Noretynodrel: Difference between revisions
No edit summary |
Lead, pharmacokinetics, etc. |
||
| Line 18: | Line 18: | ||
| legal_status = |
| legal_status = |
||
| routes_of_administration = [[Oral administration|By mouth]] |
| routes_of_administration = [[Oral administration|By mouth]] |
||
| class = [[Progestin]]; [[Progestogen]] |
| class = [[Progestin]]; [[Progestogen]]; [[Estrogen (medication)|Estrogen]] |
||
<!--Pharmacokinetic data--> |
<!--Pharmacokinetic data--> |
||
| bioavailability = |
| bioavailability = |
||
| protein_bound = Noretynodrel: to [[human serum albumin|albumin]] and not to {{abbrlink|SHBG|sex hormone-binding globulin}} or {{abbrlink|CBG|corticosteroid-binding globulin}}<ref name="pmid2170822" /> |
|||
| protein_bound = |
|||
| metabolism = [[Liver]], [[intestine]]s ([[hydroxylation]], [[isomerization]], [[conjugation (biochemistry)|conjugation]])<ref name="pmid2170822" /><ref name="pmid16112947" /> |
|||
| metabolism = |
|||
| metabolites = • 3α-Hydroxynoretynodrel<ref name="pmid22210085" /><br />• 3β-Hydroxynoretynodrel<ref name="pmid22210085" /><br />• [[Norethisterone]]<ref name="pmid22210085" /><ref name="pmid2170822" /><ref name="pmid16112947" /><br />• [[Ethinylestradiol]]<ref name="pmid16112947" /><ref name="Kuhl2011" />• [[Conjugation (biochemistry)|Conjugate]]s<ref name="pmid16112947" /> |
|||
| elimination_half-life = |
|||
| elimination_half-life = Very short (< 30 minutes)<ref name="pmid2256526" /> |
|||
| excretion = |
|||
| excretion = [[Breast milk]]: 1%<ref name="Martindale" /> |
|||
<!--Identifiers--> |
<!--Identifiers--> |
||
| Line 40: | Line 41: | ||
| UNII_Ref = {{fdacite|correct|FDA}} |
| UNII_Ref = {{fdacite|correct|FDA}} |
||
| UNII = 88181ACA0M |
| UNII = 88181ACA0M |
||
| ChEBI_Ref = {{ebicite|correct|EBI}} |
|||
| ChEBI = 34895 |
|||
| ChEMBL_Ref = {{ebicite|correct|EBI}} |
| ChEMBL_Ref = {{ebicite|correct|EBI}} |
||
| ChEMBL = 1387 |
| ChEMBL = 1387 |
||
| KEGG = D05207 |
|||
| synonyms = Norethynodrel; NSC-15432; 5(10)-Norethisterone; 17α-Ethynyl-δ<sup>5(10)</sup>-19-nortestosterone; 17α-Ethynylestr-5(10)-en-17β-ol-3-one |
| synonyms = Norethynodrel; Noretinodrel Norethinodrel; SC-4642; NSC-15432; 5(10)-Norethisterone; 17α-Ethinyl-19-nor-5(10)-testosterone; 17α-Ethynyl-δ<sup>5(10)</sup>-19-nortestosterone; 17α-Ethynylestr-5(10)-en-17β-ol-3-one; 19-Nor-17α-pregn-5(10)-en-20-yn-17β-ol-3-one |
||
<!--Chemical data--> |
<!--Chemical data--> |
||
| Line 53: | Line 57: | ||
| StdInChIKey = ICTXHFFSOAJUMG-SLHNCBLASA-N |
| StdInChIKey = ICTXHFFSOAJUMG-SLHNCBLASA-N |
||
}} |
}} |
||
<!-- Definition and medical uses --> |
|||
'''Noretynodrel''', or '''norethynodrel''', sold under the brand name '''Enovid''' among others, is a [[progestin]] medication which was previously used in [[birth control pill]]s and in the treatment of [[gynecological disorder]]s but is now no longer marketed.<ref name="Martindale">{{cite book |editor=Sweetman, Sean C. |chapter=Sex hormones and their modulators |title=Martindale: The Complete Drug Reference |edition=36th |year=2009 |pages=2120–2121 |publisher=Pharmaceutical Press |location=London|isbn=978-0-85369-840-1|url=https://www.medicinescomplete.com/mc/martindale/2009/9079-k.htm}}</ref><ref name="Jucker2013">{{cite book|author=Jucker|title=Progress in Drug Research / Fortschritte der Arzneimittelforschung / Progrès des recherches pharmaceutiques|url=https://books.google.com/books?id=Y8HzBwAAQBAJ&pg=PA85|date=21 December 2013|publisher=Birkhäuser|isbn=978-3-0348-7065-8|pages=85–88}}</ref><ref name="Marks2010">{{cite book|author=Lara Marks|title=Sexual Chemistry: A History of the Contraceptive Pill|url=https://books.google.com/books?id=_i-s4biQs7MC&pg=PA75|year=2010|publisher=Yale University Press|isbn=0-300-16791-1|pages=74–75}}</ref> It was available both alone and in combination with an [[estrogen (medication)|estrogen]].<ref name="WHO1974" /><ref name="Jucker2013" /><ref name="Marks2010" /> The medication is taken [[oral administration|by mouth]].<ref name="Jucker2013" /> |
|||
<!-- Side effects and mechanism --> |
|||
'''Noretynodrel''', or '''norethynodrel''', is a [[progestin]] of the [[19-nortestosterone]] group and an [[isomer]] of [[norethisterone]].<ref name="Elks2014">{{cite book|author=J. Elks|title=The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies|url=https://books.google.com/books?id=0vXTBwAAQBAJ&pg=PA886|date=14 November 2014|publisher=Springer|isbn=978-1-4757-2085-3|pages=886–}}</ref><ref name="IndexNominum2000">{{cite book|title=Index Nominum 2000: International Drug Directory|url=https://books.google.com/books?id=5GpcTQD_L2oC&pg=PP1|date=January 2000|publisher=Taylor & Francis|isbn=978-3-88763-075-1|pages=1–}}</ref> Noretynodrel was introduced in 1957 in '''Enovid''', a [[combination drug|combination formulation]] of noretynodrel and [[mestranol]], for the treatment of [[gynecological disorder|gynecological]] and [[menstrual disorder]]s.<ref name="Marks2010">{{cite book|author=Lara Marks|title=Sexual Chemistry: A History of the Contraceptive Pill|url=https://books.google.com/books?id=_i-s4biQs7MC&pg=PA75|year=2010|publisher=Yale University Press|isbn=0-300-16791-1|pages=74–75}}</ref> A few years later, in May 1960, Enovid was also approved as the first [[combined oral contraceptive pill|oral contraceptive]].<ref name="Marks2010" /><ref name="Hollinger2007">{{cite book|author=Mannfred A. Hollinger|title=Introduction to Pharmacology, Third Edition|url=https://books.google.com/books?id=qfrLBQAAQBAJ&pg=PA160|date=19 October 2007|publisher=CRC Press|isbn=978-1-4200-4742-4|pages=160–}}</ref> |
|||
Noretynodrel is a progestin, or a [[synthetic compound|synthetic]] [[progestogen]], and hence is an [[agonist]] of the [[progesterone receptor]], the [[biological target]] of progestogens like [[progesterone]].<ref name="pmid16112947" /> It is a relatively weak progestogen.<ref name="WilliamsFoye2002">{{cite book|author1=David A. Williams|author2=William O. Foye|author3=Thomas L. Lemke|title=Foye's Principles of Medicinal Chemistry|url=https://books.google.com/books?id=qLJ6Bs1Qml4C&pg=PA700|date=January 2002|publisher=Lippincott Williams & Wilkins|isbn=978-0-683-30737-5|pages=700–}}</ref> The medication has weak [[estrogen (medication)|estrogen]]ic activity, no or only very weak [[androgen]]ic activity, and no other important [[hormonal agent|hormonal]] activity.<ref name="pmid16112947" /><ref name="Marks2010" /><ref name="RunnebaumRabe2012">{{cite book|author1=Benno Clemens Runnebaum|author2=Thomas Rabe|author3=Ludwig Kiesel|title=Female Contraception: Update and Trends|url=https://books.google.com/books?id=LtT6CAAAQBAJ&pg=PA36|date=6 December 2012|publisher=Springer Science & Business Media|isbn=978-3-642-73790-9|pages=36–}}</ref><ref name="Sloane2002">{{cite book|author=Ethel Sloane|title=Biology of Women|url=https://books.google.com/books?id=kqcYyk7zlHYC&pg=PA426|year=2002|publisher=Cengage Learning|isbn=0-7668-1142-5|pages=426–}}</ref> It is a [[prodrug]] of various [[active metabolite]]s in the body, such as [[norethisterone]] among others.<ref name="pmid16112947" /><ref name="pmid12215716" /> |
|||
<!-- History, society, and culture --> |
|||
Noretynodrel, unlike most progestins but similarly to [[etynodiol diacetate]], has some [[estrogen (medication)|estrogen]]ic activity.<ref name="RunnebaumRabe2012">{{cite book|author1=Benno Clemens Runnebaum|author2=Thomas Rabe|author3=Ludwig Kiesel|title=Female Contraception: Update and Trends|url=https://books.google.com/books?id=LtT6CAAAQBAJ&pg=PA36|date=6 December 2012|publisher=Springer Science & Business Media|isbn=978-3-642-73790-9|pages=36–}}</ref> It has little or no [[androgen]]ic activity.<ref name="Marks2010" /><ref name="RunnebaumRabe2012"/><ref name="Sloane2002">{{cite book|author=Ethel Sloane|title=Biology of Women|url=https://books.google.com/books?id=kqcYyk7zlHYC&pg=PA426|year=2002|publisher=Cengage Learning|isbn=0-7668-1142-5|pages=426–}}</ref> The drug is a relatively weak [[progestogen]], with only about one-tenth of the progestogenic activity of norethisterone, and in relation to this fact, is no longer used in oral contraceptives.<ref name="WilliamsFoye2002">{{cite book|author1=David A. Williams|author2=William O. Foye|author3=Thomas L. Lemke|title=Foye's Principles of Medicinal Chemistry|url=https://books.google.com/books?id=qLJ6Bs1Qml4C&pg=PA700|date=January 2002|publisher=Lippincott Williams & Wilkins|isbn=978-0-683-30737-5|pages=700–}}</ref> |
|||
Noretynodrel was introduced for medical use in 1957.<ref name="Marks2010">{{cite book|author=Lara Marks|title=Sexual Chemistry: A History of the Contraceptive Pill|url=https://books.google.com/books?id=_i-s4biQs7MC&pg=PA75|year=2010|publisher=Yale University Press|isbn=0-300-16791-1|pages=74–75}}</ref> It was specifically approved at this time in combination with [[mestranol]] for the treatment of [[gynecological disorder|gynecological]] and [[menstrual disorder]]s.<ref name="Marks2010" /> Subsequently, in 1960, this formulation was approved for use as a birth control pill.<ref name="Marks2010" /><ref name="Hollinger2007">{{cite book|author=Mannfred A. Hollinger|title=Introduction to Pharmacology, Third Edition|url=https://books.google.com/books?id=qfrLBQAAQBAJ&pg=PA160|date=19 October 2007|publisher=CRC Press|isbn=978-1-4200-4742-4|pages=160–}}</ref> It was the first birth control pill to be introduced, and was followed by birth control pills containing norethisterone and other progestins shortly thereafter.<ref name="Marks2010" /><ref name="Hollinger2007" /><ref name="Ravina2011" /> Due to its nature as a relatively weak progestogen, noretynodrel is no longer used in medicine.<ref name="WilliamsFoye2002" /> As such, it is no longer marketed.<ref name="Drugs.com" /><ref name="Martindale" /> |
|||
{{TOC limit|3}} |
{{TOC limit|3}} |
||
==Medical uses== |
==Medical uses== |
||
Noretynodrel was formerly used in combination with the [[estrogen (medication)|estrogen]] [[mestranol]] in the treatment of [[gynecological disorder|gynecological]] and [[menstrual disorder]]s and as a [[combined oral contraceptive]].<ref name="Marks2010" /><ref name="Hollinger2007" /> It has |
Noretynodrel was formerly used in combination with the [[estrogen (medication)|estrogen]] [[mestranol]] in the treatment of [[gynecological disorder|gynecological]] and [[menstrual disorder]]s and as a [[combined oral contraceptive|combined birth control pill]].<ref name="Marks2010" /><ref name="Hollinger2007" /> It has also been used in the treatment of [[endometriosis]] at high dosages of 40 to 100 mg/day.<ref name="ThomasRock2012" /> The medication has been discontinued, and is no longer marketed or used medically.<ref name="WilliamsFoye2002" /><ref name="IndexNominum2000" /><ref name="Drugs.com" /> |
||
==Contraindications== |
|||
Noretynodrel has been studied in the treatment of [[endometriosis]] at high dosages of 40 to 100 mg/day.<ref name="ThomasRock2012" /> |
|||
No adverse effects have been observed in [[breastfeeding]] infants whose mothers were treated with noretynodrel.<ref name="Martindale" /> Because of this, the [[American Academy of Pediatrics]] has considered noretynodrel to be usually compatible with breastfeeding.<ref name="Martindale" /> |
|||
==Side effects== |
==Side effects== |
||
{{See also|Progestin#Side effects}} |
{{See also|Progestin#Side effects}} |
||
There is a reported case of signs of [[virilization|masculinization]] in a female infant whose mother was treated with noretynodrel for [[threatened miscarriage]] during [[pregnancy]].<ref name="Martindale" /><ref name="pmid13844748">{{cite journal | vauthors = WILKINS L | title = Masculinization of female fetus due to use of orally given progestins | journal = J Am Med Assoc | volume = 172 | issue = | pages = 1028–32 | date = March 1960 | pmid = 13844748 | doi = 10.1007/978-94-011-6621-8_31 | url = }}</ref><ref name="pmid13753182" /> |
|||
==Pharmacology== |
==Pharmacology== |
||
===Pharmacodynamics=== |
===Pharmacodynamics=== |
||
Noretynodrel is very closely related to norethisterone and tibolone, which are the δ<sup>4</sup>-isomer and the 7α-[[methyl group|methyl]] derivative of noretynodrel, respectively.<ref name="pmid22210085">{{cite journal |vauthors=Jin Y, Duan L, Chen M, Penning TM, Kloosterboer HJ |title=Metabolism of the synthetic progestogen norethynodrel by human ketosteroid reductases of the aldo-keto reductase superfamily |journal=J. Steroid Biochem. Mol. Biol. |volume=129 |issue=3-5 |pages=139–44 |year=2012 |pmid=22210085 |pmc=3303946 |doi=10.1016/j.jsbmb.2011.12.002 |url=}}</ref><ref name="pmid12475720">{{cite journal |vauthors=de Gooyer ME, Deckers GH, Schoonen WG, Verheul HA, Kloosterboer HJ |title=Receptor profiling and endocrine interactions of tibolone |journal=Steroids |volume=68 |issue=1 |pages=21–30 |year=2003 |pmid=12475720 |doi= 10.1016/s0039-128x(02)00112-5|url=}}</ref> |
Noretynodrel is very closely related to norethisterone and tibolone, which are the δ<sup>4</sup>-isomer and the 7α-[[methyl group|methyl]] derivative of noretynodrel, respectively.<ref name="pmid22210085">{{cite journal |vauthors=Jin Y, Duan L, Chen M, Penning TM, Kloosterboer HJ |title=Metabolism of the synthetic progestogen norethynodrel by human ketosteroid reductases of the aldo-keto reductase superfamily |journal=J. Steroid Biochem. Mol. Biol. |volume=129 |issue=3-5 |pages=139–44 |year=2012 |pmid=22210085 |pmc=3303946 |doi=10.1016/j.jsbmb.2011.12.002 |url=}}</ref><ref name="pmid12475720">{{cite journal |vauthors=de Gooyer ME, Deckers GH, Schoonen WG, Verheul HA, Kloosterboer HJ |title=Receptor profiling and endocrine interactions of tibolone |journal=Steroids |volume=68 |issue=1 |pages=21–30 |year=2003 |pmid=12475720 |doi= 10.1016/s0039-128x(02)00112-5|url=}}</ref> It is metabolized in a very similar manner to tibolone, whereas the metabolism of norethisterone differs.<ref name="pmid22210085" /> Both noretynodrel and tibolone are transformed into 3α- and 3β-hydroxylated metabolites and a δ<sup>4</sup>-isomer metabolite (in the case of noretynodrel, this being norethisterone), whereas norethisterone is not 3α- or 3β-hydroxylated (and of course does not form a δ<sup>4</sup>-isomer metabolite).<ref name="pmid22210085" /><ref name="pmid12475720" /> The major [[metabolite]]s of noretynodrel are 3α-hydroxynoretynodrel and to a lesser extent 3β-hydroxynoretynodrel, formed respectively by [[3α-hydroxysteroid dehydrogenase|3α-]] and [[3β-hydroxysteroid dehydrogenase]]s ([[AKR1C1]]–[[AKR1C4|4]]), while the δ<sup>4</sup>-isomer norethisterone is a minor metabolite formed in small amounts.<ref name="pmid22210085" /> Tibolone is considered to be a [[prodrug]] of both its 3α- and 3β-hydroxylated and δ<sup>4</sup>-isomerized metabolites.<ref name="pmid22210085" /> Noretynodrel is also thought to be a prodrug, as it is rapidly [[metabolism|metabolized]] and cleared from circulation (within 30 minutes)<ref name="pmid2256526">{{cite journal | vauthors = Hammerstein J | title = Prodrugs: advantage or disadvantage? | journal = Am. J. Obstet. Gynecol. | volume = 163 | issue = 6 Pt 2 | pages = 2198–203 | year = 1990 | pmid = 2256526 | doi = | url = }}</ref> and shows very weak relative affinity for the [[progesterone receptor]] (PR), although noretynodrel appears to form norethisterone in only minor quantities.<ref name="pmid22210085" /><ref name="pmid12215716">{{cite journal |last1= Stanczyk |first1= Frank Z. |title= Pharmacokinetics and Potency of Progestins used for Hormone Replacement Therapy and Contraception |journal= Reviews in Endocrine and Metabolic Disorders |volume=3 |issue=3 |date= Sep 2002 |pages= 211–224 |issn= 1389-9155 |doi= 10.1023/A:1020072325818 | pmid = 12215716 | url = https://link.springer.com/content/pdf/10.1023/A:1020072325818.pdf | quote = Although there is no convincing evidence for the ''in vivo'' transformation of norethynodrel to norethindrone, data from receptor-binding tests and bioassays suggest that norethynodrel is also a prodrug.}}</ref> |
||
====Progestogenic activity==== |
====Progestogenic activity==== |
||
In terms of the PR, noretynodrel possesses only about 6 to 19% of the [[affinity (pharmacology)|affinity]] of norethisterone for the PR<sub>A</sub>, whereas the affinity of the two drugs for the PR<sub>B</sub> is similar (noretynodrel possesses 94% of the affinity of norethisterone for the PR<sub>B</sub>).<ref name="pmid12475720" /> Tibolone and the δ<sup>4</sup>-isomer metabolite of tibolone have similar affinity for the PRs as noretynodrel and norethisterone, respectively, whereas the 3α- and 3β-hydroxylated metabolites of tibolone are virtually devoid of affinity for the |
Noretynodrel is a relatively weak [[progestogen]], with only about one-tenth of the progestogenic activity of norethisterone.<ref name="WilliamsFoye2002" /> The [[ovulation]]-inhibiting dosage of noretynodrel is 4.0 mg/day, relative to 0.4 mg/day in the case of norethisterone.<ref name="pmid2170822" /> In terms of the PR, noretynodrel possesses only about 6 to 19% of the [[affinity (pharmacology)|affinity]] of norethisterone for the PR<sub>A</sub>, whereas the affinity of the two drugs for the PR<sub>B</sub> is similar (noretynodrel possesses 94% of the affinity of norethisterone for the PR<sub>B</sub>).<ref name="pmid12475720" /> Tibolone and the δ<sup>4</sup>-isomer metabolite of tibolone have similar affinity for the PRs as noretynodrel and norethisterone, respectively, whereas the 3α- and 3β-hydroxylated metabolites of tibolone are virtually devoid of affinity for the PRs.<ref name="pmid12475720" /> Since the structurally related [[anabolic–androgenic steroid]] [[trestolone]] (7α-methyl-19-nortestosterone) is known to be a potent progestogen,<ref name="BeriKumar1998">{{cite journal|last1=Beri|first1=Ripla|last2=Kumar|first2=Narender|last3=Savage|first3=T.|last4=Benalcazar|first4=L.|last5=Sundaram|first5=Kalyan |title= Estrogenic and progestational activity of 7α-methyl-19-nortestosterone, a synthetic androgen |journal=The Journal of Steroid Biochemistry and Molecular Biology |volume=67 |issue=3 |year=1998 |pages=275–283 |issn=0960-0760 |doi= 10.1016/S0960-0760(98)00114-9 }}</ref> suggesting that a 7α-methyl substitution does not interfere with progestogenic activity, 3α- and 3β-hydroxynoretynodrel likely are devoid of affinity for the PR similarly to the 3α- and 3β-hydroxylated metabolites of tibolone.<ref name="pmid12475720" /> |
||
====Androgenic activity==== |
====Androgenic activity==== |
||
Noretynodrel has been said to possess no or only very weak [[androgen]]ic activity.<ref name="Marks2010" /><ref name="RunnebaumRabe2012" /><ref name="Sloane2002" /> This is in contrast to norethisterone, which shows mild but significant androgenicity.<ref name="Marks2010" /><ref name="pmid16112947" /> Relative to norethisterone, noretynodrel has 45 to 81% lower affinity for the [[androgen receptor]] (AR).<ref name="pmid12475720" /> In accordance, no androgenic effects (such as [[hirsutism]], [[clitoromegaly|clitoral enlargement]], or [[voice change]]s) have been observed with noretynodrel even when used in large dosages (e.g., 60 mg/day) for prolonged periods of time (9–12 months) in the treatment of women with [[endometriosis]].<ref name="pmid14129897">{{cite journal | vauthors = Kistner RW | title = Steroid compounds with progestational activity | journal = Postgrad Med | volume = 35 | issue = | pages = 225–32 | year = 1964 | pmid = 14129897 | doi = 10.1080/00325481.1964.11695038| url = | quote = This difference is important clinically since no androgenic effects (hirsutism, enlarged clitoris, voice change) have been reported even with large dosages of norethynodrel (60 mg. daily) continued from 9 to 12 months in patients with endometriosis.}}</ref> Additionally, noretynodrel has not been found to virilize female [[fetus]]es, in contrast to many other [[testosterone]]-derived progestins including [[ethisterone]], norethisterone, and [[norethisterone acetate]].<ref name="Simpson1998">{{cite book |author1=Simpson, Joe Leigh |author2=Kaufman, Raymond H. |year=1998 |chapter=Fetal effects of estrogens, progestogens and diethylstilbestrol |editor=Fraser, Ian S. |title=Estrogens and Progestogens in Clinical Practice |edition=3rd |location=London |publisher=Churchill Livingstone |pages=533–53 |isbn=0-443-04706-5}}</ref> However, at least one case of [[pseudohermaphroditism]] (virilized genitalia) has been observed that may have been due to noretynodrel.<ref name="pmid13753182">{{cite journal |vauthors=Korn GW |title=The use of norethynodrel (enovid) in clinical practice |journal=Can Med Assoc J |volume=84 |issue= |pages=584–7 |year=1961 |pmid=13753182 |pmc=1939348 |doi= |url= | quote = Pseudohermaphroditism should not be a problem in these patients as it appears that norethynodrel does not possess androgenic properties, but it is believed that Wilkins has now found one such case in a patient who has been on norethynodrel therapy.}}</ref> The δ<sup>4</sup>-isomer metabolite of tibolone shows dramatically and disproportionately increased affinity for the AR relative to norethisterone and noretynodrel (5.7- to 18.5-fold greater than that of norethisterone), indicating that the 7α-methyl group of tibolone markedly increases its androgenic activity and is responsible for the greater androgenic effects of tibolone relative to noretynodrel.<ref name="pmid12475720" /> |
|||
====Estrogenic activity==== |
====Estrogenic activity==== |
||
Whereas norethisterone has virtually no affinity for the [[estrogen receptor]]s (ERs), noretynodrel shows some, albeit very weak affinity for both the [[ERα]] and the [[ERβ]] (in terms of [[relative binding affinity]], 0.7% and 0.22% of that of [[estradiol (medication)|estradiol]], respectively).<ref name="pmid12475720" /><ref name="pmid9048584">{{cite journal |vauthors = Kuiper GG, Carlsson B, Grandien K, Enmark E, Häggblad J, Nilsson S, Gustafsson JA |title= Comparison of the ligand binding specificity and transcript tissue distribution of estrogen receptors alpha and beta |journal= Endocrinology |volume= 138 |issue= 3 |pages= 863–70 |year= 1997 |pmid= 9048584 |doi= 10.1210/endo.138.3.4979 |url= }}</ref> The estrogenic activity of 3α- and 3β-hydroxynoretynodrel has never been assessed.<ref name="pmid22210085" /> However, while tibolone shows similar affinity for the ERs as noretynodrel, the 3α- and 3β-hydroxylated metabolites of tibolone have several-fold increased affinity for the ERs.<ref name="pmid22210085" /><ref name="pmid12475720" /> As such, the 3α- and 3β-hydroxylated metabolites of noretynodrel may also show increased estrogenic activity, and this may account for the known estrogenic effects of noretynodrel.<ref name="pmid22210085" /><ref name="pmid12475720" / |
Noretynodrel, unlike most progestins but similarly to [[etynodiol diacetate]], has some [[estrogen (medication)|estrogen]]ic activity.<ref name="RunnebaumRabe2012" /> Relative to other 19-nortestosterone progestins, noretynodrel is said to possess much stronger estrogenic activity.<ref name="pmid2256526" /> In the Allen–Doisy test of estrogenicity in animals, noretynodrel has been reported to possess 100-fold greater estrogenic activity than norethisterone.<ref name="pmid16112947" /> Whereas norethisterone has virtually no affinity for the [[estrogen receptor]]s (ERs), noretynodrel shows some, albeit very weak affinity for both the [[ERα]] and the [[ERβ]] (in terms of [[relative binding affinity]], 0.7% and 0.22% of that of [[estradiol (medication)|estradiol]], respectively).<ref name="pmid12475720" /><ref name="pmid9048584">{{cite journal |vauthors = Kuiper GG, Carlsson B, Grandien K, Enmark E, Häggblad J, Nilsson S, Gustafsson JA |title= Comparison of the ligand binding specificity and transcript tissue distribution of estrogen receptors alpha and beta |journal= Endocrinology |volume= 138 |issue= 3 |pages= 863–70 |year= 1997 |pmid= 9048584 |doi= 10.1210/endo.138.3.4979 |url= }}</ref> The estrogenic activity of 3α- and 3β-hydroxynoretynodrel has never been assessed.<ref name="pmid22210085" /> However, while tibolone shows similar affinity for the ERs as noretynodrel, the 3α- and 3β-hydroxylated metabolites of tibolone have several-fold increased affinity for the ERs.<ref name="pmid22210085" /><ref name="pmid12475720" /> As such, the 3α- and 3β-hydroxylated metabolites of noretynodrel may also show increased estrogenic activity, and this may account for the known estrogenic effects of noretynodrel.<ref name="pmid22210085" /><ref name="pmid12475720" /> |
||
The δ<sup>4</sup>-isomer of tibolone, similarly to norethisterone, is virtually devoid of affinity for the ERs.<ref name="pmid12475720" /> Neither tibolone nor its metabolites are aromatized, whereas trestolone is readily aromatized similarly to testosterone and 19-nortestosterone, and for these reasons, it is unlikely that noretynodrel or its metabolites, aside from norethisterone, are aromatized either.<ref name="de GooyerOppers-Tiemissen2003">{{cite journal |last1=de Gooyer|first1=Marcel E. |last2= Oppers-Tiemissen|first2=Hendrika M. |last3= Leysen |first3=Dirk |last4=Verheul |first4=Herman A.M. |last5= Kloosterboer |first5= Helenius J. |title= Tibolone is not converted by human aromatase to 7α-methyl-17α-ethynylestradiol (7α-MEE): |journal= Steroids |volume=68 |issue=3 |year=2003 |pages= 235–243 |issn= 0039-128X |doi= 10.1016/S0039-128X(02)00184-8 }}</ref> As such, aromatization likely does not play a role in the estrogenic activity of tibolone or noretynodrel.<ref name="de GooyerOppers-Tiemissen2003" /> However, controversy on this matter exists, and other researchers have suggested that tibolone and noretynodrel may be aromatized in small amounts to highly potent estrogens ([[ethinylestradiol]] and its 7α-methyl derivative, respectively).<ref name="KuhlWiegratz2009">{{cite journal |last1=Kuhl |first1=H. |last2= Wiegratz |first2=I. |title= Can 19-nortestosterone derivatives be aromatized in the liver of adult humans? Are there clinical implications?|journal= Climacteric |volume=10 |issue=4 |year=2009 |pages=344–353 |issn= 1369-7137 |doi= 10.1080/13697130701380434 }}</ref><ref name="Kloosterboer2009">{{cite journal |title= Tibolone is not aromatized in postmenopausal women |journal= Climacteric |volume=11 |issue=2 |year=2009 |pages= 175–176 |issn= 1369-7137 |doi= 10.1080/13697130701752087}}</ref> |
|||
Relative to other 19-nortestosterone progestins, noretynodrel is said to possess much stronger estrogenic activity.<ref name="pmid2256526" /> |
|||
===Pharmacokinetics=== |
|||
Noretynodrel is rapidly [[absorption (pharmacokinetics)|absorbed]] upon [[oral administration]] and is rapidly [[metabolism|metabolized]], disappearing from the circulation within 30 minutes.<ref name="Seyffart2012">{{cite book|author=G. Seyffart|title=Drug Dosage in Renal Insufficiency|url=https://books.google.com/books?id=OavnCAAAQBAJ&pg=PA423|date=6 December 2012|publisher=Springer Science & Business Media|isbn=978-94-011-3804-8|pages=423–}}</ref><ref name="pmid2256526" /> In terms of [[plasma protein binding]], noretynodrel is bound to [[human serum albumin|albumin]], and show no [[affinity (pharmacology)|affinity]] itself for [[sex hormone-binding globulin]] or [[corticosteroid-binding globulin]].<ref name="pmid2170822" /> The plasma protein binding of its [[metabolite]]s, such as [[norethisterone]], may differ however.<ref name="pmid16112947" /> |
|||
The major metabolites of noretynodrel in the circulation are 3α-hydroxynoretynodrel (formed by {{abbrlink|3α-HSD|3α-hydroxysteroid dehydrogenase}}) and to a lesser extent 3β-hydroxynoretynodrel (formed by {{abbrlink|3β-HSD|3β-hydroxysteroid dehydrogenase}}), and more minor metabolites of noretynodrel are [[norethisterone]] (formed by [[3β-hydroxysteroid dehydrogenase|δ<sup>5-4</sup>-isomerase]]) and possibly [[ethinylestradiol]] (formed by [[aromatase]] or possibly other [[cytochrome P450]] [[enzyme]]s, most likely [[monooxygenase]]s).<ref name="pmid22210085" /><ref name="Seyffart2012" /><ref name="pmid16112947" /><ref name="Kuhl2011">{{cite journal | author = Kuhl H | title = Pharmacology of Progestogens | journal = J Reproduktionsmed Endokrinol | year = 2011 | volume = 8 | issue = 1 | pages = 157–177 | format = PDF | url = http://www.kup.at/kup/pdf/10168.pdf}}</ref> Due to its very short [[elimination half-life]] and its low [[affinity (pharmacology)|affinities]] for [[steroid hormone receptor]]s in [[ligand binding assay|receptor binding assay]]s, noretynodrel is considered to be a [[prodrug]] which is rapidly [[biotransformation|transformed]] into its [[active metabolite]]s in the [[intestine]]s and [[liver]] following oral administration.<ref name="pmid2170822">{{cite journal | vauthors = Kuhl H | title = Pharmacokinetics of oestrogens and progestogens | journal = Maturitas | volume = 12 | issue = 3 | pages = 171–97 | date = September 1990 | pmid = 2170822 | doi = | url = }}</ref><ref name="pmid16112947">{{cite journal | vauthors = Kuhl H | title = Pharmacology of estrogens and progestogens: influence of different routes of administration | journal = Climacteric | volume = 8 Suppl 1 | issue = | pages = 3–63 | year = 2005 | pmid = 16112947 | doi = 10.1080/13697130500148875 | url = http://hormonebalance.org/images/documents/Kuhl%2005%20%20Pharm%20Estro%20Progest%20Climacteric_1313155660.pdf}}</ref><ref name="pmid12215716" /><ref name="pmid2256526" /> Some researchers have stated that it is specifically a prodrug of norethisterone.<ref name="pmid2170822" /><ref name="pmid16112947" /><ref name="pmid12215716" /> According to other researchers however, there is, due to a lack of research, insufficient data to unequivocally show this to be the case at present.<ref name="pmid12215716" /> |
|||
About 1% of an oral dose of noretynodrel is detected in [[breast milk]].<ref name="Martindale" /> |
|||
==Chemistry== |
==Chemistry== |
||
{{See also|List of progestogens}} |
{{See also|List of progestogens}} |
||
Noretynodrel, also known as 17α-ethynyl-δ<sup>5(10)</sup>-19-nortestosterone or as 17α-ethynylestr-5(10)-en-17β-ol-3-one, is a [[synthetic compound|synthetic]] [[estrane]] [[steroid]] and a [[chemical derivative|derivative]] of [[testosterone (medication)|testosterone]].<ref name="Elks2014" /><ref name="IndexNominum2000" /> It is specifically a derivative of testosterone that has been [[ethynyl group|ethynyl]]ated at the C17α position, [[desmethyl|demethyl]]ated at the C19 position, and [[dehydrogenation|dehydrogenated]] (i.e., has a [[double bond]]) between the C5 and C10 positions).<ref name="Elks2014" /><ref name="IndexNominum2000" /> As such, noretynodrel is also a combined derivative of [[nandrolone]] (19-nortestosterone) and [[ethisterone]] (17α-ethynyltestosterone).<ref name="Elks2014" /><ref name="IndexNominum2000" /> In addition, it is an [[isomer]] of norethisterone (17α-ethynyl-19-nortestosterone) in which the C4 double bond has been replaced with a double bond between the C5 and C10 positions.<ref name="Elks2014" /><ref name="IndexNominum2000" /> For this reason, noretynodrel is also known as 5(10)-norethisterone.<ref name="Elks2014" /><ref name="IndexNominum2000" /> Few other 19-nortestosterone progestins share the C5(10) double bond of noretynodrel, but |
Noretynodrel, also known as 17α-ethynyl-δ<sup>5(10)</sup>-19-nortestosterone or as 17α-ethynylestr-5(10)-en-17β-ol-3-one, is a [[synthetic compound|synthetic]] [[estrane]] [[steroid]] and a [[chemical derivative|derivative]] of [[testosterone (medication)|testosterone]].<ref name="Elks2014" /><ref name="IndexNominum2000" /> It is specifically a derivative of testosterone that has been [[ethynyl group|ethynyl]]ated at the C17α position, [[desmethyl|demethyl]]ated at the C19 position, and [[dehydrogenation|dehydrogenated]] (i.e., has a [[double bond]]) between the C5 and C10 positions).<ref name="Elks2014" /><ref name="IndexNominum2000" /> As such, noretynodrel is also a combined derivative of [[nandrolone]] (19-nortestosterone) and [[ethisterone]] (17α-ethynyltestosterone).<ref name="Elks2014" /><ref name="IndexNominum2000" /> In addition, it is an [[isomer]] of norethisterone (17α-ethynyl-19-nortestosterone) in which the C4 double bond has been replaced with a double bond between the C5 and C10 positions.<ref name="Elks2014" /><ref name="IndexNominum2000" /> For this reason, noretynodrel is also known as 5(10)-norethisterone.<ref name="Elks2014" /><ref name="IndexNominum2000" /> Few other 19-nortestosterone progestins share the C5(10) double bond of noretynodrel, but examples of a couple that do include [[tibolone]], the C7α [[methyl group|methyl]] derivative of noretynodrel (i.e., 7α-methylnoretynodrel), and [[norgesterone]], the C17α [[vinyl group|vinyl]] [[structural analog|analogue]] of noretynodrel.<ref name="Elks2014" /><ref name="IndexNominum2000" /> |
||
==History== |
==History== |
||
Noretynodrel was first synthesized by [[Frank B. Colton]] of [[G.D. Searle, LLC|G. D. Searle & Company]] in 1952, and this was preceded by the synthesis of norethisterone by [[Luis E. Miramontes]] and [[Carl Djerassi]] of [[Syntex]] in 1951.<ref name="Marks2010" /> In 1957, both noretynodrel and norethisterone were approved in the [[United States]] for the treatment of [[menstrual disorder]]s.<ref name="Ravina2011">{{cite book |author= Enrique Ravina |title= The Evolution of Drug Discovery: From Traditional Medicines to Modern Drugs |url= https://books.google.com/books?id=iDNy0XxGqT8C&pg=PA190 |date= 11 January 2011 |publisher= John Wiley & Sons |isbn= 978-3-527-32669-3 |pages= 190–}}</ref> In 1960, noretynodrel, in combination with |
Noretynodrel was first synthesized by [[Frank B. Colton]] of [[G.D. Searle, LLC|G. D. Searle & Company]] in 1952, and this was preceded by the synthesis of norethisterone by [[Luis E. Miramontes]] and [[Carl Djerassi]] of [[Syntex]] in 1951.<ref name="Marks2010" /> In 1957, both noretynodrel and norethisterone, in combination with [[mestranol]], were approved in the [[United States]] for the treatment of [[menstrual disorder]]s.<ref name="Ravina2011">{{cite book |author= Enrique Ravina |title= The Evolution of Drug Discovery: From Traditional Medicines to Modern Drugs |url= https://books.google.com/books?id=iDNy0XxGqT8C&pg=PA190 |date= 11 January 2011 |publisher= John Wiley & Sons |isbn= 978-3-527-32669-3 |pages= 190–}}</ref> In 1960, noretynodrel, in combination with mestranol (as ''Enovid''), was introduced in the United States as the first oral contraceptive, and the combination of norethisterone and mestranol followed in 1963 as the second oral contraceptive to be introduced.<ref name="Ravina2011" /> In 1988, Enovid, along with other oral contraceptives containing high doses of estrogen, was discontinued.<ref>{{cite news |author= Reuters News Service |title=Searle, 2 others to stop making high-estrogen pill |work= St. Louis Post-Dispatch |pages=7D |date= 1988-04-15 |url= http://nl.newsbank.com/nl-search/we/Archives?p_text_direct-0=0EB327685B9D39FF&p_field_direct-0=document_id |accessdate=2009-08-29}}</ref><ref>{{cite news |title=High-estrogen 'pill' going off market |work= San Jose Mercury News |date= 1988-04-15 |url= http://nl.newsbank.com/nl-search/we/Archives?p_text_direct-0=0EB72D898C02F468&p_field_direct-0=document_id |accessdate= 2009-08-29}}</ref> |
||
Noretynodrel was first studied in the treatment of [[endometriosis]] in 1961 and was the first progestin to be investigated for the treatment of the condition.<ref name="ThomasRock2012">{{cite book|author1=Eric J. Thomas|author2=J. Rock|title=Modern Approaches to Endometriosis|url=https://books.google.com/books?id=AFP1CAAAQBAJ&pg=PA223|date=6 December 2012|publisher=Springer Science & Business Media|isbn=978-94-011-3864-2|pages=223–}}</ref> |
Noretynodrel was first studied in the treatment of [[endometriosis]] in 1961 and was the first progestin to be investigated for the treatment of the condition.<ref name="ThomasRock2012">{{cite book|author1=Eric J. Thomas|author2=J. Rock|title=Modern Approaches to Endometriosis|url=https://books.google.com/books?id=AFP1CAAAQBAJ&pg=PA223|date=6 December 2012|publisher=Springer Science & Business Media|isbn=978-94-011-3864-2|pages=223–}}</ref> |
||
| Line 97: | Line 115: | ||
===Generic names=== |
===Generic names=== |
||
''Noretynodrel'' is the {{abbrlink|INN|International Nonproprietary Name}} of the drug while ''norethynodrel'' is its {{abbrlink|USAN|United States Adopted Name}} and {{abbrlink|BAN|British Approved Name}}.<ref name="Elks2014" /><ref name="IndexNominum2000" /><ref name="Drugs.com" /> |
''Noretynodrel'' is the {{abbrlink|INN|International Nonproprietary Name}} of the drug while ''norethynodrel'' is its {{abbrlink|USAN|United States Adopted Name}} and {{abbrlink|BAN|British Approved Name}}.<ref name="Elks2014">{{cite book|author=J. Elks|title=The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies|url=https://books.google.com/books?id=0vXTBwAAQBAJ&pg=PA886|date=14 November 2014|publisher=Springer|isbn=978-1-4757-2085-3|pages=886–}}</ref><ref name="IndexNominum2000">{{cite book|title=Index Nominum 2000: International Drug Directory|url=https://books.google.com/books?id=5GpcTQD_L2oC&pg=PP1|date=January 2000|publisher=Taylor & Francis|isbn=978-3-88763-075-1|pages=1–}}</ref><ref name="Martindale" /><ref name="Drugs.com" /> It is also known by its developmental code names ''SC-4642'' and ''NSC-15432''.<ref name="Elks2014" /><ref name="IndexNominum2000" /><ref name="Martindale" /><ref name="Drugs.com" /> |
||
===Brand names=== |
===Brand names=== |
||
| Line 113: | Line 131: | ||
{{Navboxes |
|||
| title = [[Medicine|Medical uses]] |
|||
| titlestyle = background:#ccccff |
|||
| list1 = |
|||
{{Progestogens and antiprogestogens}} |
{{Progestogens and antiprogestogens}} |
||
{{Androgens and antiandrogens}} |
|||
{{Estrogens and antiestrogens}} |
{{Estrogens and antiestrogens}} |
||
}} |
|||
{{Navboxes |
|||
| title = [[Pharmacodynamics]] |
|||
| titlestyle = background:#ccccff |
|||
| list1 = |
|||
{{Androgen receptor modulators}} |
|||
{{Progesterone receptor modulators}} |
{{Progesterone receptor modulators}} |
||
{{Estrogen receptor modulators}} |
{{Estrogen receptor modulators}} |
||
}} |
|||
[[Category:Abandoned drugs]] |
[[Category:Abandoned drugs]] |
||
[[Category:Alcohols]] |
[[Category:Alcohols]] |
||
[[Category:Alkynes]] |
[[Category:Alkynes]] |
||
[[Category:Androgens and anabolic steroids]] |
|||
[[Category:Estranes]] |
[[Category:Estranes]] |
||
[[Category:Hormonal contraception]] |
[[Category:Hormonal contraception]] |
||
Revision as of 23:54, 23 March 2018
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Enovid (with mestranol), others |
| Other names | Norethynodrel; Noretinodrel Norethinodrel; SC-4642; NSC-15432; 5(10)-Norethisterone; 17α-Ethinyl-19-nor-5(10)-testosterone; 17α-Ethynyl-δ5(10)-19-nortestosterone; 17α-Ethynylestr-5(10)-en-17β-ol-3-one; 19-Nor-17α-pregn-5(10)-en-20-yn-17β-ol-3-one |
| Routes of administration | By mouth |
| Drug class | Progestin; Progestogen; Estrogen |
| ATC code | |
| Pharmacokinetic data | |
| Protein binding | Noretynodrel: to albumin and not to SHBG or CBG[1] |
| Metabolism | Liver, intestines (hydroxylation, isomerization, conjugation)[1][3] |
| Metabolites | • 3α-Hydroxynoretynodrel[2] • 3β-Hydroxynoretynodrel[2] • Norethisterone[2][1][3] • Ethinylestradiol[3][4]• Conjugates[3] |
| Elimination half-life | Very short (< 30 minutes)[5] |
| Excretion | Breast milk: 1%[6] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.000.620 |
| Chemical and physical data | |
| Formula | C20H26O2 |
| Molar mass | 298.419 g/mol g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Noretynodrel, or norethynodrel, sold under the brand name Enovid among others, is a progestin medication which was previously used in birth control pills and in the treatment of gynecological disorders but is now no longer marketed.[6][7][8] It was available both alone and in combination with an estrogen.[9][7][8] The medication is taken by mouth.[7]
Noretynodrel is a progestin, or a synthetic progestogen, and hence is an agonist of the progesterone receptor, the biological target of progestogens like progesterone.[3] It is a relatively weak progestogen.[10] The medication has weak estrogenic activity, no or only very weak androgenic activity, and no other important hormonal activity.[3][8][11][12] It is a prodrug of various active metabolites in the body, such as norethisterone among others.[3][13]
Noretynodrel was introduced for medical use in 1957.[8] It was specifically approved at this time in combination with mestranol for the treatment of gynecological and menstrual disorders.[8] Subsequently, in 1960, this formulation was approved for use as a birth control pill.[8][14] It was the first birth control pill to be introduced, and was followed by birth control pills containing norethisterone and other progestins shortly thereafter.[8][14][15] Due to its nature as a relatively weak progestogen, noretynodrel is no longer used in medicine.[10] As such, it is no longer marketed.[16][6]
Medical uses
Noretynodrel was formerly used in combination with the estrogen mestranol in the treatment of gynecological and menstrual disorders and as a combined birth control pill.[8][14] It has also been used in the treatment of endometriosis at high dosages of 40 to 100 mg/day.[17] The medication has been discontinued, and is no longer marketed or used medically.[10][18][16]
Contraindications
No adverse effects have been observed in breastfeeding infants whose mothers were treated with noretynodrel.[6] Because of this, the American Academy of Pediatrics has considered noretynodrel to be usually compatible with breastfeeding.[6]
Side effects
There is a reported case of signs of masculinization in a female infant whose mother was treated with noretynodrel for threatened miscarriage during pregnancy.[6][19][20]
Pharmacology
Pharmacodynamics
Noretynodrel is very closely related to norethisterone and tibolone, which are the δ4-isomer and the 7α-methyl derivative of noretynodrel, respectively.[2][21] It is metabolized in a very similar manner to tibolone, whereas the metabolism of norethisterone differs.[2] Both noretynodrel and tibolone are transformed into 3α- and 3β-hydroxylated metabolites and a δ4-isomer metabolite (in the case of noretynodrel, this being norethisterone), whereas norethisterone is not 3α- or 3β-hydroxylated (and of course does not form a δ4-isomer metabolite).[2][21] The major metabolites of noretynodrel are 3α-hydroxynoretynodrel and to a lesser extent 3β-hydroxynoretynodrel, formed respectively by 3α- and 3β-hydroxysteroid dehydrogenases (AKR1C1–4), while the δ4-isomer norethisterone is a minor metabolite formed in small amounts.[2] Tibolone is considered to be a prodrug of both its 3α- and 3β-hydroxylated and δ4-isomerized metabolites.[2] Noretynodrel is also thought to be a prodrug, as it is rapidly metabolized and cleared from circulation (within 30 minutes)[5] and shows very weak relative affinity for the progesterone receptor (PR), although noretynodrel appears to form norethisterone in only minor quantities.[2][13]
Progestogenic activity
Noretynodrel is a relatively weak progestogen, with only about one-tenth of the progestogenic activity of norethisterone.[10] The ovulation-inhibiting dosage of noretynodrel is 4.0 mg/day, relative to 0.4 mg/day in the case of norethisterone.[1] In terms of the PR, noretynodrel possesses only about 6 to 19% of the affinity of norethisterone for the PRA, whereas the affinity of the two drugs for the PRB is similar (noretynodrel possesses 94% of the affinity of norethisterone for the PRB).[21] Tibolone and the δ4-isomer metabolite of tibolone have similar affinity for the PRs as noretynodrel and norethisterone, respectively, whereas the 3α- and 3β-hydroxylated metabolites of tibolone are virtually devoid of affinity for the PRs.[21] Since the structurally related anabolic–androgenic steroid trestolone (7α-methyl-19-nortestosterone) is known to be a potent progestogen,[22] suggesting that a 7α-methyl substitution does not interfere with progestogenic activity, 3α- and 3β-hydroxynoretynodrel likely are devoid of affinity for the PR similarly to the 3α- and 3β-hydroxylated metabolites of tibolone.[21]
Androgenic activity
Noretynodrel has been said to possess no or only very weak androgenic activity.[8][11][12] This is in contrast to norethisterone, which shows mild but significant androgenicity.[8][3] Relative to norethisterone, noretynodrel has 45 to 81% lower affinity for the androgen receptor (AR).[21] In accordance, no androgenic effects (such as hirsutism, clitoral enlargement, or voice changes) have been observed with noretynodrel even when used in large dosages (e.g., 60 mg/day) for prolonged periods of time (9–12 months) in the treatment of women with endometriosis.[23] Additionally, noretynodrel has not been found to virilize female fetuses, in contrast to many other testosterone-derived progestins including ethisterone, norethisterone, and norethisterone acetate.[24] However, at least one case of pseudohermaphroditism (virilized genitalia) has been observed that may have been due to noretynodrel.[20] The δ4-isomer metabolite of tibolone shows dramatically and disproportionately increased affinity for the AR relative to norethisterone and noretynodrel (5.7- to 18.5-fold greater than that of norethisterone), indicating that the 7α-methyl group of tibolone markedly increases its androgenic activity and is responsible for the greater androgenic effects of tibolone relative to noretynodrel.[21]
Estrogenic activity
Noretynodrel, unlike most progestins but similarly to etynodiol diacetate, has some estrogenic activity.[11] Relative to other 19-nortestosterone progestins, noretynodrel is said to possess much stronger estrogenic activity.[5] In the Allen–Doisy test of estrogenicity in animals, noretynodrel has been reported to possess 100-fold greater estrogenic activity than norethisterone.[3] Whereas norethisterone has virtually no affinity for the estrogen receptors (ERs), noretynodrel shows some, albeit very weak affinity for both the ERα and the ERβ (in terms of relative binding affinity, 0.7% and 0.22% of that of estradiol, respectively).[21][25] The estrogenic activity of 3α- and 3β-hydroxynoretynodrel has never been assessed.[2] However, while tibolone shows similar affinity for the ERs as noretynodrel, the 3α- and 3β-hydroxylated metabolites of tibolone have several-fold increased affinity for the ERs.[2][21] As such, the 3α- and 3β-hydroxylated metabolites of noretynodrel may also show increased estrogenic activity, and this may account for the known estrogenic effects of noretynodrel.[2][21]
The δ4-isomer of tibolone, similarly to norethisterone, is virtually devoid of affinity for the ERs.[21] Neither tibolone nor its metabolites are aromatized, whereas trestolone is readily aromatized similarly to testosterone and 19-nortestosterone, and for these reasons, it is unlikely that noretynodrel or its metabolites, aside from norethisterone, are aromatized either.[26] As such, aromatization likely does not play a role in the estrogenic activity of tibolone or noretynodrel.[26] However, controversy on this matter exists, and other researchers have suggested that tibolone and noretynodrel may be aromatized in small amounts to highly potent estrogens (ethinylestradiol and its 7α-methyl derivative, respectively).[27][28]
Pharmacokinetics
Noretynodrel is rapidly absorbed upon oral administration and is rapidly metabolized, disappearing from the circulation within 30 minutes.[29][5] In terms of plasma protein binding, noretynodrel is bound to albumin, and show no affinity itself for sex hormone-binding globulin or corticosteroid-binding globulin.[1] The plasma protein binding of its metabolites, such as norethisterone, may differ however.[3]
The major metabolites of noretynodrel in the circulation are 3α-hydroxynoretynodrel (formed by 3α-HSD) and to a lesser extent 3β-hydroxynoretynodrel (formed by 3β-HSD), and more minor metabolites of noretynodrel are norethisterone (formed by δ5-4-isomerase) and possibly ethinylestradiol (formed by aromatase or possibly other cytochrome P450 enzymes, most likely monooxygenases).[2][29][3][4] Due to its very short elimination half-life and its low affinities for steroid hormone receptors in receptor binding assays, noretynodrel is considered to be a prodrug which is rapidly transformed into its active metabolites in the intestines and liver following oral administration.[1][3][13][5] Some researchers have stated that it is specifically a prodrug of norethisterone.[1][3][13] According to other researchers however, there is, due to a lack of research, insufficient data to unequivocally show this to be the case at present.[13]
About 1% of an oral dose of noretynodrel is detected in breast milk.[6]
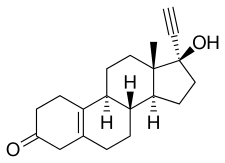
Chemistry
Noretynodrel, also known as 17α-ethynyl-δ5(10)-19-nortestosterone or as 17α-ethynylestr-5(10)-en-17β-ol-3-one, is a synthetic estrane steroid and a derivative of testosterone.[30][18] It is specifically a derivative of testosterone that has been ethynylated at the C17α position, demethylated at the C19 position, and dehydrogenated (i.e., has a double bond) between the C5 and C10 positions).[30][18] As such, noretynodrel is also a combined derivative of nandrolone (19-nortestosterone) and ethisterone (17α-ethynyltestosterone).[30][18] In addition, it is an isomer of norethisterone (17α-ethynyl-19-nortestosterone) in which the C4 double bond has been replaced with a double bond between the C5 and C10 positions.[30][18] For this reason, noretynodrel is also known as 5(10)-norethisterone.[30][18] Few other 19-nortestosterone progestins share the C5(10) double bond of noretynodrel, but examples of a couple that do include tibolone, the C7α methyl derivative of noretynodrel (i.e., 7α-methylnoretynodrel), and norgesterone, the C17α vinyl analogue of noretynodrel.[30][18]
History
Noretynodrel was first synthesized by Frank B. Colton of G. D. Searle & Company in 1952, and this was preceded by the synthesis of norethisterone by Luis E. Miramontes and Carl Djerassi of Syntex in 1951.[8] In 1957, both noretynodrel and norethisterone, in combination with mestranol, were approved in the United States for the treatment of menstrual disorders.[15] In 1960, noretynodrel, in combination with mestranol (as Enovid), was introduced in the United States as the first oral contraceptive, and the combination of norethisterone and mestranol followed in 1963 as the second oral contraceptive to be introduced.[15] In 1988, Enovid, along with other oral contraceptives containing high doses of estrogen, was discontinued.[31][32]
Noretynodrel was first studied in the treatment of endometriosis in 1961 and was the first progestin to be investigated for the treatment of the condition.[17]
Society and culture
Generic names
Noretynodrel is the INN of the drug while norethynodrel is its USAN and BAN.[30][18][6][16] It is also known by its developmental code names SC-4642 and NSC-15432.[30][18][6][16]
Brand names
Noretynodrel has been marketed by alone under the brand names Enidrel, Orgametril, and Previson and in combination with mestranol under the brand names Conovid, Conovid E, Enavid, Enavid E, Enovid, Enovid E, Norolen, and Singestol.[9]
Availability
Noretynodrel is no longer available in any formulation in the U.S.,[33] nor does it appear to still be marketed in any other country.[16][18]
See also
References
- ^ a b c d e f g Kuhl H (September 1990). "Pharmacokinetics of oestrogens and progestogens". Maturitas. 12 (3): 171–97. PMID 2170822.
- ^ a b c d e f g h i j k l m Jin Y, Duan L, Chen M, Penning TM, Kloosterboer HJ (2012). "Metabolism of the synthetic progestogen norethynodrel by human ketosteroid reductases of the aldo-keto reductase superfamily". J. Steroid Biochem. Mol. Biol. 129 (3–5): 139–44. doi:10.1016/j.jsbmb.2011.12.002. PMC 3303946. PMID 22210085.
- ^ a b c d e f g h i j k l m Kuhl H (2005). "Pharmacology of estrogens and progestogens: influence of different routes of administration" (PDF). Climacteric. 8 Suppl 1: 3–63. doi:10.1080/13697130500148875. PMID 16112947.
- ^ a b Kuhl H (2011). "Pharmacology of Progestogens" (PDF). J Reproduktionsmed Endokrinol. 8 (1): 157–177.
- ^ a b c d e Hammerstein J (1990). "Prodrugs: advantage or disadvantage?". Am. J. Obstet. Gynecol. 163 (6 Pt 2): 2198–203. PMID 2256526.
- ^ a b c d e f g h i Sweetman, Sean C., ed. (2009). "Sex hormones and their modulators". Martindale: The Complete Drug Reference (36th ed.). London: Pharmaceutical Press. pp. 2120–2121. ISBN 978-0-85369-840-1.
- ^ a b c Jucker (21 December 2013). Progress in Drug Research / Fortschritte der Arzneimittelforschung / Progrès des recherches pharmaceutiques. Birkhäuser. pp. 85–88. ISBN 978-3-0348-7065-8.
- ^ a b c d e f g h i j k Lara Marks (2010). Sexual Chemistry: A History of the Contraceptive Pill. Yale University Press. pp. 74–75. ISBN 0-300-16791-1.
- ^ a b IARC Working Group on the Evaluation of the Carcinogenic Risk of Chemicals to Man (1974). IARC Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Man: Sex Hormones (PDF). World Health Organization. p. 88,191.
- ^ a b c d David A. Williams; William O. Foye; Thomas L. Lemke (January 2002). Foye's Principles of Medicinal Chemistry. Lippincott Williams & Wilkins. pp. 700–. ISBN 978-0-683-30737-5.
- ^ a b c Benno Clemens Runnebaum; Thomas Rabe; Ludwig Kiesel (6 December 2012). Female Contraception: Update and Trends. Springer Science & Business Media. pp. 36–. ISBN 978-3-642-73790-9.
- ^ a b Ethel Sloane (2002). Biology of Women. Cengage Learning. pp. 426–. ISBN 0-7668-1142-5.
- ^ a b c d e Stanczyk, Frank Z. (Sep 2002). "Pharmacokinetics and Potency of Progestins used for Hormone Replacement Therapy and Contraception" (PDF). Reviews in Endocrine and Metabolic Disorders. 3 (3): 211–224. doi:10.1023/A:1020072325818. ISSN 1389-9155. PMID 12215716.
Although there is no convincing evidence for the in vivo transformation of norethynodrel to norethindrone, data from receptor-binding tests and bioassays suggest that norethynodrel is also a prodrug.
- ^ a b c Mannfred A. Hollinger (19 October 2007). Introduction to Pharmacology, Third Edition. CRC Press. pp. 160–. ISBN 978-1-4200-4742-4.
- ^ a b c Enrique Ravina (11 January 2011). The Evolution of Drug Discovery: From Traditional Medicines to Modern Drugs. John Wiley & Sons. pp. 190–. ISBN 978-3-527-32669-3.
- ^ a b c d e https://www.drugs.com/international/noretynodrel.html
- ^ a b Eric J. Thomas; J. Rock (6 December 2012). Modern Approaches to Endometriosis. Springer Science & Business Media. pp. 223–. ISBN 978-94-011-3864-2.
- ^ a b c d e f g h i j Index Nominum 2000: International Drug Directory. Taylor & Francis. January 2000. pp. 1–. ISBN 978-3-88763-075-1.
- ^ WILKINS L (March 1960). "Masculinization of female fetus due to use of orally given progestins". J Am Med Assoc. 172: 1028–32. doi:10.1007/978-94-011-6621-8_31. PMID 13844748.
- ^ a b Korn GW (1961). "The use of norethynodrel (enovid) in clinical practice". Can Med Assoc J. 84: 584–7. PMC 1939348. PMID 13753182.
Pseudohermaphroditism should not be a problem in these patients as it appears that norethynodrel does not possess androgenic properties, but it is believed that Wilkins has now found one such case in a patient who has been on norethynodrel therapy.
- ^ a b c d e f g h i j k de Gooyer ME, Deckers GH, Schoonen WG, Verheul HA, Kloosterboer HJ (2003). "Receptor profiling and endocrine interactions of tibolone". Steroids. 68 (1): 21–30. doi:10.1016/s0039-128x(02)00112-5. PMID 12475720.
- ^ Beri, Ripla; Kumar, Narender; Savage, T.; Benalcazar, L.; Sundaram, Kalyan (1998). "Estrogenic and progestational activity of 7α-methyl-19-nortestosterone, a synthetic androgen". The Journal of Steroid Biochemistry and Molecular Biology. 67 (3): 275–283. doi:10.1016/S0960-0760(98)00114-9. ISSN 0960-0760.
- ^ Kistner RW (1964). "Steroid compounds with progestational activity". Postgrad Med. 35: 225–32. doi:10.1080/00325481.1964.11695038. PMID 14129897.
This difference is important clinically since no androgenic effects (hirsutism, enlarged clitoris, voice change) have been reported even with large dosages of norethynodrel (60 mg. daily) continued from 9 to 12 months in patients with endometriosis.
- ^ Simpson, Joe Leigh; Kaufman, Raymond H. (1998). "Fetal effects of estrogens, progestogens and diethylstilbestrol". In Fraser, Ian S. (ed.). Estrogens and Progestogens in Clinical Practice (3rd ed.). London: Churchill Livingstone. pp. 533–53. ISBN 0-443-04706-5.
- ^ Kuiper GG, Carlsson B, Grandien K, Enmark E, Häggblad J, Nilsson S, Gustafsson JA (1997). "Comparison of the ligand binding specificity and transcript tissue distribution of estrogen receptors alpha and beta". Endocrinology. 138 (3): 863–70. doi:10.1210/endo.138.3.4979. PMID 9048584.
- ^ a b de Gooyer, Marcel E.; Oppers-Tiemissen, Hendrika M.; Leysen, Dirk; Verheul, Herman A.M.; Kloosterboer, Helenius J. (2003). "Tibolone is not converted by human aromatase to 7α-methyl-17α-ethynylestradiol (7α-MEE):". Steroids. 68 (3): 235–243. doi:10.1016/S0039-128X(02)00184-8. ISSN 0039-128X.
- ^ Kuhl, H.; Wiegratz, I. (2009). "Can 19-nortestosterone derivatives be aromatized in the liver of adult humans? Are there clinical implications?". Climacteric. 10 (4): 344–353. doi:10.1080/13697130701380434. ISSN 1369-7137.
- ^ "Tibolone is not aromatized in postmenopausal women". Climacteric. 11 (2): 175–176. 2009. doi:10.1080/13697130701752087. ISSN 1369-7137.
- ^ a b G. Seyffart (6 December 2012). Drug Dosage in Renal Insufficiency. Springer Science & Business Media. pp. 423–. ISBN 978-94-011-3804-8.
- ^ a b c d e f g h J. Elks (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 886–. ISBN 978-1-4757-2085-3.
- ^ Reuters News Service (1988-04-15). "Searle, 2 others to stop making high-estrogen pill". St. Louis Post-Dispatch. pp. 7D. Retrieved 2009-08-29.
{{cite news}}:|author=has generic name (help) - ^ "High-estrogen 'pill' going off market". San Jose Mercury News. 1988-04-15. Retrieved 2009-08-29.
- ^ "Drugs@FDA: FDA Approved Drug Products". United States Food and Drug Administration. Retrieved 27 November 2016.
