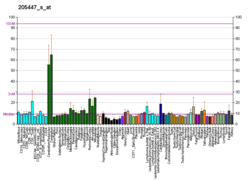
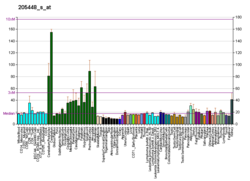
MAP3K12
Mitogen-activated protein kinase 12 is an enzyme that in humans is encoded by the MAP3K12 gene.[5][6]
Function
[edit]The protein encoded by this gene is a member of serine/threonine protein kinase family. This kinase contains a leucine-zipper domain, and is predominately expressed in neuronal cells. The phosphorylation state of this kinase in synaptic terminals was shown to be regulated by membrane depolarization via calcineurin. This kinase forms heterodimers with leucine zipper containing transcription factors, such as cAMP responsive element binding protein (CREB) and MYC, and thus may play a regulatory role in PKA or retinoic acid induced neuronal differentiation.[6]
Interactions
[edit]MAP3K12 has been shown to interact with MAPK8IP1,[7] MAP2K7[8] and MAPK8IP2.[7]
Role in development
[edit]MAP3K12, otherwise known as DLK, can initiate coordinated signalling cascades that culminate in the phosphorylation of C-Jun N-terminal kinases or JNK. Several experiments have implicated this interaction as having a role in the developing mammalian nervous system.[9] For example, neuronal migration and axon growth are critical components of neuronal development. DLK null mice have defects in neuronal migration, hypoplasia of several different axonal tracts and reduced axon number in various areas of the brain such as the cingulum and internal capsule.[9] In addition, inhibition of DLK or JNK delays radial migration and disrupts the formation of the neocortex in mice.[9] Another important function of the developing mammalian nervous system is neuronal apoptosis. The absence of DLK also protects cultured mice sensory neurons from apoptosis that would normally be triggered by a lack of NGF.[9] This, among other experiments, heavily implicates it as having a role in neuronal apoptosis.
DLK has several different interactions that contribute to mammalian nervous system development. For axon growth, DLK phosphorylates MAP2K4/7 which then phosphorylates JNK, activating it.[9] In neuronal migration DLK phosphorylates MAP2K4/7 which phosphorylates JNK, and also interacts with JIP which then interacts with MAP2K4/7 and JNK.[9] There is a similar interaction for neuronal apoptosis, where DLK phosphorylates JIP3 and MAP2K7, which both phosphorylate JNK.[9] It is evident then that DLK interactions are a versatile and critical part of neuronal development in mammals.
References
[edit]- ^ a b c GRCh38: Ensembl release 89: ENSG00000139625 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000023050 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Reddy UR, Pleasure D (Aug 1994). "Cloning of a novel putative protein kinase having a leucine zipper domain from human brain". Biochem Biophys Res Commun. 202 (1): 613–20. doi:10.1006/bbrc.1994.1972. PMID 8037767.
- ^ a b "Entrez Gene: MAP3K12 mitogen-activated protein kinase kinase kinase 12".
- ^ a b Yasuda J, Whitmarsh AJ, Cavanagh J, Sharma M, Davis RJ (Oct 1999). "The JIP group of mitogen-activated protein kinase scaffold proteins". Mol. Cell. Biol. 19 (10): 7245–54. doi:10.1128/mcb.19.10.7245. PMC 84717. PMID 10490659.
- ^ Merritt SE, Mata M, Nihalani D, Zhu C, Hu X, Holzman LB (Apr 1999). "The mixed lineage kinase DLK utilizes MKK7 and not MKK4 as substrate". J. Biol. Chem. 274 (15): 10195–202. doi:10.1074/jbc.274.15.10195. PMID 10187804.
- ^ a b c d e f g Tedeschi A, Bradke F (May 2013). "The DLK signalling pathway—a double-edged sword in neural development and regeneration". EMBO Reports. 14 (7): 605–614. doi:10.1038/embor.2013.64. PMC 3701236. PMID 23681442.
Further reading
[edit]- Reddy UR, Nycum L, Slavc I, Biegel JA (1995). "Localization of the human zipper protein kinase gene (ZPK) to chromosome 12q13 by fluorescence in situ hybridization and somatic cell hybrid analysis". Genomics. 25 (2): 597–8. doi:10.1016/0888-7543(95)80069-X. PMID 7790002.
- Holzman LB, Merritt SE, Fan G (1995). "Identification, molecular cloning, and characterization of dual leucine zipper bearing kinase. A novel serine/threonine protein kinase that defines a second subfamily of mixed lineage kinases". J. Biol. Chem. 269 (49): 30808–17. doi:10.1016/S0021-9258(18)47353-X. PMID 7983011.
- Hirai S, Izawa M, Osada S, Spyrou G, Ohno S (1996). "Activation of the JNK pathway by distantly related protein kinases, MEKK and MUK". Oncogene. 12 (3): 641–50. PMID 8637721.
- Mata M, Merritt SE, Fan G, Yu GG, Holzman LB (1996). "Characterization of dual leucine zipper-bearing kinase, a mixed lineage kinase present in synaptic terminals whose phosphorylation state is regulated by membrane depolarization via calcineurin". J. Biol. Chem. 271 (28): 16888–96. doi:10.1074/jbc.271.28.16888. PMID 8663324.
- Sakuma H, Ikeda A, Oka S, Kozutsumi Y, Zanetta JP, Kawasaki T (1997). "Molecular cloning and functional expression of a cDNA encoding a new member of mixed lineage protein kinase from human brain". J. Biol. Chem. 272 (45): 28622–9. doi:10.1074/jbc.272.45.28622. PMID 9353328.
- Merritt SE, Mata M, Nihalani D, Zhu C, Hu X, Holzman LB (1999). "The mixed lineage kinase DLK utilizes MKK7 and not MKK4 as substrate". J. Biol. Chem. 274 (15): 10195–202. doi:10.1074/jbc.274.15.10195. PMID 10187804.
- Reddy UR, Basu A, Bannerman P, Ikegaki N, Reddy CD, Pleasure D (1999). "ZPK inhibits PKA induced transcriptional activation by CREB and blocks retinoic acid induced neuronal differentiation". Oncogene. 18 (31): 4474–84. doi:10.1038/sj.onc.1202813. PMID 10442638.
- Douziech M, Laberge G, Grondin G, Daigle N, Blouin R (1999). "Localization of the mixed-lineage kinase DLK/MUK/ZPK to the Golgi apparatus in NIH 3T3 cells". J. Histochem. Cytochem. 47 (10): 1287–96. doi:10.1177/002215549904701008. PMID 10490457.
- Fukuyama K, Yoshida M, Yamashita A, Deyama T, Baba M, Suzuki A, Mohri H, Ikezawa Z, Nakajima H, Hirai S, Ohno S (2000). "MAPK upstream kinase (MUK)-binding inhibitory protein, a negative regulator of MUK/dual leucine zipper-bearing kinase/leucine zipper protein kinase". J. Biol. Chem. 275 (28): 21247–54. doi:10.1074/jbc.M001488200. PMID 10801814.
- Hébert SS, Daviau A, Grondin G, Latreille M, Aubin RA, Blouin R (2000). "The mixed lineage kinase DLK is oligomerized by tissue transglutaminase during apoptosis". J. Biol. Chem. 275 (42): 32482–90. doi:10.1074/jbc.M006528200. PMID 10922377.
- Figueroa C, Tarras S, Taylor J, Vojtek AB (2004). "Akt2 negatively regulates assembly of the POSH-MLK-JNK signaling complex". J. Biol. Chem. 278 (48): 47922–7. doi:10.1074/jbc.M307357200. PMID 14504284.
- Itoh A, Wang Z, Ito Y, Reddy UR, Itoh T (2004). "SP3 acts as a positive regulator on the core promoter of human ZPK gene". Biochem. Biophys. Res. Commun. 313 (3): 612–8. doi:10.1016/j.bbrc.2003.11.152. PMID 14697235.
- Graves PR, Winkfield KM, Haystead TA (2005). "Regulation of zipper-interacting protein kinase activity in vitro and in vivo by multisite phosphorylation". J. Biol. Chem. 280 (10): 9363–74. doi:10.1074/jbc.M412538200. PMID 15611134.
- Robitaille H, Proulx R, Robitaille K, Blouin R, Germain L (2005). "The mitogen-activated protein kinase kinase kinase dual leucine zipper-bearing kinase (DLK) acts as a key regulator of keratinocyte terminal differentiation". J. Biol. Chem. 280 (13): 12732–41. doi:10.1074/jbc.M411619200. hdl:20.500.11794/16685. PMID 15695824.
- Stelzl U, Worm U, Lalowski M, Haenig C, Brembeck FH, Goehler H, Stroedicke M, Zenkner M, Schoenherr A, Koeppen S, Timm J, Mintzlaff S, Abraham C, Bock N, Kietzmann S, Goedde A, Toksöz E, Droege A, Krobitsch S, Korn B, Birchmeier W, Lehrach H, Wanker EE (2005). "A human protein-protein interaction network: a resource for annotating the proteome". Cell. 122 (6): 957–68. doi:10.1016/j.cell.2005.08.029. hdl:11858/00-001M-0000-0010-8592-0. PMID 16169070. S2CID 8235923.