical uses== |
||
===Depression=== |
===Depression=== |
||
[[Placebo]]-controlled [[double-blind]] [[clinical studies]] have confirmed the efficacy of bupropion for [[Major depressive disorder|clinical depression]].<ref>{{cite journal | author = Fava M, Rush AJ, Thase ME, Clayton A, Stahl SM, Pradko JF, Johnston JA. | title = 15 years of clinical experience with bupropion HCl: from bupropion to bupropion SR to bupropion XL | journal = Prim Care Companion J Clin Psychiatry| volume = 7|issue = 3|pages = 106–113| year = 2005 |pmid=16027765 |url=http://www.psychiatrist.com/pcc/pccpdf/v07n03/v07n0305.pdf|format=PDF|accessdate=2008-12-30 | pmc = 1163271 | doi = 10.4088/PCC.v07n0305}}</ref> Comparative clinical studies demonstrated the equivalency of bupropion and [[sertraline]] (Zoloft), [[fluoxetine]] (Prozac), [[paroxetine]] (Paxil)<ref>{{cite journal | author = Thase ME, Haight BR, Richard N, Rockett CB, Mitton M, Modell JG, VanMeter S, Harriett AE, Wang Y| title = Remission rates following antidepressant therapy with bupropion or selective serotonin reuptake inhibitors: a meta-analysis of original data from 7 randomized controlled trials | journal = J Clin Psychiatry| volume = 66| issue=6 |pages = 974–981 | year = 2005|pmid = 16086611 | doi = 10.4088/JCP.v66n0803}}</ref> and [[escitalopram]] (Lexapro)<ref>{{cite journal | author = Clayton AH, Croft HA, Horrigan JP, Wightman DS, Krishen A, Richard NE, Modell JG| title = Bupropion extended release compared with escitalopram: effects on sexual functioning and antidepressant efficacy in 2 randomized, double-blind, placebo-controlled studies | journal = J Clin Psychiatry| volume = 67| issue=5 |pages = 736–746 | year = 2006|pmid = 1684162 | doi = 10.4088/JCP.v67n0507}}</ref> as antidepressants. |
[[Placebo]]-controlled [[double-blind]] [[clinical studies]] have confirmed the efficacy of bupropion for [[Major depressive disorder|clinical depression]].<ref>{{cite journal | author = Fava M, Rush AJ, Thase ME, Clayton A, Stahl SM, Pradko JF, Johnston JA. | title = 15 years of clinical experience with bupropion HCl: from bupropion to bupropion SR to bupropion XL | journal = Prim Care Companion J Clin Psychiatry| volume = 7|issue = 3|pages = 106–113| year = 2005 |pmid=16027765 |url=http://www.psychiatrist.com/pcc/pccpdf/v07n03/v07n0305.pdf|format=PDF|accessdate=2008-12-30 | pmc = 1163271 | doi = 10.4088/PCC.v07n0305}}</ref> Comparative clinical studies demonstrated the equivalency of bupropion and [[sertraline]] (Zoloft), [[fluoxetine]] (Prozac), [[paroxetine]] (Paxil)<ref>{{cite journal | author = Thase ME, Haight BR, Richard N, Rockett CB, Mitton M, Modell JG, VanMeter S, Harriett AE, Wang Y| title = Remission rates following antidepressant therapy with bupropion or selective serotonin reuptake inhibitors: a meta-analysis of original data from 7 randomized controlled trials | journal = J Clin Psychiatry| volume = 66| issue=6 |pages = 974–981 | year = 2005|pmid = 16086611 | doi = 10.4088/JCP.v66n0803}}</ref> and [[escitalopram]] (Lexapro)<ref>{{cite journal | author = Clayton AH, Croft HA, Horrigan JP, Wightman DS, Krishen A, Richard NE, Modell JG| title = Bupropion extended release compared with escitalopram: effects on sexual functioning and antidepressant efficacy in 2 randomized, double-blind, placebo-controlled studies | journal = J Clin Psychiatry| volume = 67| issue=5 |pages = 736–746 | year = 2006|pmid = 1684162 | doi = 10.4088/JCP.v67n0507}}</ref> as antidepressants. One study reported a significantly higher [[remission (medicine)|remission]] rate with bupropion treatment than for [[venlafaxine]] (Effexor).<ref>{{cite journal | author = Thase ME, Clayton AH, Haight BR, Thompson AH, Modell JG, Johnston JA| title = A double-blind comparison between bupropion XL and venlafaxine XR: sexual functioning, antidepressant efficacy, and tolerability | journal = J Clin Psychopharmacol| volume = 26| issue=5 |pages = 482–488 | year = 2006|pmid = 16974189 | doi = 10.1097/01.jcp.0000239790.83707.ab}}</ref> Unlike all other antidepressants, except [[mirtazapine]] (Remeron) and [[tianeptine]] (Stablon), bupropion does not cause sexual dysfunction and the occurrence of sexual side effects is not different from placebo.<ref>For the review, see: {{cite journal | author = Clayton AH| title = Antidepressant-Associated Sexual Dysfunction: A Potentially Avoidable Therapeutic Challenge | journal = Primary Psychiatry| volume = 10| issue=1 |pages = 55–61 | year = 2003}}</ref><ref>For another review, see: {{cite journal | author = Kanaly KA, Berman JR| title = Sexual side effects of SSRI medications: potential treatment strategies for SSRI-induced female sexual dysfunction | journal = Curr Women's Health Rep| volume = 2| issue=6 |pages = 409–16 | year = 2002|pmid=12429073}}</ref> Bupropion treatment is not associated with weight gain; on the contrary, at the end of every study comparing bupropion with placebo or other antidepressants the bupropion group had a lower average weight.<ref name="ReferenceA"/> Bupropion is more effective than [[SSRI]]s at improving symptoms of [[hypersomnia]] and fatigue in depressed patients.<ref>{{cite journal | author = Baldwin DS, Papakostas GI| title = Symptoms of Fatigue and Sleepiness in Major Depressive Disorder | journal = J Clin Psychiatry| volume = 67 (suppl 6)| pages = 9–15 | year = 2006 |pmid=16848671}}</ref> In a comparative meta-analysis, there appeared to be a modest advantage for the SSRIs compared to bupropion in the treatment of depression with high anxiety, while these medications were equivalent for depression with moderate or low anxiety.<ref name="pmid18605812">{{cite journal|title=Efficacy of Bupropion and the Selective Serotonin Reuptake Inhibitors in the Treatment of Major Depressive Disorder With High Levels of Anxiety (Anxious Depression): A Pooled Analysis of 10 Studies |journal=J Clin Psychiatry |volume= 69|issue= 8|pages=e1–e6 |year=2008|pmid=18605812 |url=http://article.psychiatrist.com/?ContentType=START&ID=10003663|last1=Papakostas|first1=GI|last2=Stahl|first2=SM|last3=Krishen|first3=A|last4=Seifert|first4=CA|last5=Tucker|first5=VL|last6=Goodale|first6=EP|last7=Fava|first7=M}}</ref> |
||
According to several surveys, the [[Augmentation (psychiatry)|augmentation]] of a prescribed [[SSRI]] with bupropion is the preferred strategy among clinicians when the patient does not respond to the SSRI.<ref>For the most recent review, see: {{cite journal |author=Zisook S, Rush AJ, Haight BR, Clines DC, Rockett CB |title=Use of bupropion in combination with serotonin reuptake inhibitors |journal=Biol Psychiatry |volume=59 |issue=3 |pages=203–10 |year=2006 |pmid=16165100 |doi=10.1016/j.biopsych.2005.06.027}}</ref> For example, the combination of bupropion and [[citalopram]] (Celexa) was observed to be more effective than switching to another antidepressant. The addition of bupropion to an SSRI (primarily fluoxetine or sertraline) resulted in a significant improvement in 70–80% of patients who had an incomplete response to the first-line antidepressant.<ref name="pmid9614595">{{cite journal |author=Spier SA |title=Use of bupropion with SRIs and venlafaxine |journal=Depression and |
According to several surveys, the [[Augmentation (psychiatry)|augmentation]] of a prescribed [[SSRI]] with bupropion is the preferred strategy among clinicians when the patient does not respond to the SSRI.<ref>For the most recent review, see: {{cite journal |author=Zisook S, Rush AJ, Haight BR, Clines DC, Rockett CB |title=Use of bupropion in combination with serotonin reuptake inhibitors |journal=Biol Psychiatry |volume=59 |issue=3 |pages=203–10 |year=2006 |pmid=16165100 |doi=10.1016/j.biopsych.2005.06.027}}</ref> For example, the combination of bupropion and [[citalopram]] (Celexa) was observed to be more effective than switching to another antidepressant. The addition of bupropion to an SSRI (primarily fluoxetine or sertraline) resulted in a significant improvement in 70–80% of patients who had an incomplete response to the first-line antidepressant.<ref name="pmid9614595">{{cite journal |author=Spier SA |title=Use of bupropion with SRIs and venlafaxine |journal=[[Depression and Anxiety]] |volume=7 |issue=2 |pages=73–5 |year=1998 |pmid=9614595|doi=10.1002/(SICI)1520-6394(1998)7:2<73::AID-DA4>3.0.CO;2-6}}</ref><ref name="pmid9164423">{{cite journal |author=Bodkin JA, Lasser RA, Wines JD, Gardner DM, Baldessarini RJ |title=Combining serotonin reuptake inhibitors and bupropion in partial responders to antidepressant monotherapy |journal=The Journal of clinical psychiatry |volume=58 |issue=4 |pages=137–45 |year=1997 |pmid=9164423 |doi=10.4088/JCP.v58n0401}}</ref> Bupropion improved ratings of "energy", which had decreased under the influence of the SSRI; also noted were improvements of mood and motivation, and some improvement of cognitive and sexual functions. Sleep quality and anxiety ratings in most cases were unchanged.<ref name="pmid9164423"/> In the STAR*D study, the patients who did not respond to [[citalopram]] (Celexa) were randomly assigned to augmentation by bupropion or [[buspirone]] (Buspar). Approximately 30% of subjects in both groups achieved a remission. However, bupropion augmentation gave better results based on the patients' self-ratings and was much better tolerated. The authors observed that "these findings reveal a consistently more favorable outcome with sustained-release bupropion than with buspirone augmentation of citalopram."<ref name="pmid16554526">{{cite journal |author=Trivedi MH, Fava M, Wisniewski SR, Thase ME, Quitkin F, Warden D, Ritz L, Nierenberg AA, Lebowitz BD, Biggs MM, Luther JF, Shores-Wilson K, Rush AJ |title=Medication augmentation after the failure of SSRIs for depression |journal=N. Engl. J. Med. |volume=354 |issue=12 |pages=1243–52 |year=2006 |pmid=16554526 |doi=10.1056/NEJMoa052964}}</ref> The same study indicated a possibility of higher remission rate when the non-responders to citalopram received bupropion augmentation instead of being switched to bupropion (30% vs. 20%).<ref name="pmid16554525">{{cite journal |author=Rush AJ, Trivedi MH, Wisniewski SR, Stewart JW, Nierenberg AA, Thase ME, Ritz L, Biggs MM, Warden D, Luther JF, Shores-Wilson K, Niederehe G, Fava M |title=Bupropion-SR, sertraline, or venlafaxine-XR after failure of SSRIs for depression |journal=N. Engl. J. Med. |volume=354 |issue=12 |pages=1231–42 |year=2006 |pmid=16554525 |doi=10.1056/NEJMoa052963}}</ref> |
||
===Anxiety=== |
===Anxiety=== |
||
Bupropion has shown some success in treating [[social anxiety disorder]]<ref name="bpsrsocial">{{cite journal |author=Emmanuel NP, Brawman-Mintzer O, Morton WA, Book SW, Johnson MR, Lorberbaum JP, Ballenger JC, Lydiard RB. |title=Bupropion-SR in treatment of social phobia |journal=Depression and Anxiety |volume=12 |issue=2 |pages=111–3 |year=2000 |pmid=11091936 |doi=10.1002/1520-6394(2000)12:2<111::AID-DA9>3.0.CO;2-3}}</ref> and [[anxiety]] |
Bupropion has shown some success in treating [[social anxiety disorder]]<ref name="bpsrsocial">{{cite journal |author=Emmanuel NP, Brawman-Mintzer O, Morton WA, Book SW, Johnson MR, Lorberbaum JP, Ballenger JC, Lydiard RB. |title=Bupropion-SR in treatment of social phobia |journal=Depression and Anxiety |volume=12 |issue=2 |pages=111–3 |year=2000 |pmid=11091936 |doi=10.1002/1520-6394(2000)12:2<111::AID-DA9>3.0.CO;2-3}}</ref> and [[anxiety]] combined with depression,<ref name="bpanxidepress">{{cite journal |author=Trivedi MH, Rush AJ, Carmody TJ, Donahue RM, Bolden-Watson C, Houser TL, Metz A.|title=Do bupropion SR and sertraline differ in their effects on anxiety in depressed patients? |journal=Journal of Clinical Psychiatry |volume=62 |issue=10 |pages=776–81 |year=2001 |pmid=11816866 |doi=10.4088/JCP.v62n1005}}</ref> but not [[panic disorder]] with [[agoraphobia]].<ref name="bppanic">{{cite journal |author=Sheehan DV, Davidson J, Manschreck T, Van Wyck Fleet J. |title=Lack of efficacy of a new antidepressant (bupropion) in the treatment of panic disorder with phobias |journal=Journal of Clinical Psychopharmacology |volume=3 |issue=1 |pages=28–31 |year=1983 |pmid=6403599}}</ref> Its [[anxiolytic]] potential has been compared to that of [[sertraline]]<ref name="bpanxidepress" /> and [[doxepin]].<ref name="bpdoxepin">{{cite journal |author=Feighner J, Hendrickson G, Miller L, Stern W.|title=Double-Blind Comparison of Doxepin Versus Bupropion in Outpatients with a Major Depressive Disorder |journal=Journal of Clinical Psychopharmacology |volume=6 |issue=1 |pages=27–32 |year=1986 |pmid=3081600}}</ref> However, it can cause agitation in some patients, especially at higher doses, and often increases anxiety, much like [[methylphenidate]].<ref name="bpsideeffect">{{cite journal |author=Settle EC. |title=Bupropion sustained release: side effect profile |journal=Journal of Clinical Psychopharmacology |volume=59 |issue=Supplement 4 |pages=32–6 |year=1998 |pmid=9554319}}</ref> |
||
===Smoking cessation=== |
===Smoking cessation=== |
||
Bupropion reduces the severity of [[nicotine]] cravings and [[withdrawal]] symptoms. |
Bupropion reduces the severity of [[nicotine]] cravings and [[withdrawal]] symptoms. |
||
After a seven-week treatment, 27% of subjects who received bupropion reported that an urge to smoke was a problem, versus 56% of those who received [[placebo]]. In the same study, 21% of the bupropion group reported mood swings, versus 32% of the placebo group.<ref name = "Tonnesen2003">{{cite journal | author = Tonnesen P, Tonstad S, Hjalmarson A, Lebargy F, Van Spiegel P I, Hider A, Sweet R, Townsend J| title = A multicentre, randomized, double-blind, placebo-controlled, 1-year study of bupropion SR for smoking cessation | journal = J Intern Med| volume = 254|issue = 2|pages = 184–192| year = 2003 |pmid=12859700 | doi = 10.1046/j.1365-2796.2003.01185.x}}</ref> The bupropion treatment course lasts for seven to twelve weeks, with the patient halting the use of tobacco about ten days into the course. Bupropion approximately doubles the chance of quitting smoking successfully after three months. One year after the treatment, the odds of sustaining smoking cessation are still 1.5 times higher in the bupropion group than in the placebo group.<ref>{{cite journal | author = Wu P, Wilson K, Dimoulas P, Mills E J | title = Effectiveness of smoking cessation therapies: a systematic review and meta-analysis | journal = BMC Public Health| volume = 6|pages = 300–315 | year = 2006 | pmid = 17156479 | pmc = 1764891 | doi = 10.1186/1471-2458-6-300}}</ref> The combination of bupropion and nicotine appears not to further increase the cessation rate. In a |
After a seven-week treatment, 27% of subjects who received bupropion reported that an urge to smoke was a problem, versus 56% of those who received [[placebo]]. In the same study, 21% of the bupropion group reported mood swings, versus 32% of the placebo group.<ref name = "Tonnesen2003">{{cite journal | author = Tonnesen P, Tonstad S, Hjalmarson A, Lebargy F, Van Spiegel P I, Hider A, Sweet R, Townsend J| title = A multicentre, randomized, double-blind, placebo-controlled, 1-year study of bupropion SR for smoking cessation | journal = J Intern Med| volume = 254|issue = 2|pages = 184–192| year = 2003 |pmid=12859700 | doi = 10.1046/j.1365-2796.2003.01185.x}}</ref> The bupropion treatment course lasts for seven to twelve weeks, with the patient halting the use of tobacco about ten days into the course. Bupropion approximately doubles the chance of quitting smoking successfully after three months. One year after the treatment, the odds of sustaining smoking cessation are still 1.5 times higher in the bupropion group than in the placebo group.<ref>{{cite journal | author = Wu P, Wilson K, Dimoulas P, Mills E J | title = Effectiveness of smoking cessation therapies: a systematic review and meta-analysis | journal = BMC Public Health| volume = 6|pages = 300–315 | year = 2006 | pmid = 17156479 | pmc = 1764891 | doi = 10.1186/1471-2458-6-300}}</ref> The combination of bupropion and nicotine appears not to further increase the cessation rate. In a single trial that compared bupropion and [[varenicline]] (Chantix) directly, varenicline showed superior efficacy: after one year, the rate of continuous abstinence was 10% for placebo, 15% for bupropion, and 23% for varenicline.<ref>{{cite journal | author = Jorenby D E, Hays J T, Rigotti N A, Azoulay S, Watsky E J, Williams K E, Billing C B, Gong J, Reeves K R| title = Efficacy of varenicline, an alpha4beta2 nicotinic acetylcholine receptor partial agonist, vs placebo or sustained-release bupropion for smoking cessation: a randomized controlled trial | journal = JAMA| volume = 296|issue = 1|pages = 56–63| year = 2006 |pmid=16820547 | doi = 10.1001/jama.296.1.56}}</ref> Bupropion slows the weight gain that often occurs in the first weeks after quitting smoking (after seven weeks, the placebo group had an average 2.7 kg increase in weight, versus 1.5 kg for the bupropion group). With time, however, this effect becomes negligible (after 26 weeks, both groups recorded an average 4.8 kg weight gain).<ref name = "Tonnesen2003"/> |
||
===Sexual dysfunction=== |
===Sexual dysfunction=== |
||
Bupropion is one of few antidepressants that |
Bupropion is one of few antidepressants that do not cause sexual dysfunction.<ref>{{cite journal |author=Serretti A, Chiesa A |title=Treatment-emergent sexual dysfunction related to antidepressants: a meta-analysis |journal=J Clin Psychopharmacol |volume=29 |issue=3 |pages=259–66 |year=2009 |pmid=19440080 |doi=10.1097/JCP.0b013e3181a5233f}}</ref> According to a survey of psychiatrists, it is the drug of choice for the treatment of [[SSRI]]-induced sexual dysfunction, although this is not an indication approved by the [[Food and Drug Administration (United States)|U.S. Food and Drug Administration]] (FDA). 36% of psychiatrists preferred switching patients with SSRI-induced sexual dysfunction to bupropion, and 43% favored the augmentation of the current medication with bupropion.<ref>{{cite journal | author =Dording CM, Mischoulon D, Petersen TJ, Kornbluh R, Gordon J, Nierenberg AA, Rosenbaum JE, Fava M.| title = The pharmacologic management of SSRI-induced side effects: a survey of psychiatrists| journal = Ann Clin Psychiatry| volume = 14|issue=3| pages = 143–7 | year = 2002|pmid=12585563 | doi =10.3109/10401230209147450}}</ref> There are studies demonstrating the efficacy of both approaches; improvement of the desire and orgasm components of sexual function were the most often noted. For the augmentation approach, the addition of at least 200 mg/day of bupropion to the SSRI regimen may be necessary to achieve an improvement since the addition of 150 mg/day of bupropion did not produce a statistically significant difference from placebo.<ref>{{cite journal | author = Walker PW, Cole JO, Gardner EA, Hughes AR, Johnston JA, Batey SR, Lineberry CG| title = Improvement in fluoxetine-associated sexual dysfunction in patients switched to bupropion| journal = J Clin Psychiatry| volume = 54 |issue=12| pages = 459–65 | year = 1993|pmid=8276736}}</ref><ref>{{cite journal | author = Dobkin RD, Menza M, Marin H, Allen LA, Rousso R, Leiblum SR| title = Bupropion improves sexual functioning in depressed minority women: an open-label switch study| journal = J Clin Psychiatry| volume = 26|issue=1| pages = 21–6 | year = 2006|pmid=16415700}}</ref><ref>{{cite journal | author = Masand PS, Ashton AK, Gupta S, Frank B| title = Sustained-release bupropion for selective serotonin reuptake inhibitor-induced sexual dysfunction: a randomized, double-blind, placebo-controlled, parallel-group study| journal = Am J Psychiatry | volume = 158|issue=5| pages = 805–807 | year = 2001|pmid=11329407 | doi = 10.1176/appi.ajp.158.5.805}}</ref><ref>{{cite journal | author =DeBattista C, Solvason B, Poirier J, Kendrick E, Loraas E|title = A placebo-controlled, randomized, double-blind study of adjunctive bupropion sustained release in the treatment of SSRI-induced sexual dysfunction| journal = J Clin Psychiatry | volume = 66|issue=7| pages = 844–8 | year = 2005 | pmid =16013899 | doi =10.4088/JCP.v66n0706}}</ref><ref>{{cite journal | author = Ashton AK, Rosen RC| title = Bupropion as an antidote for serotonin reuptake inhibitor-induced sexual dysfunction| journal = J Clin Psychiatry| volume = 59|issue=3| pages = 112–5 | year = 1998|pmid=9541153 | doi = 10.4088/JCP.v59n0304}}</ref><ref>{{cite journal | author =Clayton AH, Warnock JK, Kornstein SG, Pinkerton R, Sheldon-Keller A, McGarvey EL| title = A placebo-controlled trial of bupropion SR as an antidote for selective serotonin reuptake inhibitor-induced sexual dysfunction| journal = J Clin Psychiatry | volume = 65|issue=1| pages = 62–7 | year = 2004|pmid=14744170 | doi =10.4088/JCP.v65n0110}}</ref> |
||
Several studies have indicated that bupropion also relieves sexual dysfunction in people who do not have depression. In a mixed-gender double-blind study, 63% of subjects on a 12-week course of bupropion rated their condition as improved or much improved, versus 3% of subjects on placebo.<ref>{{cite journal | author =Crenshaw TL, Goldberg JP, Stern WC| title = Pharmacologic modification of psychosexual dysfunction| journal = J Sex Marital Ther| volume = 13|issue=4| pages = 239–52 | year = 1987|pmid=3121861| doi=10.1080/00926238708403896}}</ref> Two studies, one of which was placebo-controlled, demonstrated the efficacy of bupropion for women with [[hypoactive sexual desire disorder]],<ref>{{cite journal | author = Segraves RT, Croft H, Kavoussi R, Ascher JA, Batey SR, Foster VJ, Bolden-Watson C, Metz A| title = Bupropion sustained release (SR) for the treatment of hypoactive sexual desire disorder (HSDD) in nondepressed women| journal = J Sex Marital Ther| volume = 27|issue=3| pages = 303–16 | year = 2001|pmid=11354935| doi = 10.1080/009262301750257155}}</ref><ref>{{cite journal | author = Segraves RT, Clayton A, Croft H, Wolf A, Warnock J.| title =Bupropion sustained release for the treatment of hypoactive sexual desire disorder in premenopausal women| journal = J Clin Psychopharmacol| volume = 24|issue=3| pages = 339–42 | year = 2004|pmid=15118489 | doi = 10.1097/01.jcp.0000125686.20338.c1}}</ref> resulting in significant improvement of arousal, orgasm and overall satisfaction. Bupropion also showed promise as a treatment for sexual dysfunction caused by [[chemotherapy]] for breast cancer<ref>{{cite journal | author =Mathias C, Cardeal Mendes CM, Ponde de Sena E, Dias de Moraes E, Bastos C, Braghiroli MI, Nunez G, Athanazio R, Alban L, Moore HC, del Giglio A| title = An open-label, fixed-dose study of bupropion effect on sexual function scores in women treated for breast cancer| journal = Ann Oncol| volume = 17|issue=12| pages = 1792–6 | year = 20060|pmid=16980597| doi = 10.1093/annonc/mdl304}}</ref> and for orgasmic dysfunction.<ref>{{cite journal | author =Modell JG, May RS, Katholi CR| title =Effect of bupropion-SR on orgasmic dysfunction in nondepressed subjects: a pilot study| journal = J Sex Marital Ther| volume = 26|issue=3| pages = 231–40 | year = 2000|pmid=10929571| doi =10.1080/00926230050084623}}</ref> As with the treatment of SSRI-induced sexual disorder, a higher dose of bupropion (300 mg) may be necessary: a randomized study employing a lower dose (150 mg) failed to find a significant difference between bupropion, sexual therapy or combined treatment.<ref>{{cite journal | author = Cabello F| title = Effectiveness of the Treatment of Female Hypoactive Sexual Desire Disorder | journal = J Sex Research |
Several studies have indicated that bupropion also relieves sexual dysfunction in people who do not have depression. In a mixed-gender double-blind study, 63% of subjects on a 12-week course of bupropion rated their condition as improved or much improved, versus 3% of subjects on placebo.<ref>{{cite journal | author =Crenshaw TL, Goldberg JP, Stern WC| title = Pharmacologic modification of psychosexual dysfunction| journal = J Sex Marital Ther| volume = 13|issue=4| pages = 239–52 | year = 1987|pmid=3121861| doi=10.1080/00926238708403896}}</ref> Two studies, one of which was placebo-controlled, demonstrated the efficacy of bupropion for women with [[hypoactive sexual desire disorder]],<ref>{{cite journal | author = Segraves RT, Croft H, Kavoussi R, Ascher JA, Batey SR, Foster VJ, Bolden-Watson C, Metz A| title = Bupropion sustained release (SR) for the treatment of hypoactive sexual desire disorder (HSDD) in nondepressed women| journal = J Sex Marital Ther| volume = 27|issue=3| pages = 303–16 | year = 2001|pmid=11354935| doi = 10.1080/009262301750257155}}</ref><ref>{{cite journal | author = Segraves RT, Clayton A, Croft H, Wolf A, Warnock J.| title =Bupropion sustained release for the treatment of hypoactive sexual desire disorder in premenopausal women| journal = J Clin Psychopharmacol| volume = 24|issue=3| pages = 339–42 | year = 2004|pmid=15118489 | doi = 10.1097/01.jcp.0000125686.20338.c1}}</ref> resulting in significant improvement of arousal, orgasm and overall satisfaction. Bupropion also showed promise as a treatment for sexual dysfunction caused by [[chemotherapy]] for breast cancer<ref>{{cite journal | author =Mathias C, Cardeal Mendes CM, Ponde de Sena E, Dias de Moraes E, Bastos C, Braghiroli MI, Nunez G, Athanazio R, Alban L, Moore HC, del Giglio A| title = An open-label, fixed-dose study of bupropion effect on sexual function scores in women treated for breast cancer| journal = Ann Oncol| volume = 17|issue=12| pages = 1792–6 | year = 20060|pmid=16980597| doi = 10.1093/annonc/mdl304}}</ref> and for [[orgasmic dysfunction]].<ref>{{cite journal | author =Modell JG, May RS, Katholi CR| title =Effect of bupropion-SR on orgasmic dysfunction in nondepressed subjects: a pilot study| journal = J Sex Marital Ther| volume = 26|issue=3| pages = 231–40 | year = 2000|pmid=10929571| doi =10.1080/00926230050084623}}</ref> As with the treatment of SSRI-induced sexual disorder, a higher dose of bupropion (300 mg) may be necessary: a randomized study employing a lower dose (150 mg) failed to find a significant difference between bupropion, sexual therapy or combined treatment.<ref>{{cite journal | author = Cabello F| title = Effectiveness of the Treatment of Female Hypoactive Sexual Desire Disorder | journal = J Sex Research| year = 2006| accessdate = 2007-04-05 | url = http://www.findarticles.com/p/articles/mi_m2372/is_1_43/ai_n16102437}}</ref> Bupropion does not adversely affect any measures of sexual functioning in healthy men.<ref>{{cite journal | author =Labbate LA, Brodrick PS, Nelson RP, Lydiard RB, Arana GW| title =Effects of bupropion sustained-release on sexual functioning and nocturnal erections in healthy men| journal = J Clin Psychopharmacol| volume = 21|issue=1| pages = 99–103 | year = 2001|pmid=11199957| doi =10.1097/00004714-200102000-00018}}</ref> |
||
===Obesity=== |
===Obesity=== |
||
A recent [[meta-analysis]] of [[anti-obesity drug|anti-obesity medication]]s pooled the results of three double-blind, placebo-controlled trials of bupropion. It confirmed the efficacy of bupropion given at 400 mg per day for treating [[obesity]]. Over a period of 6 to 12 months, weight loss in the bupropion group (4.4 kg) was significantly greater than in the placebo group (1.7 kg). The same review found the differences in weight loss between bupropion and other established weight-loss medications, such as [[sibutramine]], [[orlistat]] and [[diethylpropion]], to be statistically |
A recent [[meta-analysis]] of [[anti-obesity drug|anti-obesity medication]]s pooled the results of three double-blind, placebo-controlled trials of bupropion. It confirmed the efficacy of bupropion given at 400 mg per day for treating [[obesity]]. Over a period of 6 to 12 months, weight loss in the bupropion group (4.4 kg) was significantly greater than in the placebo group (1.7 kg). The same review found the differences in weight loss between bupropion and other established weight-loss medications, such as [[sibutramine]], [[orlistat]] and [[diethylpropion]], not to be statistically significant.<ref>{{cite journal | author =Li Z, Maglione M, Tu W, Mojica W, Arterburn D, Shugarman LR, Hilton L, Suttorp M, Solomon V, Shekelle PG, Morton SC.| title = Meta-analysis: pharmacologic treatment of obesity| journal = Ann Intern Med| volume = 142|issue=7| pages = 532–46 | year = 2005|pmid=15809465 }}</ref> Combinations of bupropion and [[zonisamide]] ([[Empatic]]) and of bupropion and [[naltrexone]] ([[Contrave]]) are currently being studied for obesity.<ref name="pmid19885278">{{cite journal |author=Klonoff DC, Greenway F |title=Drugs in the pipeline for the obesity market |journal=J Diabetes Sci Technol |volume=2 |issue=5 |pages=913–8 |year=2008 |pmid=19885278 |pmc=2769782}}</ref> |
||
===Attention-deficit hyperactivity disorder=== |
===Attention-deficit hyperactivity disorder=== |
||
Although [[attention-deficit hyperactivity disorder]] (ADHD) is not an approved indication, bupropion was found to be effective for adult ADHD.<ref name="pmid15820237">{{cite journal |author=Wilens TE, Haight BR, Horrigan JP, Hudziak JJ, Rosenthal NE, Connor DF, Hampton KD, Richard NE, Modell JG |title=Bupropion XL in adults with attention-deficit/hyperactivity disorder: a randomized, placebo-controlled study |journal=Biol. Psychiatry |volume=57 |issue=7 |pages=793–801 |year=2005 |pmid=15820237 |doi=10.1016/j.biopsych.2005.01.027}}</ref> There have been many positive case studies and other uncontrolled clinical studies of bupropion for ADHD in minors.<ref name="pmid9554326">For the review, see: {{cite journal |author=Cantwell DP |title=ADHD through the life span: the role of bupropion in treatment |journal=The Journal of clinical psychiatry |volume=59 Suppl 4 |
Although [[attention-deficit hyperactivity disorder]] (ADHD) is not an approved indication, bupropion was found to be effective for adult ADHD.<ref name="pmid15820237">{{cite journal |author=Wilens TE, Haight BR, Horrigan JP, Hudziak JJ, Rosenthal NE, Connor DF, Hampton KD, Richard NE, Modell JG |title=Bupropion XL in adults with attention-deficit/hyperactivity disorder: a randomized, placebo-controlled study |journal=Biol. Psychiatry |volume=57 |issue=7 |pages=793–801 |year=2005 |pmid=15820237 |doi=10.1016/j.biopsych.2005.01.027}}</ref> There have been many positive case studies and other uncontrolled clinical studies of bupropion for ADHD in minors.<ref name="pmid9554326">For the review, see: {{cite journal |author=Cantwell DP |title=ADHD through the life span: the role of bupropion in treatment |journal=The Journal of clinical psychiatry |volume=59 Suppl 4 |pages=92–4 |year=1998 |pmid=9554326}}</ref> However, in the largest to date double-blind study, which was conducted by GlaxoSmithKline, the results were inconclusive. Aggression and hyperactivity as rated by the children's teachers were significantly improved in comparison to placebo; in contrast, parents and clinicians could not distinguish between the effects of bupropion and placebo.<ref name="pmid9554326"/> The 2007 guideline on the ADHD treatment from American Academy of Child and Adolescent Psychiatry notes that the evidence for bupropion is "far weaker" than for the FDA-approved treatments. Its effect may also be "considerably less than of the approved agents... Thus it may be prudent for the clinician to recommend a trial of behavior therapy at this point, before moving to these second-line agents."<ref>{{cite web |url=http://www.aacap.org/galleries/practiceparameters/jaacap_adhd_2007.pdf |title= Practice Parameter For The Assessment And Treatment Of Children And Adolescents With Attention-Deficit/Hyperactivity Disorder |accessdate=2011-01-25 |author= Steven Pliszka ''et al.'' |year= 2007 |format=PDF |publisher=American Academy of Child and Adolescent Psychiatry |page=16}}</ref> Similarly, the 2006 guideline from the Texas Department of State Health Services recommends considering bupropion or a [[tricyclic antidepressant]] as a fourth-line treatment after trying two different stimulants and [[atomoxetine]] (Strattera).<ref>{{cite web |url=http://www.dshs.state.tx.us/mhprograms/adhdpage.shtm |title= The Texas Children's Medication Algorithm Project: attention-deficit/hyperactivity disorder|accessdate=2011-01-25 |author= Pliszka SR ''et al.'' |year=2006 |publisher=Texas Department of State Health Services }}</ref><ref>{{cite web |url=http://www.dshs.state.tx.us/mhprograms/ADHD_Algo_Schematics_With_NO_Sig_Co_Dis.pdf |title= Algorithm Stages Flowsheets. ADHD with no significant comorbidity algorithm |accessdate=2011-01-25 |month=May | year=2006 |format=PDF|publisher=Texas Department of State Health Services |archiveurl=http://web.archive.org/web/20060822172713/http://www.dshs.state.tx.us/mhprograms/ADHD_Algo_Schematics_With_NO_Sig_Co_Dis.pdf|archivedate=August 22, 2006}}</ref> |
||
A study of prophylactic bupropion for the prevention of smoking among teenagers with ADHD yielded unexpected results. The teenagers taking bupropion were two times more likely (close to statistical significance) to begin smoking than the teenagers in the placebo group. However, because significance was not reached, this difference can be attributed entirely to chance |
A study of prophylactic bupropion for the prevention of smoking among teenagers with ADHD yielded unexpected results. The teenagers taking bupropion were two times more likely (close to statistical significance) to begin smoking than the teenagers in the placebo group. However, because significance was not reached, this difference can be attributed entirely to chance. At the same time, the sub-group of patients taking stimulants in addition to bupropion or placebo had a five times lower risk of smoking initiation.<ref name="pmid17685748">{{cite journal |author=Monuteaux MC, Spencer TJ, Faraone SV, Wilson AM, Biederman J |title=A randomized, placebo-controlled clinical trial of bupropion for the prevention of smoking in children and adolescents with attention-deficit/hyperactivity disorder |journal=The Journal of clinical psychiatry |volume=68 |issue=7 |pages=1094–101 |year=2007 |pmid=17685748 |doi=10.4088/JCP.v68n0718}}</ref> |
||
===Other uses=== |
===Other uses=== |
||
Bupropion improves mood by increasing the concentration of [[dopamine]] in emotional brain regions. The mechanism may improve the negative mood associated with methamphetamine withdrawal. Though it has demonstrated no effectiveness in the treatment of cocaine dependence, there is weak evidence that it may be useful in methamphetamine dependence.<ref name=Kampman>{{cite journal |author=Kampman KM |title=The search for medications to treat stimulant dependence |journal=Addict Sci Clin Pract |volume=4 |issue=2 |pages=28–35 |year=2008 |pmid=18497715 |pmc=2797110}}</ref> |
|||
| ⚫ | Bupropion was approved by the [[Food and Drug Administration (United States)|U.S. Food and Drug Administration]] (FDA)<ref name="pmid17328102">{{cite journal |
||
| ⚫ | Bupropion was approved by the [[Food and Drug Administration (United States)|U.S. Food and Drug Administration]] (FDA)<ref name="pmid17328102">{{cite journal |title=First drug for seasonal depression |journal=FDA Consumer |volume=40 |issue=5 |page=7 |year=2006 |pmid=17328102 | url = http://permanent.access.gpo.gov/lps1609/www.fda.gov/fdac/departs/2006/506_upd.html#depression | publisher = [[Food and Drug Administration (United States)|U.S. Food and Drug Administration]] (FDA) }}</ref> for the prevention of [[seasonal affective disorder]].<ref name="pmid16271314">{{cite journal |author=Modell JG, Rosenthal NE, Harriett AE, Krishen A, Asgharian A, Foster VJ, Metz A, Rockett CB, Wightman DS |title=Seasonal affective disorder and its prevention by anticipatory treatment with bupropion XL |journal=Biol Psychiatry |volume=58 |issue=8 |pages=658–67 |year=2005 |pmid=16271314 |doi=10.1016/j.biopsych.2005.07.021}}</ref> According to several case studies and a pilot study, bupropion lowers the level of an inflammatory mediator [[Tumor necrosis factor-alpha|TNF-alpha]]<ref name="pmid14552325"/> and may be useful in autoinflammatory conditions such as [[Crohn's disease]]<ref name="pmid14552325">{{cite journal |author=Kane S, Altschuler EL, Kast RE |title=Crohn's disease remission on bupropion |journal=Gastroenterology |volume=125 |issue=4 |page=1290 |year=2003 |pmid=14552325 |doi= 10.1016/j.gastro.2003.02.004}}</ref><ref name="pmid11706830">{{cite journal |author=Kast RE, Altschuler EL |title=Remission of Crohn's disease on bupropion |journal=Gastroenterology |volume=121 |issue=5 |pages=1260–1 |year=2001 |pmid=11706830 |doi= 10.1053/gast.2001.29467}}</ref> and [[psoriasis]].<ref name="pmid12883127">{{cite journal |author=Altschuler EL, Kast RE |title=Bupropion in psoriasis and atopic dermatitis: decreased tumor necrosis factor-alpha? |journal=Psychosom Med |volume=65 |issue=4 |page=719 |year=2003 |pmid=12883127 |doi= 10.1097/01.PSY.0000073874.55003.EE}}</ref><ref name="pmid12271115">{{cite journal |author=Modell JG, Boyce S, Taylor E, Katholi C |title=Treatment of atopic dermatitis and psoriasis vulgaris with bupropion-SR: a pilot study |journal=Psychosom Med |volume=64 |issue=5 |pages=835–40 |year=2002 |pmid=12271115 |doi= 10.1097/01.PSY.0000021954.59258.9B}}</ref> |
||
| ⚫ | No properly controlled double-blind studies of bupropion for [[Parkinson's disease]] have been conducted. A small 1984 study funded by bupropion's manufacturer found that addition of bupropion to [[carbidopa]] or [[levodopa]] improved Parkinson's symptoms in ten out of twenty patients; however, the side effects, particularly nausea and vomiting, were frequent.<ref name="pmid6431314">{{cite journal |author=Goetz CG, Tanner CM, Klawans HL |title=Bupropion in Parkinson's disease |journal=Neurology |volume=34 |issue=8 |pages=1092–4 |year=1984 |pmid=6431314 |
||
| ⚫ | No properly controlled double-blind studies of bupropion for [[Parkinson's disease]] have been conducted. A small 1984 study funded by bupropion's manufacturer found that addition of bupropion to [[carbidopa]] or [[levodopa]] improved Parkinson's symptoms in ten out of twenty patients; however, the side effects, particularly nausea and vomiting, were frequent.<ref name="pmid6431314">{{cite journal |author=Goetz CG, Tanner CM, Klawans HL |title=Bupropion in Parkinson's disease |journal=Neurology |volume=34 |issue=8 |pages=1092–4 |year=1984 |pmid=6431314}}</ref> The [[American Psychiatric Association]] notes that, "there is no evidence favoring any particular antidepressant medication from the standpoint of therapeutic efficacy in patients with Parkinson's disease complicated by major depressive disorder".<ref>{{cite web |url=http://www.psychiatryonline.com/pracGuide/PracticePDFs/PG_Depression3rdEd.pdf |title=Practice guideline for the treatment of patients with major depressive disorder. Third edition |accessdate=2011-01-25 |author=American Psychiatric Association |year= 2000|format= PDF|page= 37}}</ref> |
||
| ⚫ | There is considerable disagreement regarding whether it is useful to add an antidepressant, including bupropion, to a [[mood stabilizer]] in patients with [[bipolar depression]].<ref name="pmid17392295">For the review indicating that antidepressants are not better than placebo, see: {{cite journal |author=Sachs GS, Nierenberg AA, Calabrese JR, Marangell LB, Wisniewski SR, Gyulai L, Friedman ES, Bowden CL, Fossey MD, Ostacher MJ, Ketter TA, Patel J, Hauser P, Rapport D, Martinez JM, Allen MH, Miklowitz DJ, Otto MW, Dennehy EB, Thase ME |title=Effectiveness of adjunctive antidepressant treatment for bipolar depression |journal=N. Engl. J. Med. |volume=356 |issue=17 |pages=1711–22 |year=2007 |pmid=17392295 |doi=10.1056/NEJMoa064135}}</ref><ref name="pmid15337640">For the review in favor of the antidepressant use, see: {{cite journal |author=Gijsman HJ, Geddes JR, Rendell JM, Nolen WA, Goodwin GM |title=Antidepressants for bipolar depression: a systematic review of randomized, controlled trials |journal=The American |
||
| ⚫ | There is considerable disagreement regarding whether it is useful to add an antidepressant, including bupropion, to a [[mood stabilizer]] in patients with [[bipolar depression]].<ref name="pmid17392295">For the review indicating that antidepressants are not better than placebo, see: {{cite journal |author=Sachs GS, Nierenberg AA, Calabrese JR, Marangell LB, Wisniewski SR, Gyulai L, Friedman ES, Bowden CL, Fossey MD, Ostacher MJ, Ketter TA, Patel J, Hauser P, Rapport D, Martinez JM, Allen MH, Miklowitz DJ, Otto MW, Dennehy EB, Thase ME |title=Effectiveness of adjunctive antidepressant treatment for bipolar depression |journal=N. Engl. J. Med. |volume=356 |issue=17 |pages=1711–22 |year=2007 |pmid=17392295 |doi=10.1056/NEJMoa064135}}</ref><ref name="pmid15337640">For the review in favor of the antidepressant use, see: {{cite journal |author=Gijsman HJ, Geddes JR, Rendell JM, Nolen WA, Goodwin GM |title=Antidepressants for bipolar depression: a systematic review of randomized, controlled trials |journal=The American Journal of Psychiatry |volume=161 |issue=9 |pages=1537–47 |year=2004 |pmid=15337640 |doi=10.1176/appi.ajp.161.9.1537}}</ref><ref name="pmid17156158">For the guidelines recommending the use of bupropion with a mood stabilizer, see: {{cite journal |author=Yatham LN, Kennedy SH, O'Donovan C, Parikh SV, MacQueen G, McIntyre RS, Sharma V, Beaulieu S |title=Canadian Network for Mood and Anxiety Treatments (CANMAT) guidelines for the management of patients with bipolar disorder: update 2007 |journal=Bipolar Disord |volume=8 |issue=6 |pages=721–39 |year=2006 |pmid=17156158 |doi=10.1111/j.1399-5618.2006.00432.x |last9=Guidelines Group}}</ref> |
||
| ⚫ | Bupropion, similarly to other antidepressants, is not efficacious in chronic low back pain.<ref>{{cite journal |author=Katz J, Pennella-Vaughan J, Hetzel RD, Kanazi GE, Dworkin RH |title=A randomized, placebo-controlled trial of bupropion sustained release in chronic low back pain |journal=J Pain |volume=6 |issue=10 |pages=656–61 |year=2005 |
||
| ⚫ | Bupropion, similarly to other antidepressants, is not efficacious in chronic low back pain.<ref>{{cite journal |author=Katz J, Pennella-Vaughan J, Hetzel RD, Kanazi GE, Dworkin RH |title=A randomized, placebo-controlled trial of bupropion sustained release in chronic low back pain |journal=J Pain |volume=6 |issue=10 |pages=656–61 |year=2005 |pmid=16202958 |doi=10.1016/j.jpain.2005.05.002}}</ref><ref>{{cite journal |author=Urquhart DM, Hoving JL, Assendelft WW, Roland M, van Tulder MW |title=Cochrane Database of Systematic Reviews |journal=Cochrane Database Syst Rev |issue=1 |pages=CD001703 |year=2008 |pmid=18253994 |doi=10.1002/14651858.CD001703.pub3 |chapter=Antidepressants for non-specific low back pain |editor1-last=Urquhart |editor1-first=Donna M}}</ref> However, it has been found to have surprisingly high efficacy in the treatment of neuropathic pain.<ref name="pmid10741812">{{cite journal | author = Semenchuk MR, Davis B | title = Efficacy of sustained-release bupropion in neuropathic pain: an open-label study | journal = The Clinical Journal of Pain | volume = 16 | issue = 1 | pages = 6–11 | year = 2000 | pmid = 10741812 | doi = 10.1097/00002508-200003000-00002}}</ref><ref name="pmid11706096">{{cite journal | author = Semenchuk MR, Sherman S, Davis B | title = Double-blind, randomized trial of bupropion SR for the treatment of neuropathic pain | journal = Neurology | volume = 57 | issue = 9 | pages = 1583–8 | year = 2001| pmid = 11706096}}</ref><ref name="pmid20185402">{{cite journal | author = Shah TH, Moradimehr A | title = Bupropion for the Treatment of Neuropathic Pain | journal = The American Journal of Hospice & Palliative Care | volume = 27| issue = 5| pages = 333–6| year = 2010 | pmid = 20185402 | doi = 10.1177/1049909110361229}}</ref> |
||
==Contraindications== |
==Contraindications== |
||
| Line 107: | Line 109: | ||
==Adverse effects== |
==Adverse effects== |
||
[[Seizure]] is the most controversial side effect of bupropion, and was responsible for its initial withdrawal from the market. The risk of seizure is highly dose-dependent: 0.1% at 100–300 mg of bupropion, 0.4% at 300–450 mg, and 2% at 600 mg. For comparison, the incidence of the first unprovoked seizure in the general population is 0. |
[[Seizure]] is the most controversial side effect of bupropion, and was responsible for its initial withdrawal from the market. The risk of seizure is highly dose-dependent: 0.1% at 100–300 mg of bupropion, 0.4% at 300–450 mg, and 2% at 600 mg.{{cn|date=November 2012}} For comparison, the incidence of the first unprovoked seizure in the general population is 0.07 to 0.09%. The risk of seizure for other antidepressants is as follows: 0.1–0.6% for [[imipramine]], depending on dosage; 0–0.06% for [[amitriptyline]], depending on dosage; 0.5% for [[clomipramine]]; 0.4% for [[maprotiline]]; and 0.2% for [[fluoxetine]] and [[fluvoxamine]].<ref>{{cite journal |author=Pisani F, Oteri G, Costa C, Di Raimondo G, Di Perri R |title=Effects of Psychotropic Drugs on Seizure Threshold |journal=Drug Safety|volume=25 |issue=2 |pages=91–110 |year=2002 |doi=10.2165/00002018-200225020-00004 |pmid=11888352}}</ref> Experiments on mice indicate that increased susceptibility to seizure is a general side effect of chronically using antidepressants that inhibit norepinephrine transporters, such as [[imipramine]], [[desipramine]] and [[reboxetine]].<ref>{{cite journal |author=Ahern TH, Javors MA, Eagles DA, Martillotti J, Mitchell HA, Liles LC, Weinshenker D |title=The effects of chronic norepinephrine transporter inactivation on seizure susceptibility in mice |journal=Neuropsychopharmacology |volume=31 |issue=4 |pages=730–8 |year=2006 |pmid=16052243 |doi=10.1038/sj.npp.1300847}}</ref> Clinical depression itself was reported to increase the occurrence of seizures two-to-sevenfold compared with the general population; in this light, the above statistics could indicate that low to moderate doses of antidepressants, including bupropion, may actually have an anti-convulsive action.<ref>{{cite journal |author=Alper K, Schwartz KA, Kolts RL, Khan A |title=Seizure Incidence in Psychopharmacological Clinical Trials: An Analysis of Food and Drug Administration (FDA) Summary Basis of Approval Reports |journal=Biol Psychiatry |volume=62 |issue=4 |pages=345–54 |year=2007 |pmid=17223086 |doi=10.1016/j.biopsych.2006.09.023}}</ref> |
||
The prescribing information notes that [[hypertension]], sometimes severe, was observed in some patients, both with and without pre-existing hypertension. The frequency of this adverse effect was under 1% and not significantly higher than that found with placebo.<ref name="WellbutrinPI"/> In a group of cardiac patients with depression, high doses of bupropion (400–500 mg/day) caused a rise in [[supine position|supine]] blood pressure but had no effect on pulse rate.<ref name="pmid1900980">{{cite journal |author=Roose SP, Dalack GW, Glassman AH, Woodring S, Walsh BT, Giardina EG |title=Cardiovascular effects of bupropion in depressed patients with heart disease |journal=Am J Psychiatry |volume=148 |issue=4 |pages=512–6 |year=1991 |pmid=1900980 |
The prescribing information notes that [[hypertension]], sometimes severe, was observed in some patients, both with and without pre-existing hypertension. The frequency of this adverse effect was under 1% and not significantly higher than that found with placebo.<ref name="WellbutrinPI"/> In a group of cardiac patients with depression, high doses of bupropion (400–500 mg/day) caused a rise in [[supine position|supine]] blood pressure but had no effect on pulse rate.<ref name="pmid1900980">{{cite journal |author=Roose SP, Dalack GW, Glassman AH, Woodring S, Walsh BT, Giardina EG |title=Cardiovascular effects of bupropion in depressed patients with heart disease |journal=Am J Psychiatry |volume=148 |issue=4 |pages=512–6 |year=1991 |pmid=1900980}}</ref> No statistically significant changes in blood pressure or heart rate occurred in patients with or without heart conditions at a lower dose of 300 mg/day.<ref name="pmid12109935">{{cite journal |author=Aubin HJ |title=Tolerability and safety of sustained-release bupropion in the management of smoking cessation |journal=Drugs |volume=62 Suppl 2 |pages=45–52 |year=2002 |pmid=12109935 |doi=10.2165/00003495-200262002-00005}}</ref> In a study of bupropion for [[ADHD]], a rise of [[systole (medicine)|systolic]] blood pressure by 6 mm Hg and of heart rate by 7 beats per minute (both statistically significant) were observed.<ref name="pmid15705013">{{cite journal |author=Wilens TE, Hammerness PG, Biederman J, Kwon A, Spencer TJ, Clark S, Scott M, Podolski A, Ditterline JW, Morris MC, Moore H |title=Blood pressure changes associated with medication treatment of adults with attention-deficit/hyperactivity disorder |journal=J Clin Psychiatry |volume=66 |issue=2 |pages=253–9 |year=2005 |pmid=15705013 |doi=10.4088/JCP.v66n0215}}</ref> A study of smokers hospitalized for heart disease found a 1.5-fold increase (close to being statistically significant) in subsequent cardiovascular events in the bupropion group, compared with the placebo group, but found no difference in blood pressure.<ref name="pmid17145253">{{cite journal |author=Rigotti NA, Thorndike AN, Regan S, McKool K, Pasternak RC, Chang Y, Swartz S, Torres-Finnerty N, Emmons KM, Singer DE |title=Bupropion for smokers hospitalized with acute cardiovascular disease |journal=Am J Med |volume=119 |issue=12 |pages=1080–7 |year=2006 |pmid=17145253 |doi=10.1016/j.amjmed.2006.04.024}}</ref> Although the cardiovascular side effects of bupropion appear to be mild, it cannot be recommended for patients with heart disease, since the safety comparison with SSRIs (such as [[sertraline]] and [[fluoxetine]], which may have a preventative effect after a [[myocardial infarction]]<ref name="pmid17112303">{{cite journal |author=van Melle JP, de Jonge P, van den Berg MP, Pot HJ, van Veldhuisen DJ |title=Treatment of depression in acute coronary syndromes with selective serotonin reuptake inhibitors |journal=Drugs |volume=66 |issue=16 |pages=2095–107 |year=2006 |pmid=17112303 |doi=10.2165/00003495-200666160-00005}}</ref>) is not in its favor. |
||
In the UK, more than 7,600 reports of suspected adverse reactions were collected in the first two years after bupropion's approval by the [[Medicines and Healthcare products Regulatory Agency|MHRA]] as part of the [[Yellow Card Scheme]], which monitored side effects. Approximately 540,000 people were treated with bupropion for smoking cessation during that period. The MHRA received 60 reports of "''suspected'' [emphasis MHRA's] adverse reactions to Zyban which had a fatal outcome". The agency concluded that "in the majority of cases the individual's underlying condition may provide an alternative explanation."<ref name=MHRA>{{cite web | title = Zyban (bupropion hydrochloride) |
In the UK, more than 7,600 reports of suspected adverse reactions were collected in the first two years after bupropion's approval by the [[Medicines and Healthcare products Regulatory Agency|MHRA]] as part of the [[Yellow Card Scheme]], which monitored side effects. Approximately 540,000 people were treated with bupropion for smoking cessation during that period. The MHRA received 60 reports of "''suspected'' [emphasis MHRA's] adverse reactions to Zyban which had a fatal outcome". The agency concluded that "in the majority of cases the individual's underlying condition may provide an alternative explanation."<ref name=MHRA>{{cite web | title = Zyban (bupropion hydrochloride) – safety update | publisher = [[Medicines and Healthcare products Regulatory Agency]] | date = 2002-07-24 | accessdate = 2006-10-07 | url = http://www.mhra.gov.uk/home/idcplg?IdcService=GET_FILE&dID=2556&noSaveAs=0&Rendition=WEB| archiveurl = http://web.archive.org/web/20070928004349/http://www.mhra.gov.uk/home/idcplg?IdcService=GET_FILE&dID=2556&noSaveAs=0&Rendition=WEB| archivedate = September 28, 2007}}</ref> This is consistent with a large, 9,300-patient safety study that showed that the mortality of smokers taking bupropion is not higher than the natural mortality of smokers of the same age.<ref name=Hubbard>{{cite journal | author = Hubbard R, Lewis S, West J, Smith C, Godfrey C, Smeeth L, Farrington P, Britton J | title = Bupropion and the risk of sudden death: a self-controlled case-series analysis using The Health Improvement Network | journal = Thorax | volume = 60 | issue = 10 | pages = 848–50 | year = 2005 | pmid = 16055620 | doi = 10.1136/thx.2005.041798 | pmc = 1747199|url=http://thorax.bmj.com/content/60/10/848.full|format=Free full text}}</ref> |
||
Other isolated adverse |
Other isolated adverse effects have been reported. Three cases of liver toxicity have been described,<ref name="pmid11785718">For the most recent report, see: {{cite journal |author=Alvaro D, Onetti-Muda A, Moscatelli R, Atili AF |title=Acute cholestatic hepatitis induced by bupropion prescribed as pharmacological support to stop smoking. A case report |journal=Digestive and liver disease|volume=33 |issue=8 |pages=703–6 |year=2001 |pmid=11785718 |doi=10.1016/S1590-8658(01)80049-9}}</ref> a very low incidence given the widespread use of the drug. A single case of clitoral priapism ([[clitorism]]) has been reported in the literature.<ref>{{cite journal | author = Levenson JL | title = Priapism associated with bupropion treatment | journal = Am J Psychiatry | volume = 152 | issue = 5 | page = 813 | year = 1995 | pmid = 7726332}}</ref> |
||
The common adverse effects associated with 12-hour sustained-release bupropion (with the greatest difference from placebo) are dry mouth, nausea, insomnia, tremor, excessive sweating and [[tinnitus]]. Those that most often resulted in interruption of the treatment in the same trial were rash (2.4%) and nausea (0.8%).<ref name="WellbutrinPI"/> |
The common adverse effects associated with 12-hour sustained-release bupropion (with the greatest difference from placebo) are dry mouth, nausea, insomnia, tremor, excessive sweating and [[tinnitus]]. Those that most often resulted in interruption of the treatment in the same trial were rash (2.4%) and nausea (0.8%).<ref name="WellbutrinPI"/> |
||
| Line 124: | Line 126: | ||
Suicidal behavior is less likely when bupropion is prescribed for smoking cessation. According to a 2007 [[Cochrane Library|Cochrane Database]] review, there have been four suicides per one million prescriptions and one case of suicidal ideation per ten thousand prescriptions of bupropion for smoking cessation in the UK. The review concludes, "Although some suicides and deaths while taking bupropion have been reported, thus far there is insufficient evidence to suggest they were caused by bupropion."<ref>{{cite journal |author = Hughes JR, Stead LF, Lancaster T|title = Cochrane Database of Systematic Reviews |journal = Cochrane Database Syst Rev. |volume=24 |issue=1|pages=CD000031 |year=2007 |pmid=17253443|doi = 10.1002/14651858.CD000031.pub3 |chapter = Antidepressants for smoking cessation |editor1-last = Hughes |editor1-first = John R}}</ref> |
Suicidal behavior is less likely when bupropion is prescribed for smoking cessation. According to a 2007 [[Cochrane Library|Cochrane Database]] review, there have been four suicides per one million prescriptions and one case of suicidal ideation per ten thousand prescriptions of bupropion for smoking cessation in the UK. The review concludes, "Although some suicides and deaths while taking bupropion have been reported, thus far there is insufficient evidence to suggest they were caused by bupropion."<ref>{{cite journal |author = Hughes JR, Stead LF, Lancaster T|title = Cochrane Database of Systematic Reviews |journal = Cochrane Database Syst Rev. |volume=24 |issue=1|pages=CD000031 |year=2007 |pmid=17253443|doi = 10.1002/14651858.CD000031.pub3 |chapter = Antidepressants for smoking cessation |editor1-last = Hughes |editor1-first = John R}}</ref> |
||
Two years later, the FDA issued a health advisory, which warned that the prescription of bupropion and [[varenicline]] for smoking cessation has been associated with reports about unusual behaviour changes, agitation and hostility. Some patients have become depressed or have had their depression worsen, have had thoughts about suicide or dying, or have attempted suicide.<ref name=warning>{{cite web |
Two years later, the FDA issued a health advisory, which warned that the prescription of bupropion and [[varenicline]] for smoking cessation has been associated with reports about unusual behaviour changes, agitation and hostility. Some patients have become depressed or have had their depression worsen, have had thoughts about suicide or dying, or have attempted suicide.<ref name=warning>{{cite web | title = Public Health Advisory: FDA Requires New Boxed Warnings for the Smoking Cessation Drugs Chantix and Zyban | date = 2009-07-01 |accessdate = 2009-07-03 | url = http://www.fda.gov/Drugs/DrugSafety/PostmarketDrugSafetyInformationforPatientsandProviders/DrugSafetyInformationforHeathcareProfessionals/PublicHealthAdvisories/ucm169988.htm | publisher = [[Food and Drug Administration (United States)|U.S. Food and Drug Administration]] (FDA) }}</ref><ref name=warning2>{{cite web | title = Information for Healthcare Professionals: Varenicline (marketed as Chantix) and Bupropion (marketed as Zyban, Wellbutrin, and generics) | date = 2009-07-01 |accessdate = 2009-07-03 | url = http://www.fda.gov/Drugs/DrugSafety/PostmarketDrugSafetyInformationforPatientsandProviders/DrugSafetyInformationforHeathcareProfessionals/ucm169986.htm | publisher = [[Food and Drug Administration (United States)|U.S. Food and Drug Administration]] (FDA) }}</ref> This advisory was based on the postmarket review of anti-smoking products, which identified 153 reports of the suicidal adverse events for varenicline over the first year it was marketed and 75 reports for bupropion over ten years. No clear association with suicidality was identified for [[nicotine patch]] products.<ref>{{cite journal |title = The Smoking Cessation Aids Varenicline (Marketed as Chantix) And Bupropion (Marketed as Zyban and Generics) Suicidal Ideation and Behavior |journal = Drug Safety Newsletter |volume=2 |issue=1|pages=1–4 |year=2009 | publisher=[[Food and Drug Administration (United States)|U.S. Food and Drug Administration]] (FDA) | url = http://www.fda.gov/downloads/Drugs/DrugSafety/DrugSafetyNewsletter/UCM107318.pdf|format=PDF}}</ref> |
||
[[Mania]] is one of the rare side effects of bupropion. The prescribing information notes that "it is generally believed (though not established in controlled trials)" that, should an episode of depression actually be the first presentation of [[bipolar disorder]], treating it with antidepressants, including bupropion, may precipitate a manic episode.<ref name="WellbutrinPI"/> More recent data indicate that the addition of newer antidepressants, including bupropion, to a [[mood stabilizer]] does not cause the switch to mania more often than the addition of placebo.<ref name="pmid15337640"/> Moreover, when added to a mood stabilizer, bupropion and sertraline had a twice lower switch risk than venlafaxine.<ref name="pmid16880481">{{cite journal |author=Post RM, Altshuler LL, Leverich GS, Frye MA, Nolen WA, Kupka RW, Suppes T, McElroy S, Keck PE, Denicoff KD, Grunze H, Walden J, Kitchen CM, Mintz J |title=Mood switch in bipolar depression: comparison of adjunctive venlafaxine, bupropion and sertraline |journal=Br J Psychiatry |volume=189 |issue= 2|pages=124–31 |year=2006 |pmid=16880481 |doi=10.1192/bjp.bp.105.013045}}</ref> |
[[Mania]] is one of the rare side effects of bupropion. The prescribing information notes that "it is generally believed (though not established in controlled trials)" that, should an episode of depression actually be the first presentation of [[bipolar disorder]], treating it with antidepressants, including bupropion, may precipitate a manic episode.<ref name="WellbutrinPI"/> More recent data indicate that the addition of newer antidepressants, including bupropion, to a [[mood stabilizer]] does not cause the switch to mania more often than the addition of placebo.<ref name="pmid15337640"/> Moreover, when added to a mood stabilizer, bupropion and sertraline had a twice lower switch risk than venlafaxine.<ref name="pmid16880481">{{cite journal |author=Post RM, Altshuler LL, Leverich GS, Frye MA, Nolen WA, Kupka RW, Suppes T, McElroy S, Keck PE, Denicoff KD, Grunze H, Walden J, Kitchen CM, Mintz J |title=Mood switch in bipolar depression: comparison of adjunctive venlafaxine, bupropion and sertraline |journal=Br J Psychiatry |volume=189 |issue= 2|pages=124–31 |year=2006 |pmid=16880481 |doi=10.1192/bjp.bp.105.013045}}</ref> |
||
| Line 130: | Line 132: | ||
Like many of the antidepressants, bupropion is associated with the potential risk for causing anxiety as a side effect. How common or severe this anxiety is remains to be fully discovered. Some studies show that it has similar anxiety side effects to the SSRI antidepressants, while others show an elevated risk for [[akathisia]], or extreme anxiety, as high as 13.9%, or three times that from sertraline.<ref>{{cite web |url=http://healthlifeandstuff.com/2010/02/wellbutrin-and-anxiety-concerns/ |title=Wellbutrin – Anxiety Treatment Concerns |publisher=Health and Life }}</ref> |
Like many of the antidepressants, bupropion is associated with the potential risk for causing anxiety as a side effect. How common or severe this anxiety is remains to be fully discovered. Some studies show that it has similar anxiety side effects to the SSRI antidepressants, while others show an elevated risk for [[akathisia]], or extreme anxiety, as high as 13.9%, or three times that from sertraline.<ref>{{cite web |url=http://healthlifeandstuff.com/2010/02/wellbutrin-and-anxiety-concerns/ |title=Wellbutrin – Anxiety Treatment Concerns |publisher=Health and Life }}</ref> |
||
[[Psychotic]] symptoms associated with bupropion are rare. They may include delusions, hallucinations, [[paranoia]], and confusion. Often, these symptoms can be reduced or eliminated by decreasing the dose or ceasing treatment.<ref name="WellbutrinPI"/><ref name="pmid3934991">{{cite journal | author = Golden RN, James SP, Sherer MA, Rudorfer MV, Sack DA, Potter WZ | title = Psychoses associated with bupropion treatment | journal = The American Journal of Psychiatry | volume = 142 | issue = 12 | pages = 1459–62 | year = 1985 | |
[[Psychotic]] symptoms associated with bupropion are rare. They may include delusions, hallucinations, [[paranoia]], and confusion. Often, these symptoms can be reduced or eliminated by decreasing the dose or ceasing treatment.<ref name="WellbutrinPI"/><ref name="pmid3934991">{{cite journal | author = Golden RN, James SP, Sherer MA, Rudorfer MV, Sack DA, Potter WZ | title = Psychoses associated with bupropion treatment | journal = The American Journal of Psychiatry | volume = 142 | issue = 12 | pages = 1459–62 | year = 1985 | pmid=3934991}}</ref><ref name="pmid3092682">{{cite journal | author = Johnston JA, Lineberry CG, Frieden CS | title = Prevalence of psychosis, delusions, and hallucinations in clinical trials with bupropion | journal = The American Journal of Psychiatry | volume = 143 | issue = 9 | pages = 1192–3 | year = 1986| pmid = 3092682}}</ref><ref name="pmid3151278">{{cite journal | author = Golden RN | title = Diethylpropion, bupropion, and psychoses | journal = The British Journal of Psychiatry : the Journal of Mental Science | volume = 153 | issue = 2| pages = 265–6 | year = 1988 | pmid = 3151278 | doi = 10.1192/bjp.153.2.265}}</ref><ref name="pmid1541606">{{cite journal | author = Ames D, Wirshing WC, Szuba MP | title = Organic mental disorders associated with bupropion in three patients | journal = The Journal of Clinical Psychiatry | volume = 53 | issue = 2 | pages = 53–5 | year = 1992 | pmid = 1541606}}</ref><ref name="pmid10588428">{{cite journal | author = Howard WT, Warnock JK | title = Bupropion-induced psychosis | journal = The American Journal of Psychiatry | volume = 156 | issue = 12 | pages = 2017–8 | year = 1999 | pmid = 10588428}}</ref><ref name="pmid12518275">{{cite journal | author = Neumann M, Livak V, Paul HW, Laux G | title = Acute psychosis after administration of bupropion hydrochloride (Zyban) | journal = Pharmacopsychiatry | volume = 35 | issue = 6 | pages = 247–8 | year = 2002| pmid = 12518275 | doi = 10.1055/s-2002-36393}}</ref><ref name="pmid18467537">{{cite journal | author = Bailey J, Waters S | title = Acute psychosis after bupropion treatment in a healthy 28-year-old woman | journal = Journal of the American Board of Family Medicine : JABFM | volume = 21 | issue = 3 | pages = 244–5 | year = 2008 | pmid = 18467537 | doi = 10.3122/jabfm.2008.03.070236 }}</ref><ref name="pmid19039677">{{cite journal | title = Two acute psychotic episodes after administration of bupropion: a case of involuntary rechallenge | journal = Pharmacy World & Science : PWS | volume = 31 | issue = 2 | pages = 238–40 | year = 2009 | pmid = 19039677 | doi = 10.1007/s11096-008-9272-x | last1 = Javelot | first1 = Hervé | last2 = Baratta | first2 = Alexandre | last3 = Weiner | first3 = Luisa | last4 = Javelot | first4 = Thierry | last5 = Nonnenmacher | first5 = Cathy | last6 = Westphal | first6 = Jean-Frédéric | last7 = Messaoudi | first7 = Michaël}}</ref><ref name="pmid19742195">{{cite journal | author = Grover S, Das PP | title = Can bupropion unmask psychosis | journal = Indian Journal of Psychiatry | volume = 51 | issue = 1 | pages = 53–4 | year = 2009 | pmid = 19742195 | pmc = 2738405 | doi = 10.4103/0019-5545.44907}}</ref> In many of these case reports, psychotic symptoms are associated with such risk factors as old age, the history of bipolar disorder or psychosis, and concomitant medications, for example, lithium or benzodiazepines.<ref name="isbn1-58562-243-5">{{cite book |author=Nemeroff, Charles B.; Schatzberg, Alan F. |title=Essentials of Clinical Psychopharmacology |publisher=American Psychiatric Publishing |location=Washington, D.C |year=2006 |page=146 |isbn=1-58562-243-5}}</ref> Most researchers have attributed the psychosis to bupropion's [[dopaminergic]] action.<ref name="pmid18467537"/><ref name="pmid17890983">{{cite journal | author = Hahn M, Hajek T, Alda M, Gorman JM | title = Psychosis induced by low-dose bupropion: sensitization of dopaminergic system by past cocaine abuse? | journal = Journal of Psychiatric Practice | volume = 13 | issue = 5 | pages = 336–8 | year = 2007| pmid = 17890983 | doi = 10.1097/01.pra.0000290673.32978.e8}}</ref> |
||
According to several case reports, stopping bupropion abruptly may result in [[withdrawal syndrome|discontinuation syndrome]] expressed as [[dystonia]], irritability, anxiety, [[mania]], headache, aches and pains.<ref>{{cite journal |
According to several case reports, stopping bupropion abruptly may result in [[withdrawal syndrome|discontinuation syndrome]] expressed as [[dystonia]], irritability, anxiety, [[mania]], headache, aches and pains.<ref>{{cite journal |
||
| pmid=17218049 |
| pmid=17218049 |
||
| year=2007 |
| year=2007 |
||
| month=April |
|||
| author=Wang, Hy; Chou, Wj; Huang, Ty; Hung, Cf |
| author=Wang, Hy; Chou, Wj; Huang, Ty; Hung, Cf |
||
| title=Acute dystonia resulting from abrupt bupropion discontinuation |
| title=Acute dystonia resulting from abrupt bupropion discontinuation |
||
| Line 153: | Line 154: | ||
| pmc=181231 |
| pmc=181231 |
||
| journal=Primary care companion to the Journal of clinical psychiatry |
| journal=Primary care companion to the Journal of clinical psychiatry |
||
| ⚫ | |||
| date= April 14, 2002 |
|||
| ⚫ | |||
| pmid=15014696 |
| pmid=15014696 |
||
| author=Berigan, Tr; Harazin, Js |
| author=Berigan, Tr; Harazin, Js |
||
| Line 167: | Line 167: | ||
| pmid=15003088 |
| pmid=15003088 |
||
| year=2004 |
| year=2004 |
||
| month=February |
|||
| author=Michael, N; Erfurth, A; Bergant, V |
| author=Michael, N; Erfurth, A; Bergant, V |
||
| title=A case report of mania related to discontinuation of bupropion therapy for smoking cessation |
| title=A case report of mania related to discontinuation of bupropion therapy for smoking cessation |
||
| Line 176: | Line 175: | ||
| journal=The Journal of clinical psychiatry |
| journal=The Journal of clinical psychiatry |
||
}}</ref> The prescribing information recommends dose tapering after bupropion has been used for [[seasonal affective disorder]];<ref name="WellbutrinPI"/> however it states that dose tapering is not required when discontinuing treatment for smoking cessation.<ref>{{cite web |accessdate = 2010-01-06 |
}}</ref> The prescribing information recommends dose tapering after bupropion has been used for [[seasonal affective disorder]];<ref name="WellbutrinPI"/> however it states that dose tapering is not required when discontinuing treatment for smoking cessation.<ref>{{cite web |accessdate = 2010-01-06 |
||
| title=PRESCRIBING INFORMATION |
| title=PRESCRIBING INFORMATION – ZYBAN (bupropion hydrochloride) Sustained-Release Tablets |
||
| url=http://us.gsk.com/products/assets/us_zyban.pdf}}</ref> |
| url=http://us.gsk.com/products/assets/us_zyban.pdf}}</ref> |
||
===Overdose=== |
===Overdose=== |
||
Overdose of bupropion results in significant clinical effects in over one-third of cases.<ref name="Balit"/> The most common symptoms include [[sinus tachycardia]], [[hypertension]], drowsiness, lethargy, agitation, nausea and vomiting, and in particular [[delirium]] and seizures.<ref name="Balit">{{cite journal |author=Balit CR, Lynch CN, Isbister GK |title=Bupropion poisoning: a case series |journal=Med. J. Aust. |volume=178 |issue=2 |pages=61–3 |year=2003 |pmid=12526723 |
Overdose of bupropion results in significant clinical effects in over one-third of cases.<ref name="Balit"/> The most common symptoms include [[sinus tachycardia]], [[hypertension]], drowsiness, lethargy, agitation, nausea and vomiting, and in particular [[delirium]] and seizures.<ref name="Balit">{{cite journal |author=Balit CR, Lynch CN, Isbister GK |title=Bupropion poisoning: a case series |journal=Med. J. Aust. |volume=178 |issue=2 |pages=61–3 |year=2003 |pmid=12526723}}</ref><ref name="Buckley">{{cite journal |author=Buckley NA, Faunce TA |title='Atypical' antidepressants in overdose: clinical considerations with respect to safety |journal=Drug safety: an international journal of medical toxicology and drug experience |volume=26 |issue=8 |pages=539–51 |year=2003 |pmid=12825968}}</ref><ref name=" Spiller">{{cite journal |author=Spiller HA, Ramoska EA, Krenzelok EP, Sheen SR, Borys DJ, Villalobos D, Muir S, Jones-Easom L |title=Bupropion overdose: a 3-year multi-center retrospective analysis |journal=The American journal of emergency medicine |volume=12 |issue=1 |pages=43–5 |year=1994 |pmid=8285970|doi=10.1016/0735-6757(94)90195-3}}</ref> Less commonly additional symptoms include auditory and visual [[hallucination]]s,<ref>{{cite journal |author=Mainie I, McGurk C, McClintock G, Robinson J |title=Seizures after buproprion overdose |journal=Lancet |volume=357 |issue=9268 |page=1624 |year=2001 |pmid=11386326 |doi=10.1016/S0140-6736(00)04770-X}}</ref> [[coma]],<ref name=" Spiller"/> and [[Electrocardiogram|ECG]] changes such as conduction disturbance or [[cardiac arrhythmia|arrhythmia]].<ref name="Shepherd">{{cite journal |author=Shepherd G, Velez LI, Keyes DC |title=Intentional bupropion overdoses |journal=The Journal of emergency medicine |volume=27 |issue=2 |pages=147–51 |year=2004 |pmid=15261357 |doi=10.1016/j.jemermed.2004.02.017}}</ref><ref>{{cite journal |author=Curry SC, Kashani JS, LoVecchio F, Holubek W |title=Intraventricular conduction delay after bupropion overdose |journal=The Journal of emergency medicine |volume=29 |issue=3 |pages=299–305 |year=2005 |pmid=16183450 |doi=10.1016/j.jemermed.2005.01.027}}</ref><ref>{{cite journal |author=Tracey JA, Cassidy N, Casey PB, Ali I |title=Bupropion (Zyban) toxicity |journal=Irish medical journal |volume=95 |issue=1 |pages=23–4 |year=2002 |pmid=11928786}}</ref> |
||
In the majority of childhood exploratory ingestions involving one or two tablets, children will remain asymptomatic.<ref name="Shepherd2">{{cite journal |author=Shepherd G, Velez LI, James DK, Keyes DC |title=Pediatric bupropion exposures reported in Texas: 1998–1999 [abstract] |journal=J Toxicol Clin Toxicol|volume=39 |
In the majority of childhood exploratory ingestions involving one or two tablets, children will remain [[asymptomatic]].<ref name="Shepherd2">{{cite journal |author=Shepherd G, Velez LI, James DK, Keyes DC |title=Pediatric bupropion exposures reported in Texas: 1998–1999 [abstract] |journal=J Toxicol Clin Toxicol|volume=39 |page=263 |year=2001}}</ref><ref name="Colbridge">{{cite journal |author=Colbridge MG, Dargan PI, Jones AL |title=Bupropion – the experience of the National Poisons Information Service (London) [abstract] |journal=J Toxicol Clin Toxicol|volume=40 |pages=398–9 |year=2002}}</ref> In teenagers and adults seizures are more commonly observed with the seizure rate increasing tenfold with doses of 600 mg daily.<ref name=" Johnston ">{{cite journal |author=Johnston JA, Lineberry CG, Ascher JA, Davidson J, Khayrallah MA, Feighner JP, Stark P |title=A 102-center prospective study of seizure in association with bupropion |journal=The Journal of clinical psychiatry |volume=52 |issue=11 |pages=450–6 |year=1991 |pmid=1744061}}</ref> One overdose study suggested a dose-dependent relationship with seizures; patients ingesting more than 4.5 g were likely to have a seizure and nearly all patients ingesting more than 9 g had a seizure.<ref name="Balit"/> |
||
There is no specific antidote for bupropion; treatment is supportive, and focuses on maintaining airway patency and controlling seizures with high dose intravenous [[benzodiazepine]]s or [[barbiturate]]s if seizures are refractory to benzodiazepines.<ref name="Buckley"/> Gastric decontamination may be of little benefit given the risk of seizures and aspiration<ref name="Buckley"/> but [[activated charcoal]] is recommended.<ref name="Balit"/> Additionally, [[whole bowel irrigation]] should be undertaken in those ingesting sustained release formulations.<ref name="Buckley"/> Toxic effects may be delayed in onset, with seizures developing as late as 32 hours.<ref name="Buckley"/> Subsequently patients should undergo [[electroencephalography|electroencephalographic]] monitoring for 48 hours.<ref name="WellbutrinPI">{{cite web | title = Wellbutrin XL Prescribing Information | month = December | year = 2008 | publisher = [[GlaxoSmithKline]] | url = http://us.gsk.com/products/assets/us_wellbutrinXL.pdf |format=PDF|accessdate=2010-01-16}}</ref> |
There is no specific antidote for bupropion; treatment is supportive, and focuses on maintaining airway patency and controlling seizures with high dose intravenous [[benzodiazepine]]s or [[barbiturate]]s if seizures are refractory to benzodiazepines.<ref name="Buckley"/> Gastric decontamination may be of little benefit given the risk of seizures and aspiration<ref name="Buckley"/> but [[activated charcoal]] is recommended.<ref name="Balit"/> Additionally, [[whole bowel irrigation]] should be undertaken in those ingesting sustained release formulations.<ref name="Buckley"/> Toxic effects may be delayed in onset, with seizures developing as late as 32 hours.<ref name="Buckley"/> Subsequently patients should undergo [[electroencephalography|electroencephalographic]] monitoring for 48 hours.<ref name="WellbutrinPI">{{cite web | title = Wellbutrin XL Prescribing Information | month = December | year = 2008 | publisher = [[GlaxoSmithKline]] | url = http://us.gsk.com/products/assets/us_wellbutrinXL.pdf |format=PDF|accessdate=2010-01-16}}</ref> |
||
Bupropion overdose rarely results in death, although cases have been reported.<ref name="Shepherd"/><ref>{{cite journal |author=Harris CR, Gualtieri J, Stark G |title=Fatal bupropion overdose |journal=J Toxicol Clin Toxicol |volume=35 |issue=3 |pages=321–4 |year=1997 |pmid=9140330 |doi=10.3109/15563659709001220}}</ref><ref>{{cite journal |author=Friel PN, Logan BK, Fligner CL |title=Three fatal drug overdoses involving bupropion |journal=Journal of analytical toxicology |volume=17 |issue=7 |pages=436–8 |year=1993 |pmid=8309220 |
Bupropion overdose rarely results in death, although cases have been reported.<ref name="Shepherd"/><ref>{{cite journal |author=Harris CR, Gualtieri J, Stark G |title=Fatal bupropion overdose |journal=J Toxicol Clin Toxicol |volume=35 |issue=3 |pages=321–4 |year=1997 |pmid=9140330 |doi=10.3109/15563659709001220}}</ref><ref>{{cite journal |author=Friel PN, Logan BK, Fligner CL |title=Three fatal drug overdoses involving bupropion |journal=Journal of analytical toxicology |volume=17 |issue=7 |pages=436–8 |year=1993 |pmid=8309220}}</ref> Fatalities are typically associated with large overdosage and related to [[metabolic acidosis]] and [[hypoxia (medical)|hypoxia]] as complications of [[status epilepticus]] with associated [[cardiorespiratory arrest]].<ref>{{cite journal |author=Paoloni R, Szekely I |title=Sustained-release bupropion overdose: a new entity for Australian emergency departments |journal=Emergency medicine (Fremantle, W.A.) |volume=14 |issue=1 |pages=109–12 |year=2002 |pmid=11993828 |doi=10.1046/j.1442-2026.2002.00295.x}}</ref> There is one published case report of successful treatment of refractory cardiac arrest in overdose of bupropion and [[lamotrigine]] using [[lipid rescue]].<ref>{{cite journal |title=Use of Lipid Emulsion in the Resuscitation of a Patient With Prolonged Cardiovascular Collapse After Overdose of Bupropion and Lamotrigine |journal=Ann Emerg Med |volume= 51|issue= 4|page=412|year=2007 |pmid=17766009 |doi=10.1016/j.annemergmed.2007.06.004 |last1=Sirianni |first1=Archie J. |last2=Osterhoudt |first2=Kevin C. |last3=Calello |first3=Diane P. |last4=Muller |first4=Allison A. |last5=Waterhouse |first5=Marie R. |last6=Goodkin |first6=Michael B. |last7=Weinberg |first7=Guy L. |last8=Henretig |first8=Fred M. |pages=412–5, 415.e1}}</ref> |
||
===Interactions=== |
===Interactions=== |
||
Since bupropion is metabolized to hydroxybupropion by the [[CYP2B6]] enzyme, drug interactions with CYP2B6 inhibitors are possible: this includes medications like [[paroxetine]], [[sertraline]], [[norfluoxetine]] (the active metabolite of [[fluoxetine]]), [[diazepam]], [[clopidogrel]], and [[orphenadrine]]. The expected result is the increase of bupropion and decrease of hydroxybupropion blood concentration. The reverse effect (decrease of bupropion and increase of hydroxybupropion) can be expected with CYP2B6 [[enzyme induction and inhibition|inducers]], such as [[carbamazepine]], [[clotrimazole]], [[rifampicin]], [[ritonavir]], [[St John's Wort]] and others.<ref name="pmid16368442">{{cite journal |author=Jefferson JW, Pradko JF, Muir KT |title=Bupropion for major depressive disorder: Pharmacokinetic and formulation considerations |journal=Clin Ther |volume=27 |issue=11 |pages=1685–95 |year=2005 |pmid=16368442 |doi=10.1016/j.clinthera.2005.11.011}}</ref> |
Since bupropion is metabolized to hydroxybupropion by the [[CYP2B6]] enzyme, drug interactions with CYP2B6 inhibitors are possible: this includes medications like [[paroxetine]], [[sertraline]], [[norfluoxetine]] (the active metabolite of [[fluoxetine]]), [[diazepam]], [[clopidogrel]], and [[orphenadrine]]. The expected result is the increase of bupropion and decrease of hydroxybupropion blood concentration. The reverse effect (decrease of bupropion and increase of hydroxybupropion) can be expected with CYP2B6 [[enzyme induction and inhibition|inducers]], such as [[carbamazepine]], [[clotrimazole]], [[rifampicin]], [[ritonavir]], [[St John's Wort]], [[phenobarbitone]], [[phenytoin]] and others.<ref name="pmid16368442">{{cite journal |author=Jefferson JW, Pradko JF, Muir KT |title=Bupropion for major depressive disorder: Pharmacokinetic and formulation considerations |journal=Clin Ther |volume=27 |issue=11 |pages=1685–95 |year=2005 |pmid=16368442 |doi=10.1016/j.clinthera.2005.11.011}}</ref> |
||
Bupropion is an inhibitor of [[CYP2D6]].<ref name="pmid16368442"/> Hydroxybupropion |
Bupropion is an inhibitor of [[CYP2D6]].<ref name="pmid16368442"/> Hydroxybupropion is an inhibitor of that enzyme ''in vitro''.<ref>{{cite journal|author=Hesse LM, Nevkatakrishnan K, Court MH, von Moltke LL, Duan SX, Shader RI, Greenblatt DJ|year=2000|title=2B6 mediates the in vitro hydroxylation of bupropion: potential drug interactions with other antidipressants|journal=Drug Metab Dispos|volume=28|pages=1176–1183}}</ref> A significant increase in the concentration of some drugs metabolized by CYP2D6 ([[venlafaxine]], [[desipramine]] and [[dextromethorphan]], but not fluoxetine or paroxetine) has been observed when they are taken with bupropion.<ref name="pmid16368442"/><ref name="pmid11926715">{{cite journal |author=Kennedy SH, McCann SM, Masellis M, McIntyre RS, Raskin J, McKay G, Baker GB |title=Combining bupropion SR with venlafaxine, paroxetine, or fluoxetine: a preliminary report on pharmacokinetic, therapeutic, and sexual dysfunction effects |journal=J Clin Psychiatry |volume=63 |issue=3 |pages=181–6 |year=2002 |pmid=11926715 |doi=10.4088/JCP.v63n0302}}</ref> |
||
Bupropion lowers the seizure threshold; accordingly, extreme care should be taken when prescribing bupropion with other medications that also lower it, such as antipsychotics, [[theophylline]], [[steroid]]s, and some [[tricyclic antidepressant]]s.<ref name="WellbutrinPI"/> Its combination with [[nicotine replacement therapy|nicotine replacement therapies]] can elevate blood pressure; since this combination is no more effective than either a nicotine patch or bupropion alone, it is not recommended. |
Bupropion lowers the seizure threshold; accordingly, extreme care should be taken when prescribing bupropion with other medications that also lower it, such as antipsychotics, [[theophylline]], [[steroid]]s, and some [[tricyclic antidepressant]]s.<ref name="WellbutrinPI"/> Its combination with [[nicotine replacement therapy|nicotine replacement therapies]] can elevate blood pressure; since this combination is no more effective than either a nicotine patch or bupropion alone, it is not recommended. |
||
The prescribing information recommends minimizing the use of alcohol, since in rare cases bupropion reduces alcohol tolerance, and because the excessive use of alcohol may lower the seizure threshold.<ref name="WellbutrinPI"/> A small study conducted by GlaxoSmithKline indicated that bupropion (100 mg) may counteract the subjective effects of small doses of alcohol (16–32 mL, slightly less than 1–2 standard US drinks). The volunteers reported feeling more sober and clear-headed and less sedated. Bupropion also reduced the detrimental effect of alcohol on auditory vigilance. The combination of bupropion (100 mg) and two drinks of alcohol increased heart rate by six beats per minute, a statistically significant increase.<ref>{{cite web |url=http://download.gsk-clinicalstudyregister.com/files/827.pdf |title=P02-31UK I Examination of Bupropion and Ethanol, Alone and in Combination, on Human Performance Tests, Subjective Rating Scales, EEG and Autonomic Responses |accessdate=2011-01-25 |
The prescribing information recommends minimizing the use of alcohol, since in rare cases bupropion reduces alcohol tolerance, and because the excessive use of alcohol may lower the seizure threshold.<ref name="WellbutrinPI"/> A small study conducted by GlaxoSmithKline indicated that bupropion (100 mg) may counteract the subjective effects of small doses of alcohol (16–32 mL, slightly less than 1–2 standard US drinks). The volunteers reported feeling more sober and clear-headed and less sedated. Bupropion also reduced the detrimental effect of alcohol on auditory vigilance. The combination of bupropion (100 mg) and two drinks of alcohol increased heart rate by six beats per minute, a statistically significant increase.<ref>{{cite web |url=http://download.gsk-clinicalstudyregister.com/files/827.pdf |title=P02-31UK I Examination of Bupropion and Ethanol, Alone and in Combination, on Human Performance Tests, Subjective Rating Scales, EEG and Autonomic Responses |accessdate=2011-01-25|format=PDF |work=GlaxoSmithKline Clinical Trial Register. Bupropion Studies }}</ref> |
||
==Detection in biological fluids== |
==Detection in biological fluids== |
||
Bupropion may be quantitated in blood, plasma or serum to monitor therapeutic use, confirm a diagnosis of poisoning or assist in a medicolegal death investigation. The drug is unstable in biological fluids at room temperature and therefore specimens should be maintained at the coldest possible temperature prior to analysis.<ref> |
Bupropion may be quantitated in blood, plasma or serum to monitor therapeutic use, confirm a diagnosis of poisoning or assist in a medicolegal death investigation. The drug is unstable in biological fluids at room temperature and therefore specimens should be maintained at the coldest possible temperature prior to analysis.<ref>Baselt, R. ''Disposition of Toxic Drugs and Chemicals in Man'', 8th edition, Biomedical Publications, Foster City, CA, 2008, pp. 193–195 ISBN 0962652350.</ref> |
||
==Mechanism of action== |
==Mechanism of action== |
||
Bupropion is a [[dopamine reuptake inhibitor|dopamine]] and [[norepinephrine reuptake inhibitor]].<ref>{{cite journal | |
Bupropion is a [[dopamine reuptake inhibitor|dopamine]] and [[norepinephrine reuptake inhibitor]] and releaser.<ref name="pmid19497387">{{cite journal |author=Arias HR |title=Is the inhibition of nicotinic acetylcholine receptors by bupropion involved in its clinical actions? |journal=Int. J. Biochem. Cell Biol. |volume=41 |issue=11 |pages=2098–108 |year=2009 |pmid=19497387 |doi=10.1016/j.biocel.2009.05.015}}</ref> It is about twice as potent an inhibitor of dopamine reuptake than of norepinephrine reuptake.<ref>{{cite journal|doi=10.1521/capn.2008.13.5.6 |title=Prescriptions Into Practice: Bupropion|year=2008|last1=Akhtar|first1=Nadeem Masood|last2=Khan|first2=Afreen Bano|journal=Child and Adolescent Psychopharmacology News|volume=13|issue=5|pages=6}}</ref> Besides reuptake inhibition of dopamine and norepinephrine, bupropion also causes the release of dopamine and norepinephrine.<ref>{{cite book|doi=10.1016/S0074-7742(09)88009-4|chapter=Chapter 9. Pharmacological and Neurotoxicological Actions Mediated By Bupropion and Diethylpropion|title=New Concepts of Psychostimulant Induced Neurotoxicity|series=International Review of Neurobiology|year=2009|last1=Arias|first1=Hugo R.|last2=Santamaría|first2=Abel|last3=Ali|first3=Syed F.|isbn=9780123745040|volume=88|pages=223}}</ref> As bupropion is rapidly converted in the body into several metabolites with differing activity, its action cannot be understood without reference to its metabolism. The occupancy of [[dopamine transporter]] (DAT) by bupropion and its metabolites in the human brain as measured by [[positron emission tomography]] was 6–22% in an independent study<ref>{{cite journal | author = Meyer J, Goulding V S, Wilson A A, Hussey D, Christensen B K, Houle S | title = Bupropion occupancy of the dopamine transporter is low during clinical treatment | journal = Psychopharmacology | volume = 163 | pages = 102–105 | year = 2002 |pmid=12185406 |doi=10.1007/s00213-002-1166-3 | issue = 1}}</ref> and 12–35% according to GlaxoSmithKline researchers.<ref>{{cite journal | author = Learned-Coughkin S M, Bergstrom M, Savitcheva I, Ascher J, Schmith V D, Langstrom B | title = ''In vivo'' activity of bupropion at the human dopamine transporter as measured by positron emission tomography | journal = Biol Psychiatry | volume = 54| pages = 800–805 | year = 2003 |pmid=14550679 | doi = 10.1016/S0006-3223(02)01834-6 | issue = 8}}</ref> Based on analogy with [[serotonin reuptake inhibitor]]s, higher than 50% inhibition of DAT would be needed for the dopamine reuptake mechanism to be a major mechanism of the drug's action. By contrast, approximately 65% occupancy or greater of DAT is required to achieve euphoria and reach abuse potential.<ref>{{cite journal|pmid=11408518|year=2001|last1=Howell|first1=LL|last2=Wilcox|first2=KM|title=The dopamine transporter and cocaine medication development: Drug self-administration in nonhuman primates|volume=298|issue=1|pages=1–6|journal=The Journal of pharmacology and experimental therapeutics|url=http://research.yerkes.emory.edu/Howell/JPET298.pdf}} |
||
</ref> However recent research indicates that dopamine is inactivated by norepinephrine reuptake in the frontal cortex, which largely lacks dopamine transporters, therefore bupropion can increase dopamine neurotransmission in this part of the brain, and this may be one possible explanation for any additional dopaminergic effects.<ref>{{cite web|url=http://stahlonline.cambridge.org/prescribers_drug.jsf?page=0521683505c09_p53-58.html.therapeutics&name=Bupropion&title=Therapeutics |title=Bupropion |work=Stahlonline.cambridge.org | publisher=[[Cambridge University Press]] |accessdate=2010-04-27}}</ref> |
|||
Bupropion does not inhibit [[monoamine oxidase]] or serotonin reuptake. However, it has been shown to indirectly enhance the firing of serotonergic neurons, via activation of downstream norepinephrine flow. Bupropion has also been shown to act as a noncompetitive |
Bupropion does not inhibit [[monoamine oxidase]] or serotonin reuptake. However, it has been shown to indirectly enhance the firing of serotonergic neurons, via activation of downstream norepinephrine flow. Bupropion has also been shown to act as a noncompetitive [[nicotinic antagonist]].<ref name="pmid19497387"/> The degree of inhibition of α<sub>3</sub>β<sub>4</sub> receptors correlates well with the decrease in self-administration of morphine and methamphetamine in rats,<ref>{{cite web | author = Glick S D| title = Ibogaine Analogues: Drug Development for Addictive Disorders. (Presentation at Addiction Medicine State of the Art 2003 Conference October 8–11, 2003, Radisson-Miyako Hotel, San Francisco) | publisher = www.csam-asam.org| accessdate = 2007-03-24 | url = http://www.csam-asam.org/pdf/misc/Glick.ppt}}</ref> and may be relevant to the effect of bupropion on nicotine addiction. The drug is supplied as a [[racemic]] mixture, and the activities of the individual enantiomers were not found to differ.<ref>{{cite journal|pmid=8240925|year=1993|last1=Musso|first1=DL|last2=Mehta|first2=NB|last3=Soroko|first3=FE|last4=Ferris|first4=RM|last5=Hollingsworth|first5=EB|last6=Kenney|first6=BT|title=Synthesis and evaluation of the antidepressant activity of the enantiomers of bupropion|volume=5|issue=7|pages=495–500|doi=10.1002/chir.530050704|journal=Chirality}}</ref> |
||
Two [[Structure-activity relationship|SAR]] studies date from 2009<ref>{{cite journal | pmid = 19821577 | year = 2009 | last1 = Carroll | first1 = FI | last2 = Blough | first2 = BE | last3 = Abraham | first3 = P | last4 = Mills | first4 = AC | last5 = Holleman | first5 = JA | last6 = Wolckenhauer | first6 = SA | last7 = Decker | first7 = AM | last8 = Landavazo | first8 = A | last9 = McElroy | first9 = KT | title = Synthesis and biological evaluation of bupropion analogues as potential pharmacotherapies for cocaine addiction | volume = 52 | issue = 21 | pages = 6768–81 | doi = 10.1021/jm901189z | journal = Journal of |
Two [[Structure-activity relationship|SAR]] studies date from 2009<ref>{{cite journal | pmid = 19821577 | year = 2009 | last1 = Carroll | first1 = FI | last2 = Blough | first2 = BE | last3 = Abraham | first3 = P | last4 = Mills | first4 = AC | last5 = Holleman | first5 = JA | last6 = Wolckenhauer | first6 = SA | last7 = Decker | first7 = AM | last8 = Landavazo | first8 = A | last9 = McElroy | first9 = KT | title = Synthesis and biological evaluation of bupropion analogues as potential pharmacotherapies for cocaine addiction | volume = 52 | issue = 21 | pages = 6768–81 | doi = 10.1021/jm901189z | journal = Journal of Medical Chemistry}}</ref> and 2010.<ref>{{cite journal | pmid =20158204 | year =2010 | last1 =Carroll | first1 =FI | last2 =Blough | first2 =BE | last3 =Mascarella | first3 =SW | last4 =Navarro | first4 =HA | pmc =2841507 | last5 =Eaton | first5 =JB | last6 =Lukas | first6 =RJ | last7 =Damaj | first7 =MI | title =Synthesis and biological evaluation of bupropion analogues as potential pharmacotherapies for smoking cessation | volume =53 | issue =5 | pages =2204–14 | doi =10.1021/jm9017465 | journal =Journal of Medical Chemistry}}</ref> |
||
==Pharmacokinetics== |
==Pharmacokinetics== |
||
[[ |
[[File:Metabolites of bupropion.png|thumb|right|176px|Important metabolites of bupropion.]] |
||
Bupropion is metabolized in the liver. |
|||
| ⚫ | |||
| ⚫ | It has several active [[metabolite]]s: ''R,R''-hydroxybupropion, ''S,S''-hydroxybupropion, ''threo''-hydrobupropion and ''erythro''-hydrobupropion, which are further metabolized to inactive metabolites and eliminated through excretion into the urine. Pharmacological data on bupropion and its metabolites are presented in Table 1. Bupropion is known to weakly inhibit the [[Alpha-1 adrenergic receptor|α<sub>1</sub> adrenergic receptor]], with a 14% potency of its dopamine uptake inhibition, and the [[histamine H1 receptor|H<sub>1</sub> receptor]], with a 9% potency.<ref name = "horst1998"/> |
||
The biological activity of bupropion can be attributed to a significant degree to its active metabolites, in particular to ''S,S''-hydroxybupropion. GlaxoSmithKline developed this metabolite as a separate drug called [[radafaxine]],<ref>{{cite press release | url = http://www.biospace.com/news_story.aspx?StoryID=18222420&full=1 | title = GlaxoSmithKline (GSK) Reviews Novel Therapeutics For CNS Disorders And Confirms Strong Pipeline Momentum | date = November 23, 2004 | accessdate = 2007-08-18 | publisher = PRNewswire}}</ref> but discontinued development in 2006 due to "an unfavourable risk/benefit |
The biological activity of bupropion can be attributed to a significant degree to its active metabolites, in particular to ''S,S''-hydroxybupropion. GlaxoSmithKline developed this metabolite as a separate drug called [[radafaxine]],<ref>{{cite press release | url = http://www.biospace.com/news_story.aspx?StoryID=18222420&full=1 | title = GlaxoSmithKline (GSK) Reviews Novel Therapeutics For CNS Disorders And Confirms Strong Pipeline Momentum | date = November 23, 2004 | accessdate = 2007-08-18 | publisher = PRNewswire}}</ref> but discontinued development in 2006 due to "an unfavourable risk/benefit |
||
| Line 214: | Line 216: | ||
GlaxoSmithKline (July 26, 2006) {{PDFlink|[http://www.gsk.com/investors/reports/gsk_q22006/q22006.pdf ''Pipeline Update'']|136 KB}}. [[Press release]]. Retrieved on 2007-08-18.</ref> |
GlaxoSmithKline (July 26, 2006) {{PDFlink|[http://www.gsk.com/investors/reports/gsk_q22006/q22006.pdf ''Pipeline Update'']|136 KB}}. [[Press release]]. Retrieved on 2007-08-18.</ref> |
||
Bupropion is metabolized to hydroxybupropion by [[CYP2B6]], an [[isoenzyme]] of the [[cytochrome P450|cytochrome P450 system]]. Alcohol causes an increase of CYP2B6 in the liver, and persons with a history of alcohol use metabolize bupropion faster. The mechanism of formation of ''erythro''-hydrobupropion and ''threo''-hydrobupropion has not been studied but is probably mediated by one of the carbonyl reductase enzymes. The metabolism of bupropion is highly variable: the effective doses of bupropion received by persons who ingest the same amount of the drug may differ by as much as 5.5 times (and the half-life from 3 to 16 hours), and of hydroxybupropion by as much as 7.5 times (and the half-life from 12 to 38 hours).<ref name="pmid14515060">{{cite journal |author=Kirchheiner J, Klein C, Meineke I, Sasse J, Zanger UM, Mürdter TE, Roots I, Brockmöller J |title=Bupropion and 4-OH-bupropion pharmacokinetics in relation to genetic polymorphisms in CYP2B6 |journal=Pharmacogenetics |volume=13 |issue=10 |pages=619–26 |year=2003 |pmid=14515060 |doi=10.1097/00008571-200310000-00005}}</ref><ref name="pmid15083067">{{cite journal |author=Hesse LM, He P, Krishnaswamy S, Hao Q, Hogan K, von Moltke LL, Greenblatt DJ, Court MH |title=Pharmacogenetic determinants of interindividual variability in bupropion hydroxylation by cytochrome P450 2B6 in human liver microsomes |journal=Pharmacogenetics |volume=14 |issue=4 |pages=225–38 |year=2004 |pmid=15083067|doi=10.1097/00008571-200404000-00002}}</ref> Based on this, some researchers have advocated monitoring of the blood level of bupropion and hydroxybupropion.<ref name="pmid1813908">{{cite journal |author=Preskorn SH |title=Should bupropion dosage be adjusted based upon therapeutic drug monitoring? |journal=Psychopharmacology bulletin |volume=27 |issue=4 |pages=637–43 |year=1991 |pmid=1813908 |
Bupropion is metabolized to hydroxybupropion by [[CYP2B6]], an [[isoenzyme]] of the [[cytochrome P450|cytochrome P450 system]]. Alcohol causes an increase of CYP2B6 in the liver, and persons with a history of alcohol use metabolize bupropion faster. The mechanism of formation of ''erythro''-hydrobupropion and ''threo''-hydrobupropion has not been studied but is probably mediated by one of the carbonyl reductase enzymes. The metabolism of bupropion is highly variable: the effective doses of bupropion received by persons who ingest the same amount of the drug may differ by as much as 5.5 times (and the half-life from 3 to 16 hours), and of hydroxybupropion by as much as 7.5 times (and the half-life from 12 to 38 hours).<ref name="pmid14515060">{{cite journal |author=Kirchheiner J, Klein C, Meineke I, Sasse J, Zanger UM, Mürdter TE, Roots I, Brockmöller J |title=Bupropion and 4-OH-bupropion pharmacokinetics in relation to genetic polymorphisms in CYP2B6 |journal=Pharmacogenetics |volume=13 |issue=10 |pages=619–26 |year=2003 |pmid=14515060 |doi=10.1097/00008571-200310000-00005}}</ref><ref name="pmid15083067">{{cite journal |author=Hesse LM, He P, Krishnaswamy S, Hao Q, Hogan K, von Moltke LL, Greenblatt DJ, Court MH |title=Pharmacogenetic determinants of interindividual variability in bupropion hydroxylation by cytochrome P450 2B6 in human liver microsomes |journal=Pharmacogenetics |volume=14 |issue=4 |pages=225–38 |year=2004 |pmid=15083067|doi=10.1097/00008571-200404000-00002}}</ref> Based on this, some researchers have advocated monitoring of the blood level of bupropion and hydroxybupropion.<ref name="pmid1813908">{{cite journal |author=Preskorn SH |title=Should bupropion dosage be adjusted based upon therapeutic drug monitoring? |journal=Psychopharmacology bulletin |volume=27 |issue=4 |pages=637–43 |year=1991 |pmid=1813908}}</ref> |
||
There are significant interspecies differences in the metabolism of bupropion, with [[guinea pig]]s' metabolism of the drug being closest to that of humans compared to mice and rats.<ref>{{cite journal |author = Suckow R F, Smith T M, Perumal A S, Cooper T B |title = Pharmacokinetics of bupropion and metabolites in plasma and brain of rats, mice, and guinea pigs |journal = Drug Metab Dispos |volume=14 |issue=6|pages=692–697 |year=1986 |pmid=2877828}}</ref> Particular caution is needed when extrapolating the results of experiments on rats to humans since hydroxybupropion, the main metabolite of bupropion in humans, is absent in rats.<ref name="pharmacological287">{{cite journal |author = Welch R M, Lai A A, Schroeder D H |title = Pharmacological significance of the species differences in bupropion metabolism |journal = Xenobiotica |volume=17 |issue=3|pages=287–289 |year=1987 |pmid=3107223 |doi = 10.3109/00498258709043939}}</ref> |
There are significant interspecies differences in the metabolism of bupropion, with [[guinea pig]]s' metabolism of the drug being closest to that of humans compared to mice and rats.<ref>{{cite journal |author = Suckow R F, Smith T M, Perumal A S, Cooper T B |title = Pharmacokinetics of bupropion and metabolites in plasma and brain of rats, mice, and guinea pigs |journal = Drug Metab Dispos |volume=14 |issue=6|pages=692–697 |year=1986 |pmid=2877828}}</ref> Particular caution is needed when extrapolating the results of experiments on rats to humans since hydroxybupropion, the main metabolite of bupropion in humans, is absent in rats.<ref name="pharmacological287">{{cite journal |author = Welch R M, Lai A A, Schroeder D H |title = Pharmacological significance of the species differences in bupropion metabolism |journal = Xenobiotica |volume=17 |issue=3|pages=287–289 |year=1987 |pmid=3107223 |doi = 10.3109/00498258709043939}}</ref> |
||
There have been two reported cases of false-positive urine amphetamine tests in persons taking bupropion. |
There have been two reported cases of false-positive urine amphetamine tests in persons taking bupropion. As substituted phenethylamines encompass the substituted amphetamines where bupropion is described as β-Keto-3-chloro-N-tert-butylamphetamine, this is likely the cause of a positive result shown in drug-screening tests on those taking the medicine. More specific follow-up tests were negative.<ref name="pmid10999247">{{cite journal |author=Weintraub D, Linder MW |title=Amphetamine positive toxicology screen secondary to bupropion |journal=Depress Anxiety |volume=12 |issue=1 |pages=53–4 |year=2000 |pmid=10999247 |doi=10.1002/1520-6394(2000)12:1<53::AID-DA8>3.0.CO;2-4}}</ref><ref name="pmid7768026">{{cite journal |author=Nixon AL, Long WH, Puopolo PR, Flood JG |title=Bupropion metabolites produce false-positive urine amphetamine results |journal=Clin. Chem. |volume=41 |issue=6 Pt 1 |pages=955–6 |year=1995 |pmid=7768026}}</ref> |
||
{| class="wikitable" style="margin:auto; margin:1em auto 1em auto;" |
{| class="wikitable" style="margin:auto; margin:1em auto 1em auto;" |
||
| Line 301: | Line 303: | ||
Bupropion is synthesized by brominating 3'-chloropropiophenone, followed by [[nucleophilic substitution]] with [[t-butylamine|''t''-butylamine]].<ref name="United States Patent: 3819706"/><ref>{{Cite journal | journal = [[J. Chem. Ed.]] | title = A Short, One-Pot Synthesis of Bupropion | author = Daniel M. Perrine, Jason T. Ross, Stephen J. Nervi, and Richard H. Zimmerman | url = http://jchemed.chem.wisc.edu/Journal/Issues/2000/nov/PlusSub/V77N11/p1479.pdf |format=PDF| year = 2000 | volume = 77 | issue = 11 | page = 1479}}</ref> It is a [[substituted cathinone]], or in other words, a derivative of [[cathinone]]. |
Bupropion is synthesized by brominating 3'-chloropropiophenone, followed by [[nucleophilic substitution]] with [[t-butylamine|''t''-butylamine]].<ref name="United States Patent: 3819706"/><ref>{{Cite journal | journal = [[J. Chem. Ed.]] | title = A Short, One-Pot Synthesis of Bupropion | author = Daniel M. Perrine, Jason T. Ross, Stephen J. Nervi, and Richard H. Zimmerman | url = http://jchemed.chem.wisc.edu/Journal/Issues/2000/nov/PlusSub/V77N11/p1479.pdf |format=PDF| year = 2000 | volume = 77 | issue = 11 | page = 1479}}</ref> It is a [[substituted cathinone]], or in other words, a derivative of [[cathinone]]. |
||
:[[ |
:[[File:Synthesis of bupropion.png|500px]] |
||
==Analogues== |
|||
A number of bupropion analogues were recently reported by [[F. Ivy Carroll]], ''et al.''<ref name=smoke>{{Cite pmid|20158204}}</ref><ref name=cocaine>{{Cite pmid|19821577}}</ref> The aims were two-fold: one [[smoking cessation]],<ref name=smoke/> and two, [[cocaine addiction]] agents.<ref name=cocaine/> |
|||
In their efforts to find [[smoking cessation]] agents, [[F. Ivy Carroll]], ''et al.'' also recently (2011) disclosed a number of 2-(substituted phenyl)-3,5,5-trimethylmorpholine analogues.<ref name=morpholine>{{Cite pmid|21319801}}</ref> |
|||
==History== |
|||
| ⚫ | |||
| ⚫ | Bupropion was invented by [[Nariman Mehta]] of Burroughs Wellcome (now [[GlaxoSmithKline]]) in 1969, and the US patent for it was granted in 1974.<ref name="United States Patent: 3819706">{{cite web |url=http://patft.uspto.gov/netacgi/nph-Parser?Sect1=PTO1&Sect2=HITOFF&d=PALL&p=1&u=%2Fnetahtml%2FPTO%2Fsrchnum.htm&r=1&f=G&l=50&s1=3819706.PN.&OS=PN/3819706&RS=PN/3819706 |title=United States Patent 3,819,706: Meta-chloro substituted α-butylamino-propiophenones |author=Mehta NB |date=1974-06-25 |
||
[[F. Ivy Carroll]], ''et al.'' have also produced [[hydroxy]]-bupropion analogues, with [[smoking cessation]] as the stated application.<ref>{{Cite pmid|20576796}}</ref><ref>{{Cite pmid|20509659}}</ref><ref>{{Cite pmid|15322260}}</ref> |
|||
Although structurally unrelated to bupropion, [[F. Ivy Carroll]], ''et al.'' recently (2011) disclosed a number of 3-[[phenyltropanes]] that had some [[nAChR]] activity.<ref>{{Cite pmid|21319801}}</ref> |
|||
| ⚫ | |||
Through reading the above citations it would be possible to draw up possible SARs. To give an example, extending the α-methyl chain in bupropion to C<sub>3</sub>H<sub>7</sub> (presumed n-propyl), improved the [<sup>3</sup>H]DA IC<sub>50</sub> from 658nM to 56nM. Extending the α-methyl to an α-ethyl also improved [<sup>3</sup>H]DA IC<sub>50</sub> to 209nM. According to work done on [[sibutramine]] though, the absolute optimum chain length in these compounds is an iso-butyl radical. This has been confirmed by [[P. Meltzer]] ''et al.'' in his seminal paper on [[naphyrone]] analogs.<ref>{{Cite pmid|16480278}}</ref> [[Mark Froimowitz]], ''et al.'' also explored the effect of replacing the carbomethoxy group in Ritalin with different alkyl groups.<ref>{{Cite pmid|21973080}}</ref><ref>{{Cite pmid|17228864}}</ref> [http://www.bindingdb.org/data/mols/tenK5030/MolStructure_50302913.html In fact, the isobutyl analog of bupropion was also studied by RTI], but [<sup>3</sup>H]DA IC<sub>50</sub> was only 140nM. |
|||
It can also be readily seen that omission of the hydroxy group in [[Radafaxine]] makes DA and NE IC<sub>50</sub> shift from 630 and 180nM, respectively, to 220 and 130nM, and 5-HT was 387nM.<ref name=morpholine/> |
|||
| ⚫ | In 1996, the FDA approved a [[sustained release|sustained-release]] formulation of bupropion called Wellbutrin SR, intended to be taken twice a day (as compared with three times a day for immediate-release Wellbutrin).<ref name="NIDA_notes">"[http:// |
||
Other analogs of [[desoxy]]-[[Radafaxine]] were also prepared. For example, replacing the ''m''-chloro group with ''m''-fluoro gave an analog that had DA and NE and 5HT IC<sub>50</sub> values of 61, 32, and 4600nM, respectively. |
|||
| ⚫ | |||
===Availability and dose forms=== |
|||
| ⚫ | |||
| ⚫ | Brand-name and generic bupropion tablets are available in three |
||
A somewhat interesting observation is that in the case of [[methcathinone]], the ''m''-chloro group is not needed as the compound is fully active already. However in the case of bupropion where we have a ''tert''-butyl-N group, [http://www.bindingdb.org/data/mols/tenK5030/MolStructure_50302912.html omission of the ''m''-chloro halogen gave a compound that had diminished activity as a MAT inhibitor]. [http://www.bindingdb.org/data/mols/tenK5030/MolStructure_50302911.html Moving the ''m''-chloro to the ''para'' position also led to a deterioration in the compounds resultant potency.] |
|||
Efficacy cannot be determined early in treatment as, with most antidepressants, the medication must reach maximum blood plasma concentration for some time to be effective (2–4 weeks in some cases). Some prescribers may also prescribe a small dose of a [[benzodiazepine]] to offset the initial side effects some patients encounter when starting bupropion treatment. These initial side effects, similar to stimulant side effects such as anxiety, agitation, nervousness, insomnia, etc. generally subside within the first 2 weeks and do not occur in every patient. People taking other products that manipulate [[norepinephrine]] and/or its reuptake (for example antidepressants like [[venlafaxine]] (Effexor, Pristiq), smoking cessation aids like Zyban, or pain medications like [[tramadol]]) should surely let their doctor know they also take these medications; however, in proper dosages, concurrent use of [[norepinephrine]] manipulating medications is usually not an issue.{{Citation needed|date=May 2011}} |
|||
==Regulatory history== |
|||
| ⚫ | On October 11, 2007, two providers of consumer information on nutritional products and supplements, [[ConsumerLab.com]] and The People's Pharmacy, released the results of comparative tests of different brands of bupropion.<ref name=peoples>{{cite web |url=http://www.peoplespharmacy.com/archives/generic_drug_problems/generic_drug_equality_questioned.php |title=Generic Drug Equality Questioned |accessdate=2007-10-13 |
||
| ⚫ | |||
| ⚫ | Bupropion was invented by [[Nariman Mehta]] of Burroughs Wellcome (now [[GlaxoSmithKline]]) in 1969, and the US patent for it was granted in 1974.<ref name="United States Patent: 3819706">{{cite web |url=http://patft.uspto.gov/netacgi/nph-Parser?Sect1=PTO1&Sect2=HITOFF&d=PALL&p=1&u=%2Fnetahtml%2FPTO%2Fsrchnum.htm&r=1&f=G&l=50&s1=3819706.PN.&OS=PN/3819706&RS=PN/3819706 |title=United States Patent 3,819,706: Meta-chloro substituted α-butylamino-propiophenones |author=Mehta NB |date=1974-06-25 |publisher=USPTO |accessdate=2008-06-02}}</ref> It was approved by the United States [[Food and Drug Administration]] (FDA) as an antidepressant on December 30, 1985, and marketed under the name Wellbutrin.<ref>Wellbutrin Label and Approval History. U.S. [[Food and Drug Administration]] [[Center for Drug Evaluation and Research]]. Retrieved on 2007-08-18. Data available for download on [http://www.accessdata.fda.gov/scripts/cder/ob/docs/obdetail.cfm?Appl_No=018644&TABLE1=OB_Rx FDA website].</ref> However, a significant incidence of [[seizures]] at the originally recommended dosage (400–600 mg) caused the withdrawal of the drug in 1986. Subsequently, the risk of seizures was found to be highly dose-dependent, and bupropion was re-introduced to the market in 1989 with a maximum recommended dose of 450 mg/day.{{cn|date=November 2012}} |
||
| ⚫ | |||
| ⚫ | In France, marketing authorization was granted on August 3, 2001, |
||
| ⚫ | In 1996, the FDA approved a [[sustained release|sustained-release]] formulation of bupropion called Wellbutrin SR, intended to be taken twice a day (as compared with three times a day for immediate-release Wellbutrin).<ref name="NIDA_notes">"[http://web.archive.org/web/20070805053452/http://drugabuse.gov/NIDA_notes/NNvol20N5/Bupropion.html Bupropion Helps People With Schizophrenia Quit Smoking]." ''[[National Institute on Drug Abuse]].'' ''Research Findings'', Vol. 20, No. 5 (April 2006). Retrieved on August 19, 2007.</ref> In 2003, the FDA approved another sustained-release formulation called Wellbutrin XL, intended for once-daily dosing. Wellbutrin SR and XL are available in [[generic drug|generic]] form in the United States, while in Canada, only the SR formulation is available in generic form. In 1997, bupropion was approved by the FDA for use as a smoking cessation aid under the name Zyban.<ref name="NIDA_notes"/> In 2006, Wellbutrin XL was similarly approved as a treatment for [[seasonal affective disorder]].<ref name="wellbutrin_seasonal">Staff Writer. "[http://www.cnn.com/HEALTH/library/DI/00069.html Seasonal affective disorder drug Wellbutrin XL wins approval]." [[CNN]]. June 14, 2006. Retrieved August 19, 2007.</ref> |
||
| ⚫ | In the |
||
There is no generic equivalent at this time.{{When|date=April 2011}} This is a once-a-day formula that is taken in slightly higher dosages due to the difference in the salts (e.g. 348 mg Aplenzin Bupropion HBr is equivalent to 300 mg Bupropion HCl). In some patients the hydrobromide salt may metabolize in a more{{Citation needed|date=October 2011}} (or less){{Citation needed|date=October 2011}} consistent fashion than the classic hydrochloride salt, delivering the medication in a more{{Citation needed|date=October 2011}} (or less){{Citation needed|date=October 2011}} consistent manner{{Citation needed|date=October 2011}}. This is just one more option available to the prescriber of once-a-day Bupropion treatments.{{Citation needed|date=October 2011}} |
|||
In 2012, the U.S. Justice Department announced that GlaxoSmithKline had agreed to plead guilty and pay a $3-billion fine, in part for promoting the unapproved use of Wellbutrin for weight loss and sexual dysfunction.<ref>{{cite news|title=Glaxo Agrees to Pay $3 Billion in Fraud Settlement|newspaper=The New York Times|date=July 2, 2012|url=http://www.nytimes.com/2012/07/03/business/glaxosmithkline-agrees-to-pay-3-billion-in-fraud-settlement.html|first1=Katie|last1=Thomas|first2=Michael S.|last2=Schmidt}}</ref> |
|||
| ⚫ | In April 2008, the FDA approved a formulation of bupropion as a hydrobromide salt instead of a hydrochloride salt, to be sold under the name Aplenzin by [[Sanofi-Aventis]].<ref>{{cite web |url=http://www.medscape.com/viewarticle/574187 |title=FDA Approvals: Advair, Relistor, Aplenzin |accessdate=2008-05-09 |author=Waknine, Yael |date=2008-05-08 |publisher=[[Medscape]]}}</ref> |
||
| ⚫ | On October 11, 2007, two providers of consumer information on nutritional products and supplements, [[ConsumerLab.com]] and The People's Pharmacy, released the results of comparative tests of different brands of bupropion.<ref name=peoples>{{cite web |url=http://www.peoplespharmacy.com/archives/generic_drug_problems/generic_drug_equality_questioned.php |title=Generic Drug Equality Questioned |accessdate=2007-10-13 }}</ref> The People's Pharmacy received multiple reports of increased side effects and decreased efficacy of generic bupropion, which prompted it to ask ConsumerLab.com to test the products in question. The tests showed that "one of a few generic versions of Wellbutrin XL 300 mg, sold as Budeprion XL 300 mg, didn't perform the same as the brand-name pill in the lab."<ref name=msnbc>{{cite web |url=http://www.msnbc.msn.com/id/21142869/ |title=Report questions generic antidepressant |accessdate=2007-10-13 |author=Jacqueline Stenson |date=2007-10-12|publisher=[[msnbc.com]]}}</ref> The FDA investigated these complaints and concluded that the Budeprion XL is equivalent to Wellbutrin XL in regard to bioavailability of bupropion and its main active metabolite hydroxybupropion. The FDA also said that coincidental natural mood variation is the most likely explanation for the apparent worsening of depression after the switch from Wellbutrin XL to Budeprion XL.<ref name="urlReview of Therapeutic Equivalence: Generic Bupropion XL 300 mg and Wellbutrin XL 300 mg">{{cite web |url=http://www.fda.gov/AboutFDA/CentersOffices/OfficeofMedicalProductsandTobacco/CDER/ucm153270.htm|archiveurl=http://web.archive.org/web/20110606092603/http://www.fda.gov/AboutFDA/CentersOffices/CDER/ucm153270.htm |archivedate=2011-06-06|title=Review of Therapeutic Equivalence: Generic Bupropion XL 300 mg and Wellbutrin XL 300 mg |accessdate=2008-04-19}}</ref> On October 3, 2012, however, the FDA reversed this opinion, announcing that "Budeprion XL 300 mg fails to demonstrate therapeutic equivalence to Wellbutrin XL 300 mg."<ref>{{cite web|title=Generic antidepressant not equivalent to original Wellbutrin: FDA|url=http://www.reuters.com/article/2012/10/06/us-fda-antidepressant-idUSBRE8950BE20121006|accessdate=2012-10-09</ref><ref>{{cite web|title=FDA Update: Budeprion XL 300 mg Not Therapeutically Equivalent to Wellbutrin XL 300 mg|url=http://www.fda.gov/Drugs/DrugSafety/PostmarketDrugSafetyInformationforPatientsandProviders/ucm322161.htm|accessdate=2012-10-09}}</ref> The FDA did not test the bioequivalence of any of the other generic versions of Wellbutrin XL 300 mg, but requested that the four manufacturers submit data on this question to the FDA by May, 2013.<ref>http://www.fda.gov/Drugs/DrugSafety/PostmarketDrugSafetyInformationforPatientsandProviders/ucm322161.htm </ref> |
||
| ⚫ | In France, marketing authorization was granted for Zyban on August 3, 2001, with a maximum daily dose of 300 mg;<ref>{{cite press release | url = http://www.afssaps.fr/Infos-de-securite/Communiques-Points-presse/ZYBAN-R-sevrage-tabagique-et-securite-d-emploi/(language)/fre-FR | title = Zyban : sevrage tabagique et sécurité d'emploi | trans_title = Zyban: smoking cessation and job security | date = January 18, 2002 | accessdate = 2011-01-25 | publisher = Agence française de sécurité sanitaire des produits de santé | language = French}}</ref> only sustained-release bupropion is available, and only as a smoking cessation aid. Bupropion was granted a licence for use in adults with major depression in the Netherlands in early 2007, with GlaxoSmithKline expecting subsequent approval in other European countries.<ref>{{cite press release | authorlink = GlaxoSmithKline | author = GlaxoSmithKline | title = GlaxoSmithKline receives first European approval for Wellbutrin XR |publisher = GlaxoSmithKline | date = 2007-01-16 | url = http://www.gsk.com/media/pressreleases/2007/2007_01_16_GSK956.htm | accessdate = 2011-01-25}}</ref> |
||
==Extended-release formulations== |
|||
<!--- will be folded into another section as soon as I figure out which one -Aurochs ---> |
|||
| ⚫ | |||
| ⚫ | Brand-name and generic bupropion tablets are available in three formulations, each as the hydrochloride [[salt (chemistry)|salt]]: immediate release (Wellbutrin), [[sustained release]] (Wellbutrin SR), and extended release (Wellbutrin XL or XR). "Sustained release" and "extended release" are generally interchangeable terms, but in this case Wellbutrin SR is intended for twice-daily dosing and Wellbutrin XL is intended for once-daily dosing. Not all generics have retained this naming scheme, and the [[United States Pharmacopeia]] requires all prolonged-release drug formulations (including generics for Wellbutrin SR) to be labeled "extended release", which has caused confusion and medication errors.<ref name="USPnews">{{cite web | title = Practitioner's Reporting News | publisher = [[United States Pharmacopeia]] | date = 2004-09-30 | url=http://www.usp.org/hqi/practitionerPrograms/newsletters/practitionerReportingNews/prn1202004-09-30.html#14 | accessdate = 2007-08-20}} {{Dead link|date=March 2012|bot=H3llBot}}</ref><ref>{{cite journal | last = Rosack | first = Jim | title = Company Tries to Clear Up Confusion About Bupropion | journal = Psychiatric News | volume=41 | issue=6 | page=16 | publisher = [[American Psychiatric Association]] | date = 2006-03-17 | url = http://psychnews.psychiatryonline.org/Mobile/article.aspx?articleid=109809| accessdate = 2007-08-20}}</ref> According to GlaxoSmithKline, the SR tablets should not be split.<ref>[http://us.gsk.com/products/assets/us_wellbutrinSR.pdf wellbutrin sr sustained-release tablets]. (PDF) . Retrieved on 2012-01-05.</ref> Merck indicates that splitting a 150 mg Wellbutrin SR tablet decreases the time to peak levels, but that it will retain its sustained-release characteristics.<ref>{{cite web |url=http://www.merck.com/mmpe/print/lexicomp/bupropion.html |title=BuPROPion: Drug Information Provided by Lexi-Comp: Merck Manual Professional |accessdate=2007-06-16}}</ref> |
||
| ⚫ | |||
===Recreational use=== |
===Recreational use=== |
||
According to the US government classification of psychiatric medications, bupropion is "non-abusable"<ref>{{cite web |url=http://www.ncbi.nlm.nih.gov/books/NBK24472/table/A36258/ |title=Exhibit 4-3 Abuse Potential of Common Psychiatric Medications |accessdate=2011-01-25 |work=Health Services/Technology Assessment Text (HSTAT)|publisher=U.S. [[National Library of Medicine]]}}</ref> |
According to the US government classification of psychiatric medications, bupropion is "non-abusable".<ref>{{cite web |url=http://www.ncbi.nlm.nih.gov/books/NBK24472/table/A36258/ |title=Exhibit 4-3 Abuse Potential of Common Psychiatric Medications |accessdate=2011-01-25 |work=Health Services/Technology Assessment Text (HSTAT)|publisher=U.S. [[National Library of Medicine]]}}</ref> |
||
Two studies on drug abusers indicated that the subjective effects of bupropion are markedly different from those of [[amphetamine]].<ref name="pmid6412263">{{cite journal |author=Miller L, Griffith J |title=A comparison of bupropion, dextroamphetamine, and placebo in mixed-substance abusers |journal=Psychopharmacology (Berl.) |volume=80 |issue=3 |pages=199–205 |year=1983 |pmid=6412263|doi=10.1007/BF00436152}}</ref><ref name="pmid6406459">{{cite journal |author=Griffith JD, Carranza J, Griffith C, Miller LL |title=Bupropion: clinical assay for amphetamine-like abuse potential |journal=J Clin Psychiatry |volume=44 |issue=5 Pt 2 |pages=206–8 |year=1983 |pmid=6406459 |
Two studies on drug abusers indicated that the subjective effects of bupropion are markedly different from those of [[amphetamine]].<ref name="pmid6412263">{{cite journal |author=Miller L, Griffith J |title=A comparison of bupropion, dextroamphetamine, and placebo in mixed-substance abusers |journal=Psychopharmacology (Berl.) |volume=80 |issue=3 |pages=199–205 |year=1983 |pmid=6412263|doi=10.1007/BF00436152}}</ref><ref name="pmid6406459">{{cite journal |author=Griffith JD, Carranza J, Griffith C, Miller LL |title=Bupropion: clinical assay for amphetamine-like abuse potential |journal=J Clin Psychiatry |volume=44 |issue=5 Pt 2 |pages=206–8 |year=1983 |pmid=6406459}}</ref> Healthy volunteers trained to discriminate amphetamine and placebo recognized bupropion (400 mg) as amphetamine 20% of the time, compared to 10% for placebo and 75% for [[methylphenidate]] (20 mg). They also reported feeling alert, vigorous, elated and energetic, reflecting the general stimulating properties of bupropion. In contrast to amphetamine and methylphenidate, there was no feeling of "liking the drug" and no desire to take it again.<ref name="pmid9526144">{{cite journal |author=Rush CR, Kollins SH, Pazzaglia PJ |title=Discriminative-stimulus and participant-rated effects of methylphenidate, bupropion, and triazolam in d-amphetamine-trained humans |journal=Experimental and clinical psychopharmacology |volume=6 |issue=1 |pages=32–44 |year=1998 |pmid=9526144|doi=10.1037/1064-1297.6.1.32}}</ref> A comparison of bupropion SR (150 mg) and [[caffeine]] (178 mg) indicated that caffeine may have higher abuse liability since it resulted in more reports of pleasant feelings and a "high" than bupropion.<ref name="pmid15001822">{{cite journal |title=Subjective effects of slow-release bupropion versus caffeine as determined in a quasi-naturalistic setting |journal=Pharmacology |volume=70 |issue=4 |pages=206–15 |year=2004 |pmid=15001822 |doi=10.1159/000075550 |last1=Zernig |first1=Gerald |last2=De Wit |first2=Harriet |last3=Telser |first3=Stefan |last4=Nienhusmeier |first4=Matthias |last5=Wakonigg |first5=Gudrun |last6=Sturm |first6=Katja |last7=Berger |first7=Iris |last8=Kemmler |first8=Georg |last9=Saria |first9=Alois}}</ref> |
||
There have been four reports of bupropion abuse in the literature. Three cases described teenagers crushing and |
There have been four reports of bupropion abuse in the literature. Three cases described teenagers crushing and insufflating (snorting) the drug, two of them resulting in seizures.<ref>{{cite journal |author=Khurshid KA, Decker DH |title=Bupropion insufflation in a teenager |journal=J Child Adolesc Psychopharmacol |volume=14 |issue=1 |pages=157–8 |year=2004 |pmid=15142406 |doi=10.1089/104454604773840634}}</ref><ref>{{cite journal |author=Lu JJ, Thompson TM, Narunatvanich D, Fischbein CB, Mycyk MB |title=Seizure after Nasal Insufflation of Bupropion [abstract] |journal= Clin Toxicol (Phila) |volume=45 |page=632 |year=2007}}</ref><ref>{{cite journal |author=Welsh CJ, Doyon S |title=Seizure induced by insufflation of bupropion |journal=N. Engl. J. Med. |volume=347 |issue=12 |page=951 |year=2002 |pmid=12239274 |doi=10.1056/NEJM200209193471222}}</ref> An additional case was reported where a teenager ingested bupropion believing the drug to be a stimulant.<ref>{{cite journal |author=McCormick J |title=Recreational bupropion abuse in a teenager |journal=Br J Clin Pharmacol |volume=53 |issue=2 |page=214 |year=2002 |pmid=11851650|doi=10.1046/j.0306-5251.2001.01538.x |pmc=1874291}}</ref> A newspaper article on medication abuse in prisons mentioned bupropion as one of the psychotropic medications commonly abused by inmates.<ref>{{cite web |url=http://www.cnsnewsonline.com/index.asp?section_id=113&show=dept&article_id=4907 |title=Intervention Reduces Psychotropic Abuse in Correctional Facility; CNS News, JUNE 2005, VOLUME: 07:06 |accessdate=2007-05-27 |author=Volpe KD | archiveurl = http://web.archive.org/web/20070929071643/http://www.cnsnewsonline.com/index.asp?section_id=113&show=dept&article_id=4907| archivedate = September 29, 2007}}</ref> |
||
==Animal research== |
==Animal research== |
||
In animal studies [[squirrel monkey]]s<ref name="pmid2529365">{{cite journal |author=Bergman J, Madras BK, Johnson SE, Spealman RD |title=Effects of cocaine and related drugs in nonhuman primates. III. Self-administration by [[squirrel monkey]]s |journal=J. Pharmacol. Exp. Ther. |volume=251 |issue=1 |pages=150–5 |year=1989 |pmid=2529365 |
In animal studies, [[squirrel monkey]]s<ref name="pmid2529365">{{cite journal |author=Bergman J, Madras BK, Johnson SE, Spealman RD |title=Effects of cocaine and related drugs in nonhuman primates. III. Self-administration by [[squirrel monkey]]s |journal=J. Pharmacol. Exp. Ther. |volume=251 |issue=1 |pages=150–5 |year=1989 |pmid=2529365}}</ref> and rats<ref name="pmid9103538">{{cite journal |author=Tella SR, Ladenheim B, Cadet JL |title=Differential regulation of dopamine transporter after chronic self-administration of bupropion and nomifensine |journal=J Pharmacol Exp Ther |volume=281 |issue=1 |pages=508–13 |year=1997 |pmid=9103538}}</ref> maintained the intravenous self-administration of bupropion. Significant interspecies differences of bupropion metabolism exist, particularly between rats and humans.<ref name="pharmacological287"/> |
||
==References== |
==References== |
||
| Line 345: | Line 361: | ||
*[http://www.merck.com/mmpe/lexicomp/bupropion.html List of international brand names for bupropion] |
*[http://www.merck.com/mmpe/lexicomp/bupropion.html List of international brand names for bupropion] |
||
* {{dmoz|Health/Pharmacy/Drugs_and_Medications/B/Bupropion/|Bupropion}} |
* {{dmoz|Health/Pharmacy/Drugs_and_Medications/B/Bupropion/|Bupropion}} |
||
* [http://www.rxlist.com/cgi/generic/buprop_cp.htm Wellbutrin Pharmacology, Pharmacokinetics, Studies, Metabolism |
* [http://www.rxlist.com/cgi/generic/buprop_cp.htm Wellbutrin Pharmacology, Pharmacokinetics, Studies, Metabolism – Bupropion – RxList Monographs] |
||
* [http://www.nami.org/Template.cfm?Section=About_Medications&template=/ContentManagement/ContentDisplay.cfm&ContentID=7388 NAMI Wellbutrin] |
* [http://www.nami.org/Template.cfm?Section=About_Medications&template=/ContentManagement/ContentDisplay.cfm&ContentID=7388 NAMI Wellbutrin] |
||
* [http://www.mentalhealth.com/drug/p30-b04.html Bupropion article from mentalhealth.com] |
* [http://www.mentalhealth.com/drug/p30-b04.html Bupropion article from mentalhealth.com] |
||
| Line 359: | Line 375: | ||
[[Category:Antidepressants]] |
[[Category:Antidepressants]] |
||
[[Category:Cathinones]] |
[[Category:Cathinones]] |
||
[[Category:GlaxoSmithKline]] |
[[Category:GlaxoSmithKline brands]] |
||
[[Category:Nicotinic antagonists]] |
[[Category:Nicotinic antagonists]] |
||
[[Category:Norepinephrine-dopamine reuptake inhibitors]] |
[[Category:Norepinephrine-dopamine reuptake inhibitors]] |
||
| Line 368: | Line 384: | ||
[[de:Bupropion]] |
[[de:Bupropion]] |
||
[[es:Bupropion]] |
[[es:Bupropion]] |
||
[[fa:بوپروپیون]] |
|||
[[fr:Bupropione]] |
[[fr:Bupropione]] |
||
[[ko:부프로피온]] |
|||
[[it:Bupropione]] |
[[it:Bupropione]] |
||
[[he:בופרופיון]] |
[[he:בופרופיון]] |
||
[[hu:Bupropion]] |
[[hu:Bupropion]] |
||
[[nl:Bupropion]] |
|||
[[ja:ブプロピオン]] |
[[ja:ブプロピオン]] |
||
[[pl:Bupropion]] |
[[pl:Bupropion]] |
||
| Line 378: | Line 397: | ||
[[simple:Bupropion]] |
[[simple:Bupropion]] |
||
[[sr:Bupropion]] |
[[sr:Bupropion]] |
||
[[fi:Bupropioni]] |
|||
[[sv:Bupropion]] |
[[sv:Bupropion]] |
||
[[zh:丁氨苯丙酮]] |
[[zh:丁氨苯丙酮]] |
||
Revision as of 20:28, 25 December 2012
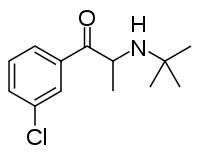 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Wellbutrin, Zyban |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a695033 |
| License data | |
| Pregnancy category |
|
| Routes of administration | Oral, insufflation, IV |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 5 to 20% in animals; no studies in humans |
| Protein binding | 84% |
| Metabolism | Hepatic—important CYP2B6 and 2D6 involvement |
| Elimination half-life | 20 hours |
| Excretion | Renal (87%), fecal (10%) |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C13H18ClNO |
| Molar mass | 239.74 g/mol g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Bupropion (/bjuːˈproʊpi.ɒn/ bew-PROH-pee-on;[2] marketed as Wellbutrin, Zyban, Voxra, Budeprion, Prexaton, Elontril or Aplenzin; and formerly known as amfebutamone[3]) is an atypical antidepressant and smoking cessation aid. Its chemical name is β-keto-3-chloro-N-tert-butylamphetamine, a substituted cathinone (β-ketoamphetamine), as well as substituted amphetamine. The drug therefore is a mild psychostimulant. Its primary pharmacological action is thought to be norepinephrine-dopamine reuptake inhibition. It binds selectively to the dopamine transporter, but its behavioural effects have often been attributed to its inhibition of norepinephrine reuptake.[4][5] It also acts as a nicotinic acetylcholine receptor antagonist.[6][7] Bupropion belongs to the chemical class of aminoketones and is similar in structure to stimulants cathinone and diethylpropion, and to phenethylamines in general. Medically, bupropion serves as a non-tricyclic antidepressant fundamentally different to most commonly prescribed antidepressants such as SSRIs.
Initially researched and marketed as an antidepressant, bupropion was subsequently found to be effective as a smoking cessation aid. With over 20 million retail prescriptions in 2007, it was the fourth-most prescribed antidepressant in the United States retail market after sertraline, escitalopram, and fluoxetine.[8]
Bupropion lowers seizure threshold, and its potential to cause seizures has been widely publicized. However, at the recommended dose, the risk of seizures is comparable to that observed for other antidepressants.[9] Bupropion is an effective antidepressant on its own, but it is particularly popular as an add-on medication in the cases of incomplete response to the first-line selective serotonin reuptake inhibitor (SSRI) antidepressant. In contrast to many other antidepressants, bupropion does not cause weight gain or sexual dysfunction; in most studies, groups placed on bupropion showed statistically significant increases in sexual functioning (libido), and mild to moderate weight loss.[10]
Medical uses
Depression
Placebo-controlled double-blind clinical studies have confirmed the efficacy of bupropion for clinical depression.[11] Comparative clinical studies demonstrated the equivalency of bupropion and sertraline (Zoloft), fluoxetine (Prozac), paroxetine (Paxil)[12] and escitalopram (Lexapro)[13] as antidepressants. One study reported a significantly higher remission rate with bupropion treatment than for venlafaxine (Effexor).[14] Unlike all other antidepressants, except mirtazapine (Remeron) and tianeptine (Stablon), bupropion does not cause sexual dysfunction and the occurrence of sexual side effects is not different from placebo.[15][16] Bupropion treatment is not associated with weight gain; on the contrary, at the end of every study comparing bupropion with placebo or other antidepressants the bupropion group had a lower average weight.[10] Bupropion is more effective than SSRIs at improving symptoms of hypersomnia and fatigue in depressed patients.[17] In a comparative meta-analysis, there appeared to be a modest advantage for the SSRIs compared to bupropion in the treatment of depression with high anxiety, while these medications were equivalent for depression with moderate or low anxiety.[18]
According to several surveys, the augmentation of a prescribed SSRI with bupropion is the preferred strategy among clinicians when the patient does not respond to the SSRI.[19] For example, the combination of bupropion and citalopram (Celexa) was observed to be more effective than switching to another antidepressant. The addition of bupropion to an SSRI (primarily fluoxetine or sertraline) resulted in a significant improvement in 70–80% of patients who had an incomplete response to the first-line antidepressant.[20][21] Bupropion improved ratings of "energy", which had decreased under the influence of the SSRI; also noted were improvements of mood and motivation, and some improvement of cognitive and sexual functions. Sleep quality and anxiety ratings in most cases were unchanged.[21] In the STAR*D study, the patients who did not respond to citalopram (Celexa) were randomly assigned to augmentation by bupropion or buspirone (Buspar). Approximately 30% of subjects in both groups achieved a remission. However, bupropion augmentation gave better results based on the patients' self-ratings and was much better tolerated. The authors observed that "these findings reveal a consistently more favorable outcome with sustained-release bupropion than with buspirone augmentation of citalopram."[22] The same study indicated a possibility of higher remission rate when the non-responders to citalopram received bupropion augmentation instead of being switched to bupropion (30% vs. 20%).[23]
Anxiety
Bupropion has shown some success in treating social anxiety disorder[24] and anxiety combined with depression,[25] but not panic disorder with agoraphobia.[26] Its anxiolytic potential has been compared to that of sertraline[25] and doxepin.[27] However, it can cause agitation in some patients, especially at higher doses, and often increases anxiety, much like methylphenidate.[28]
Smoking cessation
Bupropion reduces the severity of nicotine cravings and withdrawal symptoms. After a seven-week treatment, 27% of subjects who received bupropion reported that an urge to smoke was a problem, versus 56% of those who received placebo. In the same study, 21% of the bupropion group reported mood swings, versus 32% of the placebo group.[29] The bupropion treatment course lasts for seven to twelve weeks, with the patient halting the use of tobacco about ten days into the course. Bupropion approximately doubles the chance of quitting smoking successfully after three months. One year after the treatment, the odds of sustaining smoking cessation are still 1.5 times higher in the bupropion group than in the placebo group.[30] The combination of bupropion and nicotine appears not to further increase the cessation rate. In a single trial that compared bupropion and varenicline (Chantix) directly, varenicline showed superior efficacy: after one year, the rate of continuous abstinence was 10% for placebo, 15% for bupropion, and 23% for varenicline.[31] Bupropion slows the weight gain that often occurs in the first weeks after quitting smoking (after seven weeks, the placebo group had an average 2.7 kg increase in weight, versus 1.5 kg for the bupropion group). With time, however, this effect becomes negligible (after 26 weeks, both groups recorded an average 4.8 kg weight gain).[29]
Sexual dysfunction
Bupropion is one of few antidepressants that do not cause sexual dysfunction.[32] According to a survey of psychiatrists, it is the drug of choice for the treatment of SSRI-induced sexual dysfunction, although this is not an indication approved by the U.S. Food and Drug Administration (FDA). 36% of psychiatrists preferred switching patients with SSRI-induced sexual dysfunction to bupropion, and 43% favored the augmentation of the current medication with bupropion.[33] There are studies demonstrating the efficacy of both approaches; improvement of the desire and orgasm components of sexual function were the most often noted. For the augmentation approach, the addition of at least 200 mg/day of bupropion to the SSRI regimen may be necessary to achieve an improvement since the addition of 150 mg/day of bupropion did not produce a statistically significant difference from placebo.[34][35][36][37][38][39]
Several studies have indicated that bupropion also relieves sexual dysfunction in people who do not have depression. In a mixed-gender double-blind study, 63% of subjects on a 12-week course of bupropion rated their condition as improved or much improved, versus 3% of subjects on placebo.[40] Two studies, one of which was placebo-controlled, demonstrated the efficacy of bupropion for women with hypoactive sexual desire disorder,[41][42] resulting in significant improvement of arousal, orgasm and overall satisfaction. Bupropion also showed promise as a treatment for sexual dysfunction caused by chemotherapy for breast cancer[43] and for orgasmic dysfunction.[44] As with the treatment of SSRI-induced sexual disorder, a higher dose of bupropion (300 mg) may be necessary: a randomized study employing a lower dose (150 mg) failed to find a significant difference between bupropion, sexual therapy or combined treatment.[45] Bupropion does not adversely affect any measures of sexual functioning in healthy men.[46]
Obesity
A recent meta-analysis of anti-obesity medications pooled the results of three double-blind, placebo-controlled trials of bupropion. It confirmed the efficacy of bupropion given at 400 mg per day for treating obesity. Over a period of 6 to 12 months, weight loss in the bupropion group (4.4 kg) was significantly greater than in the placebo group (1.7 kg). The same review found the differences in weight loss between bupropion and other established weight-loss medications, such as sibutramine, orlistat and diethylpropion, not to be statistically significant.[47] Combinations of bupropion and zonisamide (Empatic) and of bupropion and naltrexone (Contrave) are currently being studied for obesity.[48]
Attention-deficit hyperactivity disorder
Although attention-deficit hyperactivity disorder (ADHD) is not an approved indication, bupropion was found to be effective for adult ADHD.[49] There have been many positive case studies and other uncontrolled clinical studies of bupropion for ADHD in minors.[50] However, in the largest to date double-blind study, which was conducted by GlaxoSmithKline, the results were inconclusive. Aggression and hyperactivity as rated by the children's teachers were significantly improved in comparison to placebo; in contrast, parents and clinicians could not distinguish between the effects of bupropion and placebo.[50] The 2007 guideline on the ADHD treatment from American Academy of Child and Adolescent Psychiatry notes that the evidence for bupropion is "far weaker" than for the FDA-approved treatments. Its effect may also be "considerably less than of the approved agents... Thus it may be prudent for the clinician to recommend a trial of behavior therapy at this point, before moving to these second-line agents."[51] Similarly, the 2006 guideline from the Texas Department of State Health Services recommends considering bupropion or a tricyclic antidepressant as a fourth-line treatment after trying two different stimulants and atomoxetine (Strattera).[52][53]
A study of prophylactic bupropion for the prevention of smoking among teenagers with ADHD yielded unexpected results. The teenagers taking bupropion were two times more likely (close to statistical significance) to begin smoking than the teenagers in the placebo group. However, because significance was not reached, this difference can be attributed entirely to chance. At the same time, the sub-group of patients taking stimulants in addition to bupropion or placebo had a five times lower risk of smoking initiation.[54]
Other uses
Bupropion improves mood by increasing the concentration of dopamine in emotional brain regions. The mechanism may improve the negative mood associated with methamphetamine withdrawal. Though it has demonstrated no effectiveness in the treatment of cocaine dependence, there is weak evidence that it may be useful in methamphetamine dependence.[55]
Bupropion was approved by the U.S. Food and Drug Administration (FDA)[56] for the prevention of seasonal affective disorder.[57] According to several case studies and a pilot study, bupropion lowers the level of an inflammatory mediator TNF-alpha[58] and may be useful in autoinflammatory conditions such as Crohn's disease[58][59] and psoriasis.[60][61]
No properly controlled double-blind studies of bupropion for Parkinson's disease have been conducted. A small 1984 study funded by bupropion's manufacturer found that addition of bupropion to carbidopa or levodopa improved Parkinson's symptoms in ten out of twenty patients; however, the side effects, particularly nausea and vomiting, were frequent.[62] The American Psychiatric Association notes that, "there is no evidence favoring any particular antidepressant medication from the standpoint of therapeutic efficacy in patients with Parkinson's disease complicated by major depressive disorder".[63]
There is considerable disagreement regarding whether it is useful to add an antidepressant, including bupropion, to a mood stabilizer in patients with bipolar depression.[64][65][66]
Bupropion, similarly to other antidepressants, is not efficacious in chronic low back pain.[67][68] However, it has been found to have surprisingly high efficacy in the treatment of neuropathic pain.[69][70][71]
Contraindications
GlaxoSmithKline advises that bupropion should not be prescribed to individuals with epilepsy or other conditions that lower the seizure threshold, such as alcohol or benzodiazepine withdrawal, anorexia nervosa, bulimia, or active brain tumors. It should be avoided in individuals who are also taking monoamine oxidase inhibitors (MAOIs). When switching from MAOIs to bupropion, it is important to include a washout period of about two weeks between the medications.[72] The prescribing information approved by the FDA recommends that caution should be exercised when treating patients with liver damage, severe kidney disease, and severe hypertension, as well as in pediatric patients, adolescents and young adults due to the increased risk of suicidal ideation.[72]
According to a retrospective case series published in 1993, bupropion treatment may exacerbate tics in children with co-occurring ADHD and Tourette syndrome.[73] No further research of this side effect has been conducted.
Adverse effects
Seizure is the most controversial side effect of bupropion, and was responsible for its initial withdrawal from the market. The risk of seizure is highly dose-dependent: 0.1% at 100–300 mg of bupropion, 0.4% at 300–450 mg, and 2% at 600 mg.[citation needed] For comparison, the incidence of the first unprovoked seizure in the general population is 0.07 to 0.09%. The risk of seizure for other antidepressants is as follows: 0.1–0.6% for imipramine, depending on dosage; 0–0.06% for amitriptyline, depending on dosage; 0.5% for clomipramine; 0.4% for maprotiline; and 0.2% for fluoxetine and fluvoxamine.[74] Experiments on mice indicate that increased susceptibility to seizure is a general side effect of chronically using antidepressants that inhibit norepinephrine transporters, such as imipramine, desipramine and reboxetine.[75] Clinical depression itself was reported to increase the occurrence of seizures two-to-sevenfold compared with the general population; in this light, the above statistics could indicate that low to moderate doses of antidepressants, including bupropion, may actually have an anti-convulsive action.[76]
The prescribing information notes that hypertension, sometimes severe, was observed in some patients, both with and without pre-existing hypertension. The frequency of this adverse effect was under 1% and not significantly higher than that found with placebo.[72] In a group of cardiac patients with depression, high doses of bupropion (400–500 mg/day) caused a rise in supine blood pressure but had no effect on pulse rate.[77] No statistically significant changes in blood pressure or heart rate occurred in patients with or without heart conditions at a lower dose of 300 mg/day.[78] In a study of bupropion for ADHD, a rise of systolic blood pressure by 6 mm Hg and of heart rate by 7 beats per minute (both statistically significant) were observed.[79] A study of smokers hospitalized for heart disease found a 1.5-fold increase (close to being statistically significant) in subsequent cardiovascular events in the bupropion group, compared with the placebo group, but found no difference in blood pressure.[80] Although the cardiovascular side effects of bupropion appear to be mild, it cannot be recommended for patients with heart disease, since the safety comparison with SSRIs (such as sertraline and fluoxetine, which may have a preventative effect after a myocardial infarction[81]) is not in its favor.
In the UK, more than 7,600 reports of suspected adverse reactions were collected in the first two years after bupropion's approval by the MHRA as part of the Yellow Card Scheme, which monitored side effects. Approximately 540,000 people were treated with bupropion for smoking cessation during that period. The MHRA received 60 reports of "suspected [emphasis MHRA's] adverse reactions to Zyban which had a fatal outcome". The agency concluded that "in the majority of cases the individual's underlying condition may provide an alternative explanation."[82] This is consistent with a large, 9,300-patient safety study that showed that the mortality of smokers taking bupropion is not higher than the natural mortality of smokers of the same age.[83]
Other isolated adverse effects have been reported. Three cases of liver toxicity have been described,[84] a very low incidence given the widespread use of the drug. A single case of clitoral priapism (clitorism) has been reported in the literature.[85]
The common adverse effects associated with 12-hour sustained-release bupropion (with the greatest difference from placebo) are dry mouth, nausea, insomnia, tremor, excessive sweating and tinnitus. Those that most often resulted in interruption of the treatment in the same trial were rash (2.4%) and nausea (0.8%).[72]
Psychiatric
The FDA requires all antidepressants, including bupropion, to carry a black box warning stating that antidepressants may increase the risk of suicide in persons younger than 25. This warning is based on a statistical analysis conducted by the FDA which found a 2-fold increase of the suicidal ideation and behavior in children and adolescents, and 1.5-fold increase of suicidality in the 18–24 age group.[86]
Suicidal ideation and behavior in clinical trials are rare. For the above analysis, the FDA combined the results of 295 trials of 11 antidepressants for psychiatric indications in order to obtain statistically significant results. Considered separately, bupropion and nine other antidepressants were not statistically different from placebo. Only fluoxetine caused a significant decrease in suicidal ideation.[86]
Suicidal behavior is less likely when bupropion is prescribed for smoking cessation. According to a 2007 Cochrane Database review, there have been four suicides per one million prescriptions and one case of suicidal ideation per ten thousand prescriptions of bupropion for smoking cessation in the UK. The review concludes, "Although some suicides and deaths while taking bupropion have been reported, thus far there is insufficient evidence to suggest they were caused by bupropion."[87]
Two years later, the FDA issued a health advisory, which warned that the prescription of bupropion and varenicline for smoking cessation has been associated with reports about unusual behaviour changes, agitation and hostility. Some patients have become depressed or have had their depression worsen, have had thoughts about suicide or dying, or have attempted suicide.[88][89] This advisory was based on the postmarket review of anti-smoking products, which identified 153 reports of the suicidal adverse events for varenicline over the first year it was marketed and 75 reports for bupropion over ten years. No clear association with suicidality was identified for nicotine patch products.[90]
Mania is one of the rare side effects of bupropion. The prescribing information notes that "it is generally believed (though not established in controlled trials)" that, should an episode of depression actually be the first presentation of bipolar disorder, treating it with antidepressants, including bupropion, may precipitate a manic episode.[72] More recent data indicate that the addition of newer antidepressants, including bupropion, to a mood stabilizer does not cause the switch to mania more often than the addition of placebo.[65] Moreover, when added to a mood stabilizer, bupropion and sertraline had a twice lower switch risk than venlafaxine.[91]
Like many of the antidepressants, bupropion is associated with the potential risk for causing anxiety as a side effect. How common or severe this anxiety is remains to be fully discovered. Some studies show that it has similar anxiety side effects to the SSRI antidepressants, while others show an elevated risk for akathisia, or extreme anxiety, as high as 13.9%, or three times that from sertraline.[92]
Psychotic symptoms associated with bupropion are rare. They may include delusions, hallucinations, paranoia, and confusion. Often, these symptoms can be reduced or eliminated by decreasing the dose or ceasing treatment.[72][93][94][95][96][97][98][99][100][101] In many of these case reports, psychotic symptoms are associated with such risk factors as old age, the history of bipolar disorder or psychosis, and concomitant medications, for example, lithium or benzodiazepines.[102] Most researchers have attributed the psychosis to bupropion's dopaminergic action.[99][103]
According to several case reports, stopping bupropion abruptly may result in discontinuation syndrome expressed as dystonia, irritability, anxiety, mania, headache, aches and pains.[104][105][106][107] The prescribing information recommends dose tapering after bupropion has been used for seasonal affective disorder;[72] however it states that dose tapering is not required when discontinuing treatment for smoking cessation.[108]
Overdose
Overdose of bupropion results in significant clinical effects in over one-third of cases.[109] The most common symptoms include sinus tachycardia, hypertension, drowsiness, lethargy, agitation, nausea and vomiting, and in particular delirium and seizures.[109][110][111] Less commonly additional symptoms include auditory and visual hallucinations,[112] coma,[111] and ECG changes such as conduction disturbance or arrhythmia.[113][114][115]
In the majority of childhood exploratory ingestions involving one or two tablets, children will remain asymptomatic.[116][117] In teenagers and adults seizures are more commonly observed with the seizure rate increasing tenfold with doses of 600 mg daily.[9] One overdose study suggested a dose-dependent relationship with seizures; patients ingesting more than 4.5 g were likely to have a seizure and nearly all patients ingesting more than 9 g had a seizure.[109]
There is no specific antidote for bupropion; treatment is supportive, and focuses on maintaining airway patency and controlling seizures with high dose intravenous benzodiazepines or barbiturates if seizures are refractory to benzodiazepines.[110] Gastric decontamination may be of little benefit given the risk of seizures and aspiration[110] but activated charcoal is recommended.[109] Additionally, whole bowel irrigation should be undertaken in those ingesting sustained release formulations.[110] Toxic effects may be delayed in onset, with seizures developing as late as 32 hours.[110] Subsequently patients should undergo electroencephalographic monitoring for 48 hours.[72]
Bupropion overdose rarely results in death, although cases have been reported.[113][118][119] Fatalities are typically associated with large overdosage and related to metabolic acidosis and hypoxia as complications of status epilepticus with associated cardiorespiratory arrest.[120] There is one published case report of successful treatment of refractory cardiac arrest in overdose of bupropion and lamotrigine using lipid rescue.[121]
Interactions
Since bupropion is metabolized to hydroxybupropion by the CYP2B6 enzyme, drug interactions with CYP2B6 inhibitors are possible: this includes medications like paroxetine, sertraline, norfluoxetine (the active metabolite of fluoxetine), diazepam, clopidogrel, and orphenadrine. The expected result is the increase of bupropion and decrease of hydroxybupropion blood concentration. The reverse effect (decrease of bupropion and increase of hydroxybupropion) can be expected with CYP2B6 inducers, such as carbamazepine, clotrimazole, rifampicin, ritonavir, St John's Wort, phenobarbitone, phenytoin and others.[122]
Bupropion is an inhibitor of CYP2D6.[122] Hydroxybupropion is an inhibitor of that enzyme in vitro.[123] A significant increase in the concentration of some drugs metabolized by CYP2D6 (venlafaxine, desipramine and dextromethorphan, but not fluoxetine or paroxetine) has been observed when they are taken with bupropion.[122][124]
Bupropion lowers the seizure threshold; accordingly, extreme care should be taken when prescribing bupropion with other medications that also lower it, such as antipsychotics, theophylline, steroids, and some tricyclic antidepressants.[72] Its combination with nicotine replacement therapies can elevate blood pressure; since this combination is no more effective than either a nicotine patch or bupropion alone, it is not recommended.
The prescribing information recommends minimizing the use of alcohol, since in rare cases bupropion reduces alcohol tolerance, and because the excessive use of alcohol may lower the seizure threshold.[72] A small study conducted by GlaxoSmithKline indicated that bupropion (100 mg) may counteract the subjective effects of small doses of alcohol (16–32 mL, slightly less than 1–2 standard US drinks). The volunteers reported feeling more sober and clear-headed and less sedated. Bupropion also reduced the detrimental effect of alcohol on auditory vigilance. The combination of bupropion (100 mg) and two drinks of alcohol increased heart rate by six beats per minute, a statistically significant increase.[125]
Detection in biological fluids
Bupropion may be quantitated in blood, plasma or serum to monitor therapeutic use, confirm a diagnosis of poisoning or assist in a medicolegal death investigation. The drug is unstable in biological fluids at room temperature and therefore specimens should be maintained at the coldest possible temperature prior to analysis.[126]
Mechanism of action
Bupropion is a dopamine and norepinephrine reuptake inhibitor and releaser.[127] It is about twice as potent an inhibitor of dopamine reuptake than of norepinephrine reuptake.[128] Besides reuptake inhibition of dopamine and norepinephrine, bupropion also causes the release of dopamine and norepinephrine.[129] As bupropion is rapidly converted in the body into several metabolites with differing activity, its action cannot be understood without reference to its metabolism. The occupancy of dopamine transporter (DAT) by bupropion and its metabolites in the human brain as measured by positron emission tomography was 6–22% in an independent study[130] and 12–35% according to GlaxoSmithKline researchers.[131] Based on analogy with serotonin reuptake inhibitors, higher than 50% inhibition of DAT would be needed for the dopamine reuptake mechanism to be a major mechanism of the drug's action. By contrast, approximately 65% occupancy or greater of DAT is required to achieve euphoria and reach abuse potential.[132] However recent research indicates that dopamine is inactivated by norepinephrine reuptake in the frontal cortex, which largely lacks dopamine transporters, therefore bupropion can increase dopamine neurotransmission in this part of the brain, and this may be one possible explanation for any additional dopaminergic effects.[133] Bupropion does not inhibit monoamine oxidase or serotonin reuptake. However, it has been shown to indirectly enhance the firing of serotonergic neurons, via activation of downstream norepinephrine flow. Bupropion has also been shown to act as a noncompetitive nicotinic antagonist.[127] The degree of inhibition of α3β4 receptors correlates well with the decrease in self-administration of morphine and methamphetamine in rats,[134] and may be relevant to the effect of bupropion on nicotine addiction. The drug is supplied as a racemic mixture, and the activities of the individual enantiomers were not found to differ.[135]
Two SAR studies date from 2009[136] and 2010.[137]
Pharmacokinetics

Bupropion is metabolized in the liver.
It has several active metabolites: R,R-hydroxybupropion, S,S-hydroxybupropion, threo-hydrobupropion and erythro-hydrobupropion, which are further metabolized to inactive metabolites and eliminated through excretion into the urine. Pharmacological data on bupropion and its metabolites are presented in Table 1. Bupropion is known to weakly inhibit the α1 adrenergic receptor, with a 14% potency of its dopamine uptake inhibition, and the H1 receptor, with a 9% potency.[138]
The biological activity of bupropion can be attributed to a significant degree to its active metabolites, in particular to S,S-hydroxybupropion. GlaxoSmithKline developed this metabolite as a separate drug called radafaxine,[139] but discontinued development in 2006 due to "an unfavourable risk/benefit assessment".[140]
Bupropion is metabolized to hydroxybupropion by CYP2B6, an isoenzyme of the cytochrome P450 system. Alcohol causes an increase of CYP2B6 in the liver, and persons with a history of alcohol use metabolize bupropion faster. The mechanism of formation of erythro-hydrobupropion and threo-hydrobupropion has not been studied but is probably mediated by one of the carbonyl reductase enzymes. The metabolism of bupropion is highly variable: the effective doses of bupropion received by persons who ingest the same amount of the drug may differ by as much as 5.5 times (and the half-life from 3 to 16 hours), and of hydroxybupropion by as much as 7.5 times (and the half-life from 12 to 38 hours).[141][142] Based on this, some researchers have advocated monitoring of the blood level of bupropion and hydroxybupropion.[143]
There are significant interspecies differences in the metabolism of bupropion, with guinea pigs' metabolism of the drug being closest to that of humans compared to mice and rats.[144] Particular caution is needed when extrapolating the results of experiments on rats to humans since hydroxybupropion, the main metabolite of bupropion in humans, is absent in rats.[145]
There have been two reported cases of false-positive urine amphetamine tests in persons taking bupropion. As substituted phenethylamines encompass the substituted amphetamines where bupropion is described as β-Keto-3-chloro-N-tert-butylamphetamine, this is likely the cause of a positive result shown in drug-screening tests on those taking the medicine. More specific follow-up tests were negative.[146][147]
| Exposure (concentration over time; bupropion exposure = 100%) and half-life | |||||
| Bupropion | R,R- Hydroxy bupropion |
S,S- Hydroxy bupropion |
Threo- hydro bupropion |
Erythro- hydro bupropion | |
|---|---|---|---|---|---|
| Exposure | 100% | 800% | 160% | 310% | 90% |
| Half-life | 10 h (IR) 17 h (SR) |
21 h | 25 h | 26 h | 26 h |
| Inhibition potency (potency of DA uptake inhibition by bupropion = 100%) | |||||
| DA uptake | 100% | 0% (rat) | 70% (rat) | 4% (rat) | No data |
| NE uptake | 27% | 0% (rat) | 106% (rat) | 16% (rat) | No data |
| 5HT uptake | 2% | 0% (rat) | 4%(rat) | 3% (rat) | No data |
| α3β4 nicotinic | 53% | 15% | 10% | 7% (rat) | No data |
| α4β2 nicotinic | 8% | 3% | 29% | No data | No data |
| α1* nicotinic | 12% | 13% | 13% | No data | No data |
| DA = dopamine; NE = norepinephrine; 5HT = serotonin. | |||||
Synthesis
Bupropion is synthesized by brominating 3'-chloropropiophenone, followed by nucleophilic substitution with t-butylamine.[152][153] It is a substituted cathinone, or in other words, a derivative of cathinone.
Analogues
A number of bupropion analogues were recently reported by F. Ivy Carroll, et al.[154][155] The aims were two-fold: one smoking cessation,[154] and two, cocaine addiction agents.[155]
In their efforts to find smoking cessation agents, F. Ivy Carroll, et al. also recently (2011) disclosed a number of 2-(substituted phenyl)-3,5,5-trimethylmorpholine analogues.[156]
F. Ivy Carroll, et al. have also produced hydroxy-bupropion analogues, with smoking cessation as the stated application.[157][158][159]
Although structurally unrelated to bupropion, F. Ivy Carroll, et al. recently (2011) disclosed a number of 3-phenyltropanes that had some nAChR activity.[160]
Through reading the above citations it would be possible to draw up possible SARs. To give an example, extending the α-methyl chain in bupropion to C3H7 (presumed n-propyl), improved the [3H]DA IC50 from 658nM to 56nM. Extending the α-methyl to an α-ethyl also improved [3H]DA IC50 to 209nM. According to work done on sibutramine though, the absolute optimum chain length in these compounds is an iso-butyl radical. This has been confirmed by P. Meltzer et al. in his seminal paper on naphyrone analogs.[161] Mark Froimowitz, et al. also explored the effect of replacing the carbomethoxy group in Ritalin with different alkyl groups.[162][163] In fact, the isobutyl analog of bupropion was also studied by RTI, but [3H]DA IC50 was only 140nM.
It can also be readily seen that omission of the hydroxy group in Radafaxine makes DA and NE IC50 shift from 630 and 180nM, respectively, to 220 and 130nM, and 5-HT was 387nM.[156]
Other analogs of desoxy-Radafaxine were also prepared. For example, replacing the m-chloro group with m-fluoro gave an analog that had DA and NE and 5HT IC50 values of 61, 32, and 4600nM, respectively.
A somewhat interesting observation is that in the case of methcathinone, the m-chloro group is not needed as the compound is fully active already. However in the case of bupropion where we have a tert-butyl-N group, omission of the m-chloro halogen gave a compound that had diminished activity as a MAT inhibitor. Moving the m-chloro to the para position also led to a deterioration in the compounds resultant potency.
Regulatory history

Bupropion was invented by Nariman Mehta of Burroughs Wellcome (now GlaxoSmithKline) in 1969, and the US patent for it was granted in 1974.[152] It was approved by the United States Food and Drug Administration (FDA) as an antidepressant on December 30, 1985, and marketed under the name Wellbutrin.[164] However, a significant incidence of seizures at the originally recommended dosage (400–600 mg) caused the withdrawal of the drug in 1986. Subsequently, the risk of seizures was found to be highly dose-dependent, and bupropion was re-introduced to the market in 1989 with a maximum recommended dose of 450 mg/day.[citation needed]

In 1996, the FDA approved a sustained-release formulation of bupropion called Wellbutrin SR, intended to be taken twice a day (as compared with three times a day for immediate-release Wellbutrin).[165] In 2003, the FDA approved another sustained-release formulation called Wellbutrin XL, intended for once-daily dosing. Wellbutrin SR and XL are available in generic form in the United States, while in Canada, only the SR formulation is available in generic form. In 1997, bupropion was approved by the FDA for use as a smoking cessation aid under the name Zyban.[165] In 2006, Wellbutrin XL was similarly approved as a treatment for seasonal affective disorder.[166]
In 2012, the U.S. Justice Department announced that GlaxoSmithKline had agreed to plead guilty and pay a $3-billion fine, in part for promoting the unapproved use of Wellbutrin for weight loss and sexual dysfunction.[167]
In April 2008, the FDA approved a formulation of bupropion as a hydrobromide salt instead of a hydrochloride salt, to be sold under the name Aplenzin by Sanofi-Aventis.[168]
On October 11, 2007, two providers of consumer information on nutritional products and supplements, ConsumerLab.com and The People's Pharmacy, released the results of comparative tests of different brands of bupropion.[169] The People's Pharmacy received multiple reports of increased side effects and decreased efficacy of generic bupropion, which prompted it to ask ConsumerLab.com to test the products in question. The tests showed that "one of a few generic versions of Wellbutrin XL 300 mg, sold as Budeprion XL 300 mg, didn't perform the same as the brand-name pill in the lab."[170] The FDA investigated these complaints and concluded that the Budeprion XL is equivalent to Wellbutrin XL in regard to bioavailability of bupropion and its main active metabolite hydroxybupropion. The FDA also said that coincidental natural mood variation is the most likely explanation for the apparent worsening of depression after the switch from Wellbutrin XL to Budeprion XL.[171] On October 3, 2012, however, the FDA reversed this opinion, announcing that "Budeprion XL 300 mg fails to demonstrate therapeutic equivalence to Wellbutrin XL 300 mg."[172][173] The FDA did not test the bioequivalence of any of the other generic versions of Wellbutrin XL 300 mg, but requested that the four manufacturers submit data on this question to the FDA by May, 2013.[174]
In France, marketing authorization was granted for Zyban on August 3, 2001, with a maximum daily dose of 300 mg;[175] only sustained-release bupropion is available, and only as a smoking cessation aid. Bupropion was granted a licence for use in adults with major depression in the Netherlands in early 2007, with GlaxoSmithKline expecting subsequent approval in other European countries.[176]
Extended-release formulations
Brand-name and generic bupropion tablets are available in three formulations, each as the hydrochloride salt: immediate release (Wellbutrin), sustained release (Wellbutrin SR), and extended release (Wellbutrin XL or XR). "Sustained release" and "extended release" are generally interchangeable terms, but in this case Wellbutrin SR is intended for twice-daily dosing and Wellbutrin XL is intended for once-daily dosing. Not all generics have retained this naming scheme, and the United States Pharmacopeia requires all prolonged-release drug formulations (including generics for Wellbutrin SR) to be labeled "extended release", which has caused confusion and medication errors.[177][178] According to GlaxoSmithKline, the SR tablets should not be split.[179] Merck indicates that splitting a 150 mg Wellbutrin SR tablet decreases the time to peak levels, but that it will retain its sustained-release characteristics.[180]
Society and culture
Recreational use
According to the US government classification of psychiatric medications, bupropion is "non-abusable".[181]
Two studies on drug abusers indicated that the subjective effects of bupropion are markedly different from those of amphetamine.[182][183] Healthy volunteers trained to discriminate amphetamine and placebo recognized bupropion (400 mg) as amphetamine 20% of the time, compared to 10% for placebo and 75% for methylphenidate (20 mg). They also reported feeling alert, vigorous, elated and energetic, reflecting the general stimulating properties of bupropion. In contrast to amphetamine and methylphenidate, there was no feeling of "liking the drug" and no desire to take it again.[184] A comparison of bupropion SR (150 mg) and caffeine (178 mg) indicated that caffeine may have higher abuse liability since it resulted in more reports of pleasant feelings and a "high" than bupropion.[185]
There have been four reports of bupropion abuse in the literature. Three cases described teenagers crushing and insufflating (snorting) the drug, two of them resulting in seizures.[186][187][188] An additional case was reported where a teenager ingested bupropion believing the drug to be a stimulant.[189] A newspaper article on medication abuse in prisons mentioned bupropion as one of the psychotropic medications commonly abused by inmates.[190]
Animal research
In animal studies, squirrel monkeys[191] and rats[192] maintained the intravenous self-administration of bupropion. Significant interspecies differences of bupropion metabolism exist, particularly between rats and humans.[145]
References
- ^ "FDA-sourced list of all drugs with black box warnings (Use Download Full Results and View Query links.)". nctr-crs.fda.gov. FDA. Retrieved 22 Oct 2023.
- ^ entry "Bupropion" at "PubMed Health" (from the US government agency NIH, National Institutes of Health, retrieved 2011-04-07.
- ^ The INN originally assigned in 1974 by the World Health Organization was "amfebutamone". In 2000, the INN was reassigned as bupropion. See World Health Organization (2000). "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 83" (PDF). WHO Drug Information. 14 (2). Archived from the original (PDF) on 2011-05-31. Retrieved 2009-06-22.
- ^ Terry P, Katz JL (1997). "Dopaminergic mediation of the discriminative stimulus effects of bupropion." Psychopharmacology (Berl). 134 (2): 201–12. doi:10.1007/s002130050443. PMID 9399385.
- ^ Learned-Coughlin SM, Bergstrom M, Savitcheva I, Ascher J, Schmith VD, Langstrom B (2003). "In vivo activity of bupropion at the human dopamine transporter." Biol Psychiatry. 54 (8): 800–5. doi:10.1016/S0006-3223(02)01834-6. PMID 14550679.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Slemmer J E, Martin R M, Damaj M I (2000). "Bupropion is a Nicotinic Antagonist". J Pharmacol Exp Ther. 295 (1): 321–327. PMID 10991997.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Fryer J D, Lukas R J (1999). "Noncompetitive functional inhibition at diverse, human nicotinic acetylcholine receptor subtypes by bupropion, phencyclidine, and ibogaine". J Pharmacol Exp Ther. 288 (6): 88–92. PMID 9862757.
- ^ The bupropion prescriptions were calculated as a total of prescriptions for Wellbutrin XL, Budeprion XL, Budeprion SR, Bupropion SR and Bupropion ER using data from the charts for generic and brand-name drugs, see: "Top 200 Generic Drugs by Units in 2007" (PDF). Drug Topics. 2008-02-18. Retrieved 2008-03-30. and Verispan (2008-02-18). "Top 200 Brand Drugs by Units in 2007" (PDF). Drug Topics. Retrieved 2008-03-30.
- ^ a b Johnston JA, Lineberry CG, Ascher JA, Davidson J, Khayrallah MA, Feighner JP, Stark P (1991). "A 102-center prospective study of seizure in association with bupropion". The Journal of clinical psychiatry. 52 (11): 450–6. PMID 1744061.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b For a short review, see: Zimmerman M, Posternak MA, Attiullah N, Friedman M, Boland RJ, Baymiller S, Berlowitz SL, Rahman S, Uy KK, Singer S, Chelminski I, Thongy, T (2005). "Dr. Zimmerman and colleagues reply to MJ Menaster". J Clin Psychiatry. 66 (10): 1336–9.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Fava M, Rush AJ, Thase ME, Clayton A, Stahl SM, Pradko JF, Johnston JA. (2005). "15 years of clinical experience with bupropion HCl: from bupropion to bupropion SR to bupropion XL" (PDF). Prim Care Companion J Clin Psychiatry. 7 (3): 106–113. doi:10.4088/PCC.v07n0305. PMC 1163271. PMID 16027765. Retrieved 2008-12-30.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Thase ME, Haight BR, Richard N, Rockett CB, Mitton M, Modell JG, VanMeter S, Harriett AE, Wang Y (2005). "Remission rates following antidepressant therapy with bupropion or selective serotonin reuptake inhibitors: a meta-analysis of original data from 7 randomized controlled trials". J Clin Psychiatry. 66 (6): 974–981. doi:10.4088/JCP.v66n0803. PMID 16086611.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Clayton AH, Croft HA, Horrigan JP, Wightman DS, Krishen A, Richard NE, Modell JG (2006). "Bupropion extended release compared with escitalopram: effects on sexual functioning and antidepressant efficacy in 2 randomized, double-blind, placebo-controlled studies". J Clin Psychiatry. 67 (5): 736–746. doi:10.4088/JCP.v67n0507. PMID 1684162.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Thase ME, Clayton AH, Haight BR, Thompson AH, Modell JG, Johnston JA (2006). "A double-blind comparison between bupropion XL and venlafaxine XR: sexual functioning, antidepressant efficacy, and tolerability". J Clin Psychopharmacol. 26 (5): 482–488. doi:10.1097/01.jcp.0000239790.83707.ab. PMID 16974189.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ For the review, see: Clayton AH (2003). "Antidepressant-Associated Sexual Dysfunction: A Potentially Avoidable Therapeutic Challenge". Primary Psychiatry. 10 (1): 55–61.
- ^ For another review, see: Kanaly KA, Berman JR (2002). "Sexual side effects of SSRI medications: potential treatment strategies for SSRI-induced female sexual dysfunction". Curr Women's Health Rep. 2 (6): 409–16. PMID 12429073.
- ^ Baldwin DS, Papakostas GI (2006). "Symptoms of Fatigue and Sleepiness in Major Depressive Disorder". J Clin Psychiatry. 67 (suppl 6): 9–15. PMID 16848671.
- ^ Papakostas, GI; Stahl, SM; Krishen, A; Seifert, CA; Tucker, VL; Goodale, EP; Fava, M (2008). "Efficacy of Bupropion and the Selective Serotonin Reuptake Inhibitors in the Treatment of Major Depressive Disorder With High Levels of Anxiety (Anxious Depression): A Pooled Analysis of 10 Studies". J Clin Psychiatry. 69 (8): e1–e6. PMID 18605812.
- ^ For the most recent review, see: Zisook S, Rush AJ, Haight BR, Clines DC, Rockett CB (2006). "Use of bupropion in combination with serotonin reuptake inhibitors". Biol Psychiatry. 59 (3): 203–10. doi:10.1016/j.biopsych.2005.06.027. PMID 16165100.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Spier SA (1998). "Use of bupropion with SRIs and venlafaxine". Depression and Anxiety. 7 (2): 73–5. doi:10.1002/(SICI)1520-6394(1998)7:2<73::AID-DA4>3.0.CO;2-6. PMID 9614595.
- ^ a b Bodkin JA, Lasser RA, Wines JD, Gardner DM, Baldessarini RJ (1997). "Combining serotonin reuptake inhibitors and bupropion in partial responders to antidepressant monotherapy". The Journal of clinical psychiatry. 58 (4): 137–45. doi:10.4088/JCP.v58n0401. PMID 9164423.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Trivedi MH, Fava M, Wisniewski SR, Thase ME, Quitkin F, Warden D, Ritz L, Nierenberg AA, Lebowitz BD, Biggs MM, Luther JF, Shores-Wilson K, Rush AJ (2006). "Medication augmentation after the failure of SSRIs for depression". N. Engl. J. Med. 354 (12): 1243–52. doi:10.1056/NEJMoa052964. PMID 16554526.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Rush AJ, Trivedi MH, Wisniewski SR, Stewart JW, Nierenberg AA, Thase ME, Ritz L, Biggs MM, Warden D, Luther JF, Shores-Wilson K, Niederehe G, Fava M (2006). "Bupropion-SR, sertraline, or venlafaxine-XR after failure of SSRIs for depression". N. Engl. J. Med. 354 (12): 1231–42. doi:10.1056/NEJMoa052963. PMID 16554525.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b Trivedi MH, Rush AJ, Carmody TJ, Donahue RM, Bolden-Watson C, Houser TL, Metz A. (2001). "Do bupropion SR and sertraline differ in their effects on anxiety in depressed patients?". Journal of Clinical Psychiatry. 62 (10): 776–81. doi:10.4088/JCP.v62n1005. PMID 11816866.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Sheehan DV, Davidson J, Manschreck T, Van Wyck Fleet J. (1983). "Lack of efficacy of a new antidepressant (bupropion) in the treatment of panic disorder with phobias". Journal of Clinical Psychopharmacology. 3 (1): 28–31. PMID 6403599.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Feighner J, Hendrickson G, Miller L, Stern W. (1986). "Double-Blind Comparison of Doxepin Versus Bupropion in Outpatients with a Major Depressive Disorder". Journal of Clinical Psychopharmacology. 6 (1): 27–32. PMID 3081600.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Settle EC. (1998). "Bupropion sustained release: side effect profile". Journal of Clinical Psychopharmacology. 59 (Supplement 4): 32–6. PMID 9554319.
- ^ a b Tonnesen P, Tonstad S, Hjalmarson A, Lebargy F, Van Spiegel P I, Hider A, Sweet R, Townsend J (2003). "A multicentre, randomized, double-blind, placebo-controlled, 1-year study of bupropion SR for smoking cessation". J Intern Med. 254 (2): 184–192. doi:10.1046/j.1365-2796.2003.01185.x. PMID 12859700.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Wu P, Wilson K, Dimoulas P, Mills E J (2006). "Effectiveness of smoking cessation therapies: a systematic review and meta-analysis". BMC Public Health. 6: 300–315. doi:10.1186/1471-2458-6-300. PMC 1764891. PMID 17156479.
{{cite journal}}: CS1 maint: multiple names: authors list (link) CS1 maint: unflagged free DOI (link) - ^ Jorenby D E, Hays J T, Rigotti N A, Azoulay S, Watsky E J, Williams K E, Billing C B, Gong J, Reeves K R (2006). "Efficacy of varenicline, an alpha4beta2 nicotinic acetylcholine receptor partial agonist, vs placebo or sustained-release bupropion for smoking cessation: a randomized controlled trial". JAMA. 296 (1): 56–63. doi:10.1001/jama.296.1.56. PMID 16820547.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Serretti A, Chiesa A (2009). "Treatment-emergent sexual dysfunction related to antidepressants: a meta-analysis". J Clin Psychopharmacol. 29 (3): 259–66. doi:10.1097/JCP.0b013e3181a5233f. PMID 19440080.
- ^ Dording CM, Mischoulon D, Petersen TJ, Kornbluh R, Gordon J, Nierenberg AA, Rosenbaum JE, Fava M. (2002). "The pharmacologic management of SSRI-induced side effects: a survey of psychiatrists". Ann Clin Psychiatry. 14 (3): 143–7. doi:10.3109/10401230209147450. PMID 12585563.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Walker PW, Cole JO, Gardner EA, Hughes AR, Johnston JA, Batey SR, Lineberry CG (1993). "Improvement in fluoxetine-associated sexual dysfunction in patients switched to bupropion". J Clin Psychiatry. 54 (12): 459–65. PMID 8276736.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Dobkin RD, Menza M, Marin H, Allen LA, Rousso R, Leiblum SR (2006). "Bupropion improves sexual functioning in depressed minority women: an open-label switch study". J Clin Psychiatry. 26 (1): 21–6. PMID 16415700.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Masand PS, Ashton AK, Gupta S, Frank B (2001). "Sustained-release bupropion for selective serotonin reuptake inhibitor-induced sexual dysfunction: a randomized, double-blind, placebo-controlled, parallel-group study". Am J Psychiatry. 158 (5): 805–807. doi:10.1176/appi.ajp.158.5.805. PMID 11329407.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ DeBattista C, Solvason B, Poirier J, Kendrick E, Loraas E (2005). "A placebo-controlled, randomized, double-blind study of adjunctive bupropion sustained release in the treatment of SSRI-induced sexual dysfunction". J Clin Psychiatry. 66 (7): 844–8. doi:10.4088/JCP.v66n0706. PMID 16013899.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Ashton AK, Rosen RC (1998). "Bupropion as an antidote for serotonin reuptake inhibitor-induced sexual dysfunction". J Clin Psychiatry. 59 (3): 112–5. doi:10.4088/JCP.v59n0304. PMID 9541153.
- ^ Clayton AH, Warnock JK, Kornstein SG, Pinkerton R, Sheldon-Keller A, McGarvey EL (2004). "A placebo-controlled trial of bupropion SR as an antidote for selective serotonin reuptake inhibitor-induced sexual dysfunction". J Clin Psychiatry. 65 (1): 62–7. doi:10.4088/JCP.v65n0110. PMID 14744170.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Crenshaw TL, Goldberg JP, Stern WC (1987). "Pharmacologic modification of psychosexual dysfunction". J Sex Marital Ther. 13 (4): 239–52. doi:10.1080/00926238708403896. PMID 3121861.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Segraves RT, Croft H, Kavoussi R, Ascher JA, Batey SR, Foster VJ, Bolden-Watson C, Metz A (2001). "Bupropion sustained release (SR) for the treatment of hypoactive sexual desire disorder (HSDD) in nondepressed women". J Sex Marital Ther. 27 (3): 303–16. doi:10.1080/009262301750257155. PMID 11354935.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Segraves RT, Clayton A, Croft H, Wolf A, Warnock J. (2004). "Bupropion sustained release for the treatment of hypoactive sexual desire disorder in premenopausal women". J Clin Psychopharmacol. 24 (3): 339–42. doi:10.1097/01.jcp.0000125686.20338.c1. PMID 15118489.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Mathias C, Cardeal Mendes CM, Ponde de Sena E, Dias de Moraes E, Bastos C, Braghiroli MI, Nunez G, Athanazio R, Alban L, Moore HC, del Giglio A (20060). "An open-label, fixed-dose study of bupropion effect on sexual function scores in women treated for breast cancer". Ann Oncol. 17 (12): 1792–6. doi:10.1093/annonc/mdl304. PMID 16980597.
{{cite journal}}: Check date values in:|year=(help)CS1 maint: multiple names: authors list (link) CS1 maint: year (link) - ^ Modell JG, May RS, Katholi CR (2000). "Effect of bupropion-SR on orgasmic dysfunction in nondepressed subjects: a pilot study". J Sex Marital Ther. 26 (3): 231–40. doi:10.1080/00926230050084623. PMID 10929571.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Cabello F (2006). "Effectiveness of the Treatment of Female Hypoactive Sexual Desire Disorder". J Sex Research. Retrieved 2007-04-05.
- ^ Labbate LA, Brodrick PS, Nelson RP, Lydiard RB, Arana GW (2001). "Effects of bupropion sustained-release on sexual functioning and nocturnal erections in healthy men". J Clin Psychopharmacol. 21 (1): 99–103. doi:10.1097/00004714-200102000-00018. PMID 11199957.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Li Z, Maglione M, Tu W, Mojica W, Arterburn D, Shugarman LR, Hilton L, Suttorp M, Solomon V, Shekelle PG, Morton SC. (2005). "Meta-analysis: pharmacologic treatment of obesity". Ann Intern Med. 142 (7): 532–46. PMID 15809465.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Klonoff DC, Greenway F (2008). "Drugs in the pipeline for the obesity market". J Diabetes Sci Technol. 2 (5): 913–8. PMC 2769782. PMID 19885278.
- ^ Wilens TE, Haight BR, Horrigan JP, Hudziak JJ, Rosenthal NE, Connor DF, Hampton KD, Richard NE, Modell JG (2005). "Bupropion XL in adults with attention-deficit/hyperactivity disorder: a randomized, placebo-controlled study". Biol. Psychiatry. 57 (7): 793–801. doi:10.1016/j.biopsych.2005.01.027. PMID 15820237.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b For the review, see: Cantwell DP (1998). "ADHD through the life span: the role of bupropion in treatment". The Journal of clinical psychiatry. 59 Suppl 4: 92–4. PMID 9554326.
- ^ Steven Pliszka; et al. (2007). "Practice Parameter For The Assessment And Treatment Of Children And Adolescents With Attention-Deficit/Hyperactivity Disorder" (PDF). American Academy of Child and Adolescent Psychiatry. p. 16. Retrieved 2011-01-25.
{{cite web}}: Explicit use of et al. in:|author=(help) - ^ Pliszka SR; et al. (2006). "The Texas Children's Medication Algorithm Project: attention-deficit/hyperactivity disorder". Texas Department of State Health Services. Retrieved 2011-01-25.
{{cite web}}: Explicit use of et al. in:|author=(help) - ^ "Algorithm Stages Flowsheets. ADHD with no significant comorbidity algorithm" (PDF). Texas Department of State Health Services. 2006. Archived from the original (PDF) on August 22, 2006. Retrieved 2011-01-25.
{{cite web}}: Unknown parameter|month=ignored (help) - ^ Monuteaux MC, Spencer TJ, Faraone SV, Wilson AM, Biederman J (2007). "A randomized, placebo-controlled clinical trial of bupropion for the prevention of smoking in children and adolescents with attention-deficit/hyperactivity disorder". The Journal of clinical psychiatry. 68 (7): 1094–101. doi:10.4088/JCP.v68n0718. PMID 17685748.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Kampman KM (2008). "The search for medications to treat stimulant dependence". Addict Sci Clin Pract. 4 (2): 28–35. PMC 2797110. PMID 18497715.
- ^ "First drug for seasonal depression". FDA Consumer. 40 (5). U.S. Food and Drug Administration (FDA): 7. 2006. PMID 17328102.
- ^ Modell JG, Rosenthal NE, Harriett AE, Krishen A, Asgharian A, Foster VJ, Metz A, Rockett CB, Wightman DS (2005). "Seasonal affective disorder and its prevention by anticipatory treatment with bupropion XL". Biol Psychiatry. 58 (8): 658–67. doi:10.1016/j.biopsych.2005.07.021. PMID 16271314.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b Kane S, Altschuler EL, Kast RE (2003). "Crohn's disease remission on bupropion". Gastroenterology. 125 (4): 1290. doi:10.1016/j.gastro.2003.02.004. PMID 14552325.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Kast RE, Altschuler EL (2001). "Remission of Crohn's disease on bupropion". Gastroenterology. 121 (5): 1260–1. doi:10.1053/gast.2001.29467. PMID 11706830.
- ^ Altschuler EL, Kast RE (2003). "Bupropion in psoriasis and atopic dermatitis: decreased tumor necrosis factor-alpha?". Psychosom Med. 65 (4): 719. doi:10.1097/01.PSY.0000073874.55003.EE. PMID 12883127.
- ^ Modell JG, Boyce S, Taylor E, Katholi C (2002). "Treatment of atopic dermatitis and psoriasis vulgaris with bupropion-SR: a pilot study". Psychosom Med. 64 (5): 835–40. doi:10.1097/01.PSY.0000021954.59258.9B. PMID 12271115.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Goetz CG, Tanner CM, Klawans HL (1984). "Bupropion in Parkinson's disease". Neurology. 34 (8): 1092–4. PMID 6431314.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ American Psychiatric Association (2000). "Practice guideline for the treatment of patients with major depressive disorder. Third edition" (PDF). p. 37. Retrieved 2011-01-25.
- ^ For the review indicating that antidepressants are not better than placebo, see: Sachs GS, Nierenberg AA, Calabrese JR, Marangell LB, Wisniewski SR, Gyulai L, Friedman ES, Bowden CL, Fossey MD, Ostacher MJ, Ketter TA, Patel J, Hauser P, Rapport D, Martinez JM, Allen MH, Miklowitz DJ, Otto MW, Dennehy EB, Thase ME (2007). "Effectiveness of adjunctive antidepressant treatment for bipolar depression". N. Engl. J. Med. 356 (17): 1711–22. doi:10.1056/NEJMoa064135. PMID 17392295.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b For the review in favor of the antidepressant use, see: Gijsman HJ, Geddes JR, Rendell JM, Nolen WA, Goodwin GM (2004). "Antidepressants for bipolar depression: a systematic review of randomized, controlled trials". The American Journal of Psychiatry. 161 (9): 1537–47. doi:10.1176/appi.ajp.161.9.1537. PMID 15337640.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ For the guidelines recommending the use of bupropion with a mood stabilizer, see: Yatham LN, Kennedy SH, O'Donovan C, Parikh SV, MacQueen G, McIntyre RS, Sharma V, Beaulieu S (2006). "Canadian Network for Mood and Anxiety Treatments (CANMAT) guidelines for the management of patients with bipolar disorder: update 2007". Bipolar Disord. 8 (6): 721–39. doi:10.1111/j.1399-5618.2006.00432.x. PMID 17156158.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Katz J, Pennella-Vaughan J, Hetzel RD, Kanazi GE, Dworkin RH (2005). "A randomized, placebo-controlled trial of bupropion sustained release in chronic low back pain". J Pain. 6 (10): 656–61. doi:10.1016/j.jpain.2005.05.002. PMID 16202958.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Urquhart DM, Hoving JL, Assendelft WW, Roland M, van Tulder MW (2008). Urquhart, Donna M (ed.). "Cochrane Database of Systematic Reviews". Cochrane Database Syst Rev (1): CD001703. doi:10.1002/14651858.CD001703.pub3. PMID 18253994.
{{cite journal}}:|chapter=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Semenchuk MR, Davis B (2000). "Efficacy of sustained-release bupropion in neuropathic pain: an open-label study". The Clinical Journal of Pain. 16 (1): 6–11. doi:10.1097/00002508-200003000-00002. PMID 10741812.
- ^ Semenchuk MR, Sherman S, Davis B (2001). "Double-blind, randomized trial of bupropion SR for the treatment of neuropathic pain". Neurology. 57 (9): 1583–8. PMID 11706096.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Shah TH, Moradimehr A (2010). "Bupropion for the Treatment of Neuropathic Pain". The American Journal of Hospice & Palliative Care. 27 (5): 333–6. doi:10.1177/1049909110361229. PMID 20185402.
- ^ a b c d e f g h i j "Wellbutrin XL Prescribing Information" (PDF). GlaxoSmithKline. 2008. Retrieved 2010-01-16.
{{cite web}}: Unknown parameter|month=ignored (help) - ^ Spencer T, Biederman J, Steingard R, Wilens T (1993). "Bupropion exacerbates tics in children with attention-deficit hyperactivity disorder and Tourette's syndrome". J Am Acad Child Adolesc Psychiatry. 32 (1): 211–4. doi:10.1097/00004583-199301000-00030. PMID 8428875.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Pisani F, Oteri G, Costa C, Di Raimondo G, Di Perri R (2002). "Effects of Psychotropic Drugs on Seizure Threshold". Drug Safety. 25 (2): 91–110. doi:10.2165/00002018-200225020-00004. PMID 11888352.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Ahern TH, Javors MA, Eagles DA, Martillotti J, Mitchell HA, Liles LC, Weinshenker D (2006). "The effects of chronic norepinephrine transporter inactivation on seizure susceptibility in mice". Neuropsychopharmacology. 31 (4): 730–8. doi:10.1038/sj.npp.1300847. PMID 16052243.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Alper K, Schwartz KA, Kolts RL, Khan A (2007). "Seizure Incidence in Psychopharmacological Clinical Trials: An Analysis of Food and Drug Administration (FDA) Summary Basis of Approval Reports". Biol Psychiatry. 62 (4): 345–54. doi:10.1016/j.biopsych.2006.09.023. PMID 17223086.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Roose SP, Dalack GW, Glassman AH, Woodring S, Walsh BT, Giardina EG (1991). "Cardiovascular effects of bupropion in depressed patients with heart disease". Am J Psychiatry. 148 (4): 512–6. PMID 1900980.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Aubin HJ (2002). "Tolerability and safety of sustained-release bupropion in the management of smoking cessation". Drugs. 62 Suppl 2: 45–52. doi:10.2165/00003495-200262002-00005. PMID 12109935.
- ^ Wilens TE, Hammerness PG, Biederman J, Kwon A, Spencer TJ, Clark S, Scott M, Podolski A, Ditterline JW, Morris MC, Moore H (2005). "Blood pressure changes associated with medication treatment of adults with attention-deficit/hyperactivity disorder". J Clin Psychiatry. 66 (2): 253–9. doi:10.4088/JCP.v66n0215. PMID 15705013.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Rigotti NA, Thorndike AN, Regan S, McKool K, Pasternak RC, Chang Y, Swartz S, Torres-Finnerty N, Emmons KM, Singer DE (2006). "Bupropion for smokers hospitalized with acute cardiovascular disease". Am J Med. 119 (12): 1080–7. doi:10.1016/j.amjmed.2006.04.024. PMID 17145253.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ van Melle JP, de Jonge P, van den Berg MP, Pot HJ, van Veldhuisen DJ (2006). "Treatment of depression in acute coronary syndromes with selective serotonin reuptake inhibitors". Drugs. 66 (16): 2095–107. doi:10.2165/00003495-200666160-00005. PMID 17112303.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ "Zyban (bupropion hydrochloride) – safety update". Medicines and Healthcare products Regulatory Agency. 2002-07-24. Archived from the original on September 28, 2007. Retrieved 2006-10-07.
- ^ Hubbard R, Lewis S, West J, Smith C, Godfrey C, Smeeth L, Farrington P, Britton J (2005). "Bupropion and the risk of sudden death: a self-controlled case-series analysis using The Health Improvement Network" (Free full text). Thorax. 60 (10): 848–50. doi:10.1136/thx.2005.041798. PMC 1747199. PMID 16055620.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ For the most recent report, see: Alvaro D, Onetti-Muda A, Moscatelli R, Atili AF (2001). "Acute cholestatic hepatitis induced by bupropion prescribed as pharmacological support to stop smoking. A case report". Digestive and liver disease. 33 (8): 703–6. doi:10.1016/S1590-8658(01)80049-9. PMID 11785718.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Levenson JL (1995). "Priapism associated with bupropion treatment". Am J Psychiatry. 152 (5): 813. PMID 7726332.
- ^ a b Levenson M, Holland C. "Antidepressants and Suicidality in Adults: Statistical Evaluation. (Presentation at Psychopharmacologic Drugs Advisory Committee; December 13, 2006)". U.S. Food and Drug Administration (FDA). Retrieved 2007-05-13.
- ^ Hughes JR, Stead LF, Lancaster T (2007). Hughes, John R (ed.). "Cochrane Database of Systematic Reviews". Cochrane Database Syst Rev. 24 (1): CD000031. doi:10.1002/14651858.CD000031.pub3. PMID 17253443.
{{cite journal}}:|chapter=ignored (help)CS1 maint: multiple names: authors list (link) - ^ "Public Health Advisory: FDA Requires New Boxed Warnings for the Smoking Cessation Drugs Chantix and Zyban". U.S. Food and Drug Administration (FDA). 2009-07-01. Retrieved 2009-07-03.
- ^ "Information for Healthcare Professionals: Varenicline (marketed as Chantix) and Bupropion (marketed as Zyban, Wellbutrin, and generics)". U.S. Food and Drug Administration (FDA). 2009-07-01. Retrieved 2009-07-03.
- ^ "The Smoking Cessation Aids Varenicline (Marketed as Chantix) And Bupropion (Marketed as Zyban and Generics) Suicidal Ideation and Behavior" (PDF). Drug Safety Newsletter. 2 (1). U.S. Food and Drug Administration (FDA): 1–4. 2009.
- ^ Post RM, Altshuler LL, Leverich GS, Frye MA, Nolen WA, Kupka RW, Suppes T, McElroy S, Keck PE, Denicoff KD, Grunze H, Walden J, Kitchen CM, Mintz J (2006). "Mood switch in bipolar depression: comparison of adjunctive venlafaxine, bupropion and sertraline". Br J Psychiatry. 189 (2): 124–31. doi:10.1192/bjp.bp.105.013045. PMID 16880481.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ "Wellbutrin – Anxiety Treatment Concerns". Health and Life.
- ^ Golden RN, James SP, Sherer MA, Rudorfer MV, Sack DA, Potter WZ (1985). "Psychoses associated with bupropion treatment". The American Journal of Psychiatry. 142 (12): 1459–62. PMID 3934991.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Johnston JA, Lineberry CG, Frieden CS (1986). "Prevalence of psychosis, delusions, and hallucinations in clinical trials with bupropion". The American Journal of Psychiatry. 143 (9): 1192–3. PMID 3092682.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Golden RN (1988). "Diethylpropion, bupropion, and psychoses". The British Journal of Psychiatry : the Journal of Mental Science. 153 (2): 265–6. doi:10.1192/bjp.153.2.265. PMID 3151278.
- ^ Ames D, Wirshing WC, Szuba MP (1992). "Organic mental disorders associated with bupropion in three patients". The Journal of Clinical Psychiatry. 53 (2): 53–5. PMID 1541606.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Howard WT, Warnock JK (1999). "Bupropion-induced psychosis". The American Journal of Psychiatry. 156 (12): 2017–8. PMID 10588428.
- ^ Neumann M, Livak V, Paul HW, Laux G (2002). "Acute psychosis after administration of bupropion hydrochloride (Zyban)". Pharmacopsychiatry. 35 (6): 247–8. doi:10.1055/s-2002-36393. PMID 12518275.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b Bailey J, Waters S (2008). "Acute psychosis after bupropion treatment in a healthy 28-year-old woman". Journal of the American Board of Family Medicine : JABFM. 21 (3): 244–5. doi:10.3122/jabfm.2008.03.070236. PMID 18467537.
- ^ Javelot, Hervé; Baratta, Alexandre; Weiner, Luisa; Javelot, Thierry; Nonnenmacher, Cathy; Westphal, Jean-Frédéric; Messaoudi, Michaël (2009). "Two acute psychotic episodes after administration of bupropion: a case of involuntary rechallenge". Pharmacy World & Science : PWS. 31 (2): 238–40. doi:10.1007/s11096-008-9272-x. PMID 19039677.
- ^ Grover S, Das PP (2009). "Can bupropion unmask psychosis". Indian Journal of Psychiatry. 51 (1): 53–4. doi:10.4103/0019-5545.44907. PMC 2738405. PMID 19742195.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ Nemeroff, Charles B.; Schatzberg, Alan F. (2006). Essentials of Clinical Psychopharmacology. Washington, D.C: American Psychiatric Publishing. p. 146. ISBN 1-58562-243-5.
{{cite book}}: CS1 maint: multiple names: authors list (link) - ^ Hahn M, Hajek T, Alda M, Gorman JM (2007). "Psychosis induced by low-dose bupropion: sensitization of dopaminergic system by past cocaine abuse?". Journal of Psychiatric Practice. 13 (5): 336–8. doi:10.1097/01.pra.0000290673.32978.e8. PMID 17890983.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Wang, Hy; Chou, Wj; Huang, Ty; Hung, Cf (2007). "Acute dystonia resulting from abrupt bupropion discontinuation". Progress in neuro-psychopharmacology & biological psychiatry. 31 (3): 766–8. doi:10.1016/j.pnpbp.2006.12.003. PMID 17218049.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Berigan, Tr (2002). "Bupropion-Associated Withdrawal Symptoms Revisited: A Case Report". Primary care companion to the Journal of clinical psychiatry. 4 (2): 78. ISSN 1523-5998. PMC 181231. PMID 15014751.
- ^ Berigan, Tr; Harazin, Js (April 1, 1999). "Bupropion-Associated Withdrawal Symptoms: A Case Report". Primary care companion to the Journal of clinical psychiatry. 1 (2): 50–51. ISSN 1523-5998. PMC 181057. PMID 15014696.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Michael, N; Erfurth, A; Bergant, V (2004). "A case report of mania related to discontinuation of bupropion therapy for smoking cessation". The Journal of clinical psychiatry. 65 (2): 277. ISSN 0160-6689. PMID 15003088.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ "PRESCRIBING INFORMATION – ZYBAN (bupropion hydrochloride) Sustained-Release Tablets" (PDF). Retrieved 2010-01-06.
- ^ a b c d Balit CR, Lynch CN, Isbister GK (2003). "Bupropion poisoning: a case series". Med. J. Aust. 178 (2): 61–3. PMID 12526723.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b c d e Buckley NA, Faunce TA (2003). "'Atypical' antidepressants in overdose: clinical considerations with respect to safety". Drug safety: an international journal of medical toxicology and drug experience. 26 (8): 539–51. PMID 12825968.
- ^ a b Spiller HA, Ramoska EA, Krenzelok EP, Sheen SR, Borys DJ, Villalobos D, Muir S, Jones-Easom L (1994). "Bupropion overdose: a 3-year multi-center retrospective analysis". The American journal of emergency medicine. 12 (1): 43–5. doi:10.1016/0735-6757(94)90195-3. PMID 8285970.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Mainie I, McGurk C, McClintock G, Robinson J (2001). "Seizures after buproprion overdose". Lancet. 357 (9268): 1624. doi:10.1016/S0140-6736(00)04770-X. PMID 11386326.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b Shepherd G, Velez LI, Keyes DC (2004). "Intentional bupropion overdoses". The Journal of emergency medicine. 27 (2): 147–51. doi:10.1016/j.jemermed.2004.02.017. PMID 15261357.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Curry SC, Kashani JS, LoVecchio F, Holubek W (2005). "Intraventricular conduction delay after bupropion overdose". The Journal of emergency medicine. 29 (3): 299–305. doi:10.1016/j.jemermed.2005.01.027. PMID 16183450.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Tracey JA, Cassidy N, Casey PB, Ali I (2002). "Bupropion (Zyban) toxicity". Irish medical journal. 95 (1): 23–4. PMID 11928786.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Shepherd G, Velez LI, James DK, Keyes DC (2001). "Pediatric bupropion exposures reported in Texas: 1998–1999 [abstract]". J Toxicol Clin Toxicol. 39: 263.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Colbridge MG, Dargan PI, Jones AL (2002). "Bupropion – the experience of the National Poisons Information Service (London) [abstract]". J Toxicol Clin Toxicol. 40: 398–9.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Harris CR, Gualtieri J, Stark G (1997). "Fatal bupropion overdose". J Toxicol Clin Toxicol. 35 (3): 321–4. doi:10.3109/15563659709001220. PMID 9140330.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Friel PN, Logan BK, Fligner CL (1993). "Three fatal drug overdoses involving bupropion". Journal of analytical toxicology. 17 (7): 436–8. PMID 8309220.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Paoloni R, Szekely I (2002). "Sustained-release bupropion overdose: a new entity for Australian emergency departments". Emergency medicine (Fremantle, W.A.). 14 (1): 109–12. doi:10.1046/j.1442-2026.2002.00295.x. PMID 11993828.
- ^ Sirianni, Archie J.; Osterhoudt, Kevin C.; Calello, Diane P.; Muller, Allison A.; Waterhouse, Marie R.; Goodkin, Michael B.; Weinberg, Guy L.; Henretig, Fred M. (2007). "Use of Lipid Emulsion in the Resuscitation of a Patient With Prolonged Cardiovascular Collapse After Overdose of Bupropion and Lamotrigine". Ann Emerg Med. 51 (4): 412. doi:10.1016/j.annemergmed.2007.06.004. PMID 17766009.
{{cite journal}}: More than one of|pages=and|page=specified (help) - ^ a b c Jefferson JW, Pradko JF, Muir KT (2005). "Bupropion for major depressive disorder: Pharmacokinetic and formulation considerations". Clin Ther. 27 (11): 1685–95. doi:10.1016/j.clinthera.2005.11.011. PMID 16368442.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Hesse LM, Nevkatakrishnan K, Court MH, von Moltke LL, Duan SX, Shader RI, Greenblatt DJ (2000). "2B6 mediates the in vitro hydroxylation of bupropion: potential drug interactions with other antidipressants". Drug Metab Dispos. 28: 1176–1183.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Kennedy SH, McCann SM, Masellis M, McIntyre RS, Raskin J, McKay G, Baker GB (2002). "Combining bupropion SR with venlafaxine, paroxetine, or fluoxetine: a preliminary report on pharmacokinetic, therapeutic, and sexual dysfunction effects". J Clin Psychiatry. 63 (3): 181–6. doi:10.4088/JCP.v63n0302. PMID 11926715.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ "P02-31UK I Examination of Bupropion and Ethanol, Alone and in Combination, on Human Performance Tests, Subjective Rating Scales, EEG and Autonomic Responses" (PDF). GlaxoSmithKline Clinical Trial Register. Bupropion Studies. Retrieved 2011-01-25.
- ^ Baselt, R. Disposition of Toxic Drugs and Chemicals in Man, 8th edition, Biomedical Publications, Foster City, CA, 2008, pp. 193–195 ISBN 0962652350.
- ^ a b Arias HR (2009). "Is the inhibition of nicotinic acetylcholine receptors by bupropion involved in its clinical actions?". Int. J. Biochem. Cell Biol. 41 (11): 2098–108. doi:10.1016/j.biocel.2009.05.015. PMID 19497387.
- ^ Akhtar, Nadeem Masood; Khan, Afreen Bano (2008). "Prescriptions Into Practice: Bupropion". Child and Adolescent Psychopharmacology News. 13 (5): 6. doi:10.1521/capn.2008.13.5.6.
- ^ Arias, Hugo R.; Santamaría, Abel; Ali, Syed F. (2009). "Chapter 9. Pharmacological and Neurotoxicological Actions Mediated By Bupropion and Diethylpropion". New Concepts of Psychostimulant Induced Neurotoxicity. International Review of Neurobiology. Vol. 88. p. 223. doi:10.1016/S0074-7742(09)88009-4. ISBN 9780123745040.
- ^ Meyer J, Goulding V S, Wilson A A, Hussey D, Christensen B K, Houle S (2002). "Bupropion occupancy of the dopamine transporter is low during clinical treatment". Psychopharmacology. 163 (1): 102–105. doi:10.1007/s00213-002-1166-3. PMID 12185406.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Learned-Coughkin S M, Bergstrom M, Savitcheva I, Ascher J, Schmith V D, Langstrom B (2003). "In vivo activity of bupropion at the human dopamine transporter as measured by positron emission tomography". Biol Psychiatry. 54 (8): 800–805. doi:10.1016/S0006-3223(02)01834-6. PMID 14550679.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Howell, LL; Wilcox, KM (2001). "The dopamine transporter and cocaine medication development: Drug self-administration in nonhuman primates" (PDF). The Journal of pharmacology and experimental therapeutics. 298 (1): 1–6. PMID 11408518.
- ^ "Bupropion". Stahlonline.cambridge.org. Cambridge University Press. Retrieved 2010-04-27.
- ^ Glick S D. "Ibogaine Analogues: Drug Development for Addictive Disorders. (Presentation at Addiction Medicine State of the Art 2003 Conference October 8–11, 2003, Radisson-Miyako Hotel, San Francisco)". www.csam-asam.org. Retrieved 2007-03-24.
- ^ Musso, DL; Mehta, NB; Soroko, FE; Ferris, RM; Hollingsworth, EB; Kenney, BT (1993). "Synthesis and evaluation of the antidepressant activity of the enantiomers of bupropion". Chirality. 5 (7): 495–500. doi:10.1002/chir.530050704. PMID 8240925.
- ^ Carroll, FI; Blough, BE; Abraham, P; Mills, AC; Holleman, JA; Wolckenhauer, SA; Decker, AM; Landavazo, A; McElroy, KT (2009). "Synthesis and biological evaluation of bupropion analogues as potential pharmacotherapies for cocaine addiction". Journal of Medical Chemistry. 52 (21): 6768–81. doi:10.1021/jm901189z. PMID 19821577.
- ^ Carroll, FI; Blough, BE; Mascarella, SW; Navarro, HA; Eaton, JB; Lukas, RJ; Damaj, MI (2010). "Synthesis and biological evaluation of bupropion analogues as potential pharmacotherapies for smoking cessation". Journal of Medical Chemistry. 53 (5): 2204–14. doi:10.1021/jm9017465. PMC 2841507. PMID 20158204.
- ^ a b Horst WD, Preskorn SH (1998). "Mechanisms of action and clinical characteristics of three atypical antidepressants: venlafaxine, nefazodone, bupropion". J Affect Disord. 51 (3): 237–54. doi:10.1016/S0165-0327(98)00222-5. PMID 10333980.
- ^ "GlaxoSmithKline (GSK) Reviews Novel Therapeutics For CNS Disorders And Confirms Strong Pipeline Momentum" (Press release). PRNewswire. November 23, 2004. Retrieved 2007-08-18.
- ^ GlaxoSmithKline (July 26, 2006) Template:PDFlink. Press release. Retrieved on 2007-08-18.
- ^ Kirchheiner J, Klein C, Meineke I, Sasse J, Zanger UM, Mürdter TE, Roots I, Brockmöller J (2003). "Bupropion and 4-OH-bupropion pharmacokinetics in relation to genetic polymorphisms in CYP2B6". Pharmacogenetics. 13 (10): 619–26. doi:10.1097/00008571-200310000-00005. PMID 14515060.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Hesse LM, He P, Krishnaswamy S, Hao Q, Hogan K, von Moltke LL, Greenblatt DJ, Court MH (2004). "Pharmacogenetic determinants of interindividual variability in bupropion hydroxylation by cytochrome P450 2B6 in human liver microsomes". Pharmacogenetics. 14 (4): 225–38. doi:10.1097/00008571-200404000-00002. PMID 15083067.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Preskorn SH (1991). "Should bupropion dosage be adjusted based upon therapeutic drug monitoring?". Psychopharmacology bulletin. 27 (4): 637–43. PMID 1813908.
- ^ Suckow R F, Smith T M, Perumal A S, Cooper T B (1986). "Pharmacokinetics of bupropion and metabolites in plasma and brain of rats, mice, and guinea pigs". Drug Metab Dispos. 14 (6): 692–697. PMID 2877828.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b Welch R M, Lai A A, Schroeder D H (1987). "Pharmacological significance of the species differences in bupropion metabolism". Xenobiotica. 17 (3): 287–289. doi:10.3109/00498258709043939. PMID 3107223.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Weintraub D, Linder MW (2000). "Amphetamine positive toxicology screen secondary to bupropion". Depress Anxiety. 12 (1): 53–4. doi:10.1002/1520-6394(2000)12:1<53::AID-DA8>3.0.CO;2-4. PMID 10999247.
- ^ Nixon AL, Long WH, Puopolo PR, Flood JG (1995). "Bupropion metabolites produce false-positive urine amphetamine results". Clin. Chem. 41 (6 Pt 1): 955–6. PMID 7768026.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Johnston AJ, Ascher J, Leadbetter R, Schmith VD, Patel DK, Durcan M, Bentley B (2002). "Pharmacokinetic optimisation of sustained-release bupropion for smoking cessation". Drugs. 62 (Suppl 2): 11–24. doi:10.2165/00003495-200262002-00002. PMID 12109932.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Xu H, Loboz KK, Gross AS, McLachlan AJ (2007). "Stereoselective analysis of hydroxybupropion and application to drug interaction studies". Chirality. 19 (3): 163–70. doi:10.1002/chir.20356. PMID 17167747.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Bondarev ML, Bondareva TS, Young R, Glennon RA (2003). "Behavioral and biochemical investigations of bupropion metabolites". Eur J Pharmacol. 474 (1): 85–93. doi:10.1016/S0014-2999(03)02010-7. PMID 12909199.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Damaj MI, Carroll FI, Eaton JB, Navarro HA, Blough BE, Mirza S, Lukas RJ, Martin BR (2004). "Enantioselective effects of hydroxy metabolites of bupropion on behavior and on function of monoamine transporters and nicotinic receptors". Mol Pharmacol. 66 (3): 675–82. doi:10.1124/mol.104.001313. PMID 15322260.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b Mehta NB (1974-06-25). "United States Patent 3,819,706: Meta-chloro substituted α-butylamino-propiophenones". USPTO. Retrieved 2008-06-02.
- ^ Daniel M. Perrine, Jason T. Ross, Stephen J. Nervi, and Richard H. Zimmerman (2000). "A Short, One-Pot Synthesis of Bupropion" (PDF). J. Chem. Ed. 77 (11): 1479.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b Attention: This template ({{cite pmid}}) is deprecated. To cite the publication identified by PMID 20158204, please use {{cite journal}} with
|pmid=20158204instead. - ^ a b Attention: This template ({{cite pmid}}) is deprecated. To cite the publication identified by PMID 19821577, please use {{cite journal}} with
|pmid=19821577instead. - ^ a b Attention: This template ({{cite pmid}}) is deprecated. To cite the publication identified by PMID 21319801, please use {{cite journal}} with
|pmid=21319801instead. - ^ Attention: This template ({{cite pmid}}) is deprecated. To cite the publication identified by PMID 20576796, please use {{cite journal}} with
|pmid=20576796instead. - ^ Attention: This template ({{cite pmid}}) is deprecated. To cite the publication identified by PMID 20509659, please use {{cite journal}} with
|pmid=20509659instead. - ^ Attention: This template ({{cite pmid}}) is deprecated. To cite the publication identified by PMID 15322260, please use {{cite journal}} with
|pmid=15322260instead. - ^ Attention: This template ({{cite pmid}}) is deprecated. To cite the publication identified by PMID 21319801, please use {{cite journal}} with
|pmid=21319801instead. - ^ Attention: This template ({{cite pmid}}) is deprecated. To cite the publication identified by PMID 16480278, please use {{cite journal}} with
|pmid=16480278instead. - ^ Attention: This template ({{cite pmid}}) is deprecated. To cite the publication identified by PMID 21973080, please use {{cite journal}} with
|pmid=21973080instead. - ^ Attention: This template ({{cite pmid}}) is deprecated. To cite the publication identified by PMID 17228864, please use {{cite journal}} with
|pmid=17228864instead. - ^ Wellbutrin Label and Approval History. U.S. Food and Drug Administration Center for Drug Evaluation and Research. Retrieved on 2007-08-18. Data available for download on FDA website.
- ^ a b "Bupropion Helps People With Schizophrenia Quit Smoking." National Institute on Drug Abuse. Research Findings, Vol. 20, No. 5 (April 2006). Retrieved on August 19, 2007.
- ^ Staff Writer. "Seasonal affective disorder drug Wellbutrin XL wins approval." CNN. June 14, 2006. Retrieved August 19, 2007.
- ^ Thomas, Katie; Schmidt, Michael S. (July 2, 2012). "Glaxo Agrees to Pay $3 Billion in Fraud Settlement". The New York Times.
- ^ Waknine, Yael (2008-05-08). "FDA Approvals: Advair, Relistor, Aplenzin". Medscape. Retrieved 2008-05-09.
- ^ "Generic Drug Equality Questioned". Retrieved 2007-10-13.
- ^ Jacqueline Stenson (2007-10-12). "Report questions generic antidepressant". msnbc.com. Retrieved 2007-10-13.
- ^ "Review of Therapeutic Equivalence: Generic Bupropion XL 300 mg and Wellbutrin XL 300 mg". Archived from the original on 2011-06-06. Retrieved 2008-04-19.
- ^ {{cite web|title=Generic antidepressant not equivalent to original Wellbutrin: FDA|url=http://www.reuters.com/article/2012/10/06/us-fda-antidepressant-idUSBRE8950BE20121006%7Caccessdate=2012-10-09
- ^ "FDA Update: Budeprion XL 300 mg Not Therapeutically Equivalent to Wellbutrin XL 300 mg". Retrieved 2012-10-09.
- ^ http://www.fda.gov/Drugs/DrugSafety/PostmarketDrugSafetyInformationforPatientsandProviders/ucm322161.htm
- ^ "Zyban : sevrage tabagique et sécurité d'emploi" (Press release) (in French). Agence française de sécurité sanitaire des produits de santé. January 18, 2002. Retrieved 2011-01-25.
{{cite press release}}: Unknown parameter|trans_title=ignored (|trans-title=suggested) (help) - ^ GlaxoSmithKline (2007-01-16). "GlaxoSmithKline receives first European approval for Wellbutrin XR" (Press release). GlaxoSmithKline. Retrieved 2011-01-25.
- ^ "Practitioner's Reporting News". United States Pharmacopeia. 2004-09-30. Retrieved 2007-08-20. [dead link]
- ^ Rosack, Jim (2006-03-17). "Company Tries to Clear Up Confusion About Bupropion". Psychiatric News. 41 (6). American Psychiatric Association: 16. Retrieved 2007-08-20.
- ^ wellbutrin sr sustained-release tablets. (PDF) . Retrieved on 2012-01-05.
- ^ "BuPROPion: Drug Information Provided by Lexi-Comp: Merck Manual Professional". Retrieved 2007-06-16.
- ^ "Exhibit 4-3 Abuse Potential of Common Psychiatric Medications". Health Services/Technology Assessment Text (HSTAT). U.S. National Library of Medicine. Retrieved 2011-01-25.
- ^ Miller L, Griffith J (1983). "A comparison of bupropion, dextroamphetamine, and placebo in mixed-substance abusers". Psychopharmacology (Berl.). 80 (3): 199–205. doi:10.1007/BF00436152. PMID 6412263.
- ^ Griffith JD, Carranza J, Griffith C, Miller LL (1983). "Bupropion: clinical assay for amphetamine-like abuse potential". J Clin Psychiatry. 44 (5 Pt 2): 206–8. PMID 6406459.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Rush CR, Kollins SH, Pazzaglia PJ (1998). "Discriminative-stimulus and participant-rated effects of methylphenidate, bupropion, and triazolam in d-amphetamine-trained humans". Experimental and clinical psychopharmacology. 6 (1): 32–44. doi:10.1037/1064-1297.6.1.32. PMID 9526144.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Zernig, Gerald; De Wit, Harriet; Telser, Stefan; Nienhusmeier, Matthias; Wakonigg, Gudrun; Sturm, Katja; Berger, Iris; Kemmler, Georg; Saria, Alois (2004). "Subjective effects of slow-release bupropion versus caffeine as determined in a quasi-naturalistic setting". Pharmacology. 70 (4): 206–15. doi:10.1159/000075550. PMID 15001822.
- ^ Khurshid KA, Decker DH (2004). "Bupropion insufflation in a teenager". J Child Adolesc Psychopharmacol. 14 (1): 157–8. doi:10.1089/104454604773840634. PMID 15142406.
- ^ Lu JJ, Thompson TM, Narunatvanich D, Fischbein CB, Mycyk MB (2007). "Seizure after Nasal Insufflation of Bupropion [abstract]". Clin Toxicol (Phila). 45: 632.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Welsh CJ, Doyon S (2002). "Seizure induced by insufflation of bupropion". N. Engl. J. Med. 347 (12): 951. doi:10.1056/NEJM200209193471222. PMID 12239274.
- ^ McCormick J (2002). "Recreational bupropion abuse in a teenager". Br J Clin Pharmacol. 53 (2): 214. doi:10.1046/j.0306-5251.2001.01538.x. PMC 1874291. PMID 11851650.
- ^ Volpe KD. "Intervention Reduces Psychotropic Abuse in Correctional Facility; CNS News, JUNE 2005, VOLUME: 07:06". Archived from the original on September 29, 2007. Retrieved 2007-05-27.
- ^ Bergman J, Madras BK, Johnson SE, Spealman RD (1989). "Effects of cocaine and related drugs in nonhuman primates. III. Self-administration by squirrel monkeys". J. Pharmacol. Exp. Ther. 251 (1): 150–5. PMID 2529365.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Tella SR, Ladenheim B, Cadet JL (1997). "Differential regulation of dopamine transporter after chronic self-administration of bupropion and nomifensine". J Pharmacol Exp Ther. 281 (1): 508–13. PMID 9103538.
{{cite journal}}: CS1 maint: multiple names: authors list (link)
External links
- Official Wellbutrin website
- List of international brand names for bupropion
- Template:Dmoz
- Wellbutrin Pharmacology, Pharmacokinetics, Studies, Metabolism – Bupropion – RxList Monographs
- NAMI Wellbutrin
- Bupropion article from mentalhealth.com

