JAG1
Jagged1 (JAG1) is one of five cell surface proteins (ligands) that interact with four receptors in the mammalian Notch signaling pathway. The Notch signaling pathway is a highly conserved pathway that functions to establish and regulate cell fate decisions in many organ systems. Once the JAG1-NOTCH (receptor-ligand) interactions take place, a cascade of proteolytic cleavages is triggered resulting in activation of the transcription for downstream target genes. Located on human chromosome 20, the JAG1 gene is expressed in multiple organ systems in the body and causes the autosomal dominant disorder Alagille syndrome (ALGS) resulting from loss of function mutations within the gene. JAG1 has also been designated as CD339 (cluster of differentiation 339).
Structure and function
[edit]JAG1 was first identified as a ligand that was able to activate notch receptors when the rat gene Jagged encoding a protein homolog was cloned in 1995.[5][6] The structure of the JAG1 protein includes a small intracellular component, a transmembrane motif, proceeded by an extracellular region containing a cystine-rich region, 16 EGF-like repeats, a DSL domain, and finally a signal peptide totaling 1218 amino acids in length over 26 coding exons.[7]
The JAG1 protein encoded by JAG1 is the human homolog of the Drosophila jagged protein.[5] Human JAG1 is one of five ligands for receptors in the NOTCH signaling pathway which helps to determine cellular fate and is active during many developmental stages. The extracellular component of the JAG1 protein physically interacts with its respective Notch receptor. This interaction kicks off a cascade of proteolytic cleavages leading to the original NOTCH intracellular domain being trafficked into the nucleus of the cell leading to the activation of different target genes.[8][9][10][11]
Expression profile and mouse studies
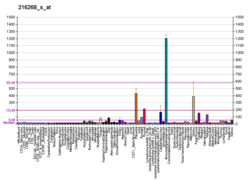
[edit]In situ hybridization and conditional gene knockout studies have helped to demonstrate the role JAG1 plays in development and its effects on different organ systems. In humans, JAG1 has broad expression in many tissue types including the pancreas, heart, placenta, prostate, lung, kidney, thymus, testis, and leucocytes in the adult.[12] In a developing embryo JAG1 expression is concentrated around the pulmonary artery, mesocardium, distal cardic outflow tract, major arteries, metanephros, branchial arches, pancreas, the portal vein, and otocyst.[12] Generally, JAG1 expression patterns correlate with organ systems affected in ALGS, although it is important to note that not all tissues where JAG1 is expressed are affected in ALGS. More recently JAG1 expression has been found to be altered in breast cancer and adrenocortical carcinoma patients.[13][14]
Mouse models where the Jag1 gene is turned off in certain tissues (conditional knockout mouse models) have been used to study the role of Jag1 in many tissue specific areas. While homozygous deletions of Jag1 have been shown to be embryonic lethal in mice, and heterozygous deletions may show only a limited phenotype (involving the eye), mice haploinsufficient for both Jag1 and Notch2 present with the ALGS phenotype.[15] Conditional gene knockout mouse models with Jag1 mutations targeted to the portal vein mesenchyme, endothelium, and cranial neural crest all exhibit features classic to those in individuals with ALGS, highlighting the role of this tissue type in disease origins[16][17][18][19][20]
Disease phenotype
[edit]ALGS is an autosomal dominant multi-system disorder affecting several body systems including the liver, heart, skeleton, eye, facial structure, kidneys and vascular system. The most clinically significant concerns stem from liver, heart, vascular or renal problems. Mutations in JAG1 were first discovered to be responsible for ALGS by researchers at The Children's Hospital of Philadelphia and the National Institutes of Health in 1997.[6] Patients who are clinically consistent with the disorder usually have a mutation in JAG1 (94%), while a smaller 2% have a mutation in NOTCH2.[21] Over half of individuals with mutations in the gene did not inherit it from either parent, and thus have a de novo mutation.[21][22] JAG1 mutation types include protein truncating (splice site, frameshift, and nonsense), missense, and whole gene deletions accounting for 80%, 7%, and 12% respectively. Since all mutation types lead to a patient phenotype, it is thought that haploinsufficiency for JAG1 is the likely disease mechanism of action.[23][24][25] Although individuals can have a range of mutation types in JAG1, all of the known mutations lead to loss of the function of one copy, and, there is no correlation between mutation type or location and disease severity. Though individuals with ALGS have several body systems affected, there is a subset of individuals with JAG1 mutations who present with tetralogy of fallot/pulmonary stenosis that do not show the other clinical signs of the syndrome.[26] Given the variable expressivity of the disease, there may be other genetic or environmental modifiers present beyond the original JAG1 mutation.
More recently, JAG1 expression changes have been implicated in many types of cancer. Specifically, up regulation of JAG1 has been correlated with both poor overall breast cancer survival rates and an enhancement of tumor proliferation in adrenocortical carcinoma patients.[13][27][28][29]
See also
[edit]- Notch signaling
- Alagille syndrome
- Autosomal dominant
- Haploinsufficiency
- Tetralogy of fallot
- In situ hybridization
- Conditional gene knockout
- Cluster of differentiation
Notes
[edit]
The 2015 version of this article was updated by an external expert under a dual publication model. The corresponding academic peer reviewed article was published in Gene and can be cited as: Christopher M Grochowski, Kathleen M Loomes, Nancy B Spinner (6 November 2015). "Jagged1 (JAG1): Structure, expression, and disease associations". Gene. Gene Wiki Review Series. 576 (1 Pt 3): 381–384. doi:10.1016/J.GENE.2015.10.065. ISSN 0378-1119. PMC 4673022. PMID 26548814. Wikidata Q28606584. |
References
[edit]- ^ a b c GRCh38: Ensembl release 89: ENSG00000101384 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000027276 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ a b Lindsell CE, Shawber CJ, Boulter J, Weinmaster G (1995). "Jagged: a mammalian ligand that activates Notch1". Cell. 80 (6): 909–17. doi:10.1016/0092-8674(95)90294-5. PMID 7697721. S2CID 11720367.
- ^ a b Oda T, Elkahloun AG, Pike BL, Okajima K, Krantz ID, Genin A, Piccoli DA, Meltzer PS, Spinner NB, Collins FS, Chandrasekharappa SC (1997). "Mutations in the human Jagged1 gene are responsible for Alagille syndrome". Nat. Genet. 16 (3): 235–42. doi:10.1038/ng0797-235. PMID 9207787. S2CID 5775213.
- ^ Guarnaccia C, Pintar A, Pongor S (2004). "Exon 6 of human Jagged-1 encodes an autonomously folding unit". FEBS Lett. 574 (1–3): 156–60. doi:10.1016/j.febslet.2004.08.022. PMID 15358557. S2CID 34651925.
- ^ Shimizu K, Chiba S, Saito T, Kumano K, Hirai H (2000). "Physical interaction of Delta1, Jagged1, and Jagged2 with Notch1 and Notch3 receptors". Biochem. Biophys. Res. Commun. 276 (1): 385–9. doi:10.1006/bbrc.2000.3469. PMID 11006133.
- ^ Shimizu K, Chiba S, Saito T, Kumano K, Takahashi T, Hirai H (July 2001). "Manic fringe and lunatic fringe modify different sites of the Notch2 extracellular region, resulting in different signaling modulation". J. Biol. Chem. 276 (28): 25753–8. doi:10.1074/jbc.M103473200. PMID 11346656.
- ^ Shimizu K, Chiba S, Kumano K, Hosoya N, Takahashi T, Kanda Y, Hamada Y, Yazaki Y, Hirai H (November 1999). "Mouse jagged1 physically interacts with notch2 and other notch receptors. Assessment by quantitative methods". J. Biol. Chem. 274 (46): 32961–9. doi:10.1074/jbc.274.46.32961. PMID 10551863.
- ^ Shimizu K, Chiba S, Hosoya N, Kumano K, Saito T, Kurokawa M, Kanda Y, Hamada Y, Hirai H (September 2000). "Binding of Delta1, Jagged1, and Jagged2 to Notch2 Rapidly Induces Cleavage, Nuclear Translocation, and Hyperphosphorylation of Notch2". Mol. Cell. Biol. 20 (18): 6913–22. doi:10.1128/MCB.20.18.6913-6922.2000. PMC 88767. PMID 10958687.
- ^ a b Jones EA, Clement-Jones M, Wilson DI (2000). "JAGGED1 expression in human embryos: correlation with the Alagille syndrome phenotype". J. Med. Genet. 37 (9): 658–62. doi:10.1136/jmg.37.9.658. PMC 1734694. PMID 10978356.
- ^ a b Reedijk M, Odorcic S, Chang L, Zhang H, Miller N, McCready DR, Lockwood G, Egan SE (2005). "High-level coexpression of JAG1 and NOTCH1 is observed in human breast cancer and is associated with poor overall survival". Cancer Res. 65 (18): 8530–7. doi:10.1158/0008-5472.CAN-05-1069. PMID 16166334.
- ^ Simon DP, Giordano TJ, Hammer GD (2012). "Upregulated JAG1 enhances cell proliferation in adrenocortical carcinoma". Clin. Cancer Res. 18 (9): 2452–64. doi:10.1158/1078-0432.CCR-11-2371. PMC 3848076. PMID 22427350.
- ^ McCright B, Lozier J, Gridley T (2002). "A mouse model of Alagille syndrome: Notch2 as a genetic modifier of Jag1 haploinsufficiency". Development. 129 (4): 1075–82. doi:10.1242/dev.129.4.1075. PMID 11861489.
- ^ Loomes KM, Underkoffler LA, Morabito J, Gottlieb S, Piccoli DA, Spinner NB, Baldwin HS, Oakey RJ (1999). "The expression of Jagged1 in the developing mammalian heart correlates with cardiovascular disease in Alagille syndrome". Hum. Mol. Genet. 8 (13): 2443–9. doi:10.1093/hmg/8.13.2443. PMID 10556292.
- ^ Hofmann JJ, Briot A, Enciso J, Zovein AC, Ren S, Zhang ZW, Radtke F, Simons M, Wang Y, Iruela-Arispe ML (2012). "Endothelial deletion of murine Jag1 leads to valve calcification and congenital heart defects associated with Alagille syndrome". Development. 139 (23): 4449–60. doi:10.1242/dev.084871. PMC 3509736. PMID 23095891.
- ^ Hofmann JJ, Zovein AC, Koh H, Radtke F, Weinmaster G, Iruela-Arispe ML (2010). "Jagged1 in the portal vein mesenchyme regulates intrahepatic bile duct development: insights into Alagille syndrome". Development. 137 (23): 4061–72. doi:10.1242/dev.052118. PMC 2976287. PMID 21062863.
- ^ High FA, Lu MM, Pear WS, Loomes KM, Kaestner KH, Epstein JA (2008). "Endothelial expression of the Notch ligand Jagged1 is required for vascular smooth muscle development". Proc. Natl. Acad. Sci. U.S.A. 105 (6): 1955–9. Bibcode:2008PNAS..105.1955H. doi:10.1073/pnas.0709663105. PMC 2538864. PMID 18245384.
- ^ Humphreys R, Zheng W, Prince LS, Qu X, Brown C, Loomes K, Huppert SS, Baldwin S, Goudy S (2012). "Cranial neural crest ablation of Jagged1 recapitulates the craniofacial phenotype of Alagille syndrome patients". Hum. Mol. Genet. 21 (6): 1374–83. doi:10.1093/hmg/ddr575. PMC 3465692. PMID 22156581.
- ^ a b Krantz ID, Colliton RP, Genin A, Rand EB, Li L, Piccoli DA, Spinner NB (1998). "Spectrum and frequency of jagged1 (JAG1) mutations in Alagille syndrome patients and their families". Am. J. Hum. Genet. 62 (6): 1361–9. doi:10.1086/301875. PMC 1377154. PMID 9585603.
- ^ Warthen DM, Moore EC, Kamath BM, Morrissette JJ, Sanchez-Lara PA, Sanchez P, Piccoli DA, Krantz ID, Spinner NB (2006). "Jagged1 (JAG1) mutations in Alagille syndrome: increasing the mutation detection rate". Hum. Mutat. 27 (5): 436–43. doi:10.1002/humu.20310. PMID 16575836. S2CID 45080348.
- ^ Penton AL, Leonard LD, Spinner NB (2012). "Notch signaling in human development and disease". Semin. Cell Dev. Biol. 23 (4): 450–7. doi:10.1016/j.semcdb.2012.01.010. PMC 3638987. PMID 22306179.
- ^ Morrissette JD, Colliton RP, Spinner NB (2001). "Defective intracellular transport and processing of JAG1 missense mutations in Alagille syndrome". Hum. Mol. Genet. 10 (4): 405–13. doi:10.1093/hmg/10.4.405. PMID 11157803.
- ^ Crosnier C, Driancourt C, Raynaud N, Dhorne-Pollet S, Pollet N, Bernard O, Hadchouel M, Meunier-Rotival M (1999). "Mutations in JAGGED1 gene are predominantly sporadic in Alagille syndrome". Gastroenterology. 116 (5): 1141–8. doi:10.1016/S0016-5085(99)70017-X. PMID 10220506.
- ^ Bauer RC, Laney AO, Smith R, Gerfen J, Morrissette JJ, Woyciechowski S, Garbarini J, Loomes KM, Krantz ID, Urban Z, Gelb BD, Goldmuntz E, Spinner NB (2010). "Jagged1 (JAG1) mutations in patients with tetralogy of Fallot or pulmonic stenosis". Hum. Mutat. 31 (5): 594–601. doi:10.1002/humu.21231. PMC 2914103. PMID 20437614.
- ^ Dickson BC, Mulligan AM, Zhang H, Lockwood G, O'Malley FP, Egan SE, Reedijk M (2007). "High-level JAG1 mRNA and protein predict poor outcome in breast cancer". Mod. Pathol. 20 (6): 685–93. doi:10.1038/modpathol.3800785. PMID 17507991.
- ^ Li D, Masiero M, Banham AH, Harris AL (2014). "The notch ligand JAGGED1 as a target for anti-tumor therapy". Front Oncol. 4: 254. doi:10.3389/fonc.2014.00254. PMC 4174884. PMID 25309874.
- ^ Lu C, Bonome T, Li Y, Kamat AA, Han LY, Schmandt R, Coleman RL, Gershenson DM, Jaffe RB, Birrer MJ, Sood AK (2007). "Gene alterations identified by expression profiling in tumor-associated endothelial cells from invasive ovarian carcinoma". Cancer Res. 67 (4): 1757–68. doi:10.1158/0008-5472.CAN-06-3700. PMID 17308118.
Further reading
[edit]- Piccoli DA, Spinner NB (2002). "Alagille syndrome and the Jagged1 gene". Semin. Liver Dis. 21 (4): 525–34. doi:10.1055/s-2001-19036. PMID 11745040. S2CID 46554750.
- Lindsell CE, Boulter J, diSibio G, Gossler A, Weinmaster G (1997). "Expression patterns of Jagged, Delta1, Notch1, Notch2, and Notch3 genes identify ligand-receptor pairs that may function in neural development". Mol. Cell. Neurosci. 8 (1): 14–27. doi:10.1006/mcne.1996.0040. PMID 8923452. S2CID 8058790.
- Zimrin AB, Pepper MS, McMahon GA, Nguyen F, Montesano R, Maciag T (1997). "An antisense oligonucleotide to the notch ligand jagged enhances fibroblast growth factor-induced angiogenesis in vitro". J. Biol. Chem. 271 (51): 32499–502. doi:10.1074/jbc.271.51.32499. PMID 8955070.
- Li L, Krantz ID, Deng Y, Genin A, Banta AB, Collins CC, Qi M, Trask BJ, Kuo WL, Cochran J, Costa T, Pierpont ME, Rand EB, Piccoli DA, Hood L, Spinner NB (1997). "Alagille syndrome is caused by mutations in human Jagged1, which encodes a ligand for Notch1". Nat. Genet. 16 (3): 243–51. doi:10.1038/ng0797-243. PMID 9207788. S2CID 8794354.
- Oda T, Elkahloun AG, Meltzer PS, Chandrasekharappa SC (1997). "Identification and cloning of the human homolog (JAG1) of the rat Jagged1 gene from the Alagille syndrome critical region at 20p12". Genomics. 43 (3): 376–9. doi:10.1006/geno.1997.4820. PMID 9268641.
- Li L, Milner LA, Deng Y, Iwata M, Banta A, Graf L, Marcovina S, Friedman C, Trask BJ, Hood L, Torok-Storb B (1998). "The human homolog of rat Jagged1 expressed by marrow stroma inhibits differentiation of 32D cells through interaction with Notch1". Immunity. 8 (1): 43–55. doi:10.1016/S1074-7613(00)80457-4. PMID 9462510.
- Hock B, Böhme B, Karn T, Yamamoto T, Kaibuchi K, Holtrich U, Holland S, Pawson T, Rübsamen-Waigmann H, Strebhardt K (1998). "PDZ-domain-mediated interaction of the Eph-related receptor tyrosine kinase EphB3 and the ras-binding protein AF6 depends on the kinase activity of the receptor". Proc. Natl. Acad. Sci. U.S.A. 95 (17): 9779–84. Bibcode:1998PNAS...95.9779H. doi:10.1073/pnas.95.17.9779. PMC 21413. PMID 9707552.
- Gray GE, Mann RS, Mitsiadis E, Henrique D, Carcangiu ML, Banks A, Leiman J, Ward D, Ish-Horowitz D, Artavanis-Tsakonas S (1999). "Human Ligands of the Notch Receptor". Am. J. Pathol. 154 (3): 785–94. doi:10.1016/S0002-9440(10)65325-4. PMC 1866435. PMID 10079256.
- Bash J, Zong WX, Banga S, Rivera A, Ballard DW, Ron Y, Gélinas C (1999). "Rel/NF-kappaB can trigger the Notch signaling pathway by inducing the expression of Jagged1, a ligand for Notch receptors". EMBO J. 18 (10): 2803–11. doi:10.1093/emboj/18.10.2803. PMC 1171361. PMID 10329626.
- Pilia G, Uda M, Macis D, Frau F, Crisponi L, Balli F, Barbera C, Colombo C, Frediani T, Gatti R, Iorio R, Marazzi MG, Marcellini M, Musumeci S, Nebbia G, Vajro P, Ruffa G, Zancan L, Cao A, DeVirgilis S (2000). "Jagged-1 mutation analysis in Italian Alagille syndrome patients". Hum. Mutat. 14 (5): 394–400. doi:10.1002/(SICI)1098-1004(199911)14:5<394::AID-HUMU5>3.0.CO;2-1. PMID 10533065. S2CID 45123109.
- Wong MK, Prudovsky I, Vary C, Booth C, Liaw L, Mousa S, Small D, Maciag T (2000). "A non-transmembrane form of Jagged-1 regulates the formation of matrix-dependent chord-like structures". Biochem. Biophys. Res. Commun. 268 (3): 853–9. doi:10.1006/bbrc.2000.2173. PMID 10679295.
- Heritage ML, MacMillan JC, Colliton RP, Genin A, Spinner NB, Anderson GJ (2000). "Jagged1 (JAG1) mutation detection in an Australian Alagille syndrome population". Hum. Mutat. 16 (5): 408–16. doi:10.1002/1098-1004(200011)16:5<408::AID-HUMU5>3.0.CO;2-9. PMID 11058898. S2CID 22168402.
- Karanu FN, Murdoch B, Gallacher L, Wu DM, Koremoto M, Sakano S, Bhatia M (2000). "The Notch Ligand Jagged-1 Represents a Novel Growth Factor of Human Hematopoietic Stem Cells". J. Exp. Med. 192 (9): 1365–72. doi:10.1084/jem.192.9.1365. PMC 2193352. PMID 11067884.
External links
[edit]- GeneReviews/NCBI/UW/NIH entry on Alagille syndrome
- OMIM entries on Alagille syndrome
- JAG1+protein,+human at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Alagille syndrome
This article incorporates text from the United States National Library of Medicine, which is in the public domain.