Benzydamine
This is an old revision of this page, as edited by 83.23.189.85 (talk) at 14:11, 11 May 2010 (→Side effects). The present address (URL) is a permanent link to this revision, which may differ significantly from the current revision.
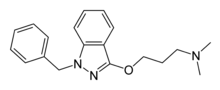 | |
| Clinical data | |
|---|---|
| Pregnancy category |
|
| Routes of administration | Oral, topical |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Protein binding | <20% |
| Elimination half-life | 13 hours |
| Excretion | Renal |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.010.354 |
| Chemical and physical data | |
| Formula | C19H23N3O |
| Molar mass | 309.405 g/mol g·mol−1 |
| 3D model (JSmol) | |
| |
| (verify) | |
Benzydamine, available as the hydrochloride, is a locally-acting nonsteroidal anti-inflammatory drug with local anaesthetic and analgesic properties providing both rapid and extended pain relief as well as a significant anti-inflammatory treatment for the painful inflammatory conditions of the mouth and throat.[1]
It selectively binds to inflamed tissues (Prostaglandin synthetase inhibitor) is virtually free of any adverse systemic effects.
It may be used alone or as an adjunct to other therapy giving the possibility of increased therapeutic effect with little risk of interaction.
Availability
It is available in a mouth wash named Tantum Verde and Tantum Rosa across Europe.
It is available in gel or oral rinse form with trade name Tantum (Elder Pharmaceutical) in India
In the UK it is available without prescription from pharmacies; it is produced in the UK by Meda Pharmaceuticals under the trade name Difflam, and is available as a spray, oral rinse and cream, and as lozenges.
In Sweden it is sold by Antula Healthcare, under he trade name Zyx, as lozenges.
In Australia it is available from 3M as Difflam-C Alcohol & Colour Free Solution, Difflam 3% Gel, Difflam Extra Strength Gel 5%, Difflam-C Solution, Difflam Solution (including Difflam Throat Spray), Difflam Cream and Difflam Lozenges.[2]
It is sold in eastern Europe without prescription as Tantum Rosa - a vaginal antiseptic and anti-inflammatory, containing 0.5 g of benzydamine hydrochloride to be reconstituted with clean water to a 0.1% (1 mg/mL) solution for vaginal enema/instillation.
In Brazil it is sold over-the-counter under the name "Benflogin", with each box containing 20 pills (50mg each).
In Pakistan it is sold by prescription under the name Tantum Capsule (50mg).
It is available in a cream and gel named Tantum Fort across Egypt by EIPICo.
It is available with prescription as a mouthwash named Novo-Benzydamine (Novopharm) in Canada.
In Mexico it is sold by prescription under the name Vantal as a mouth rinse.
In South Africa it is sold as Andolex/Andolex-C Oral Rinse, Spray and Lozenges and is manufactured by 3M Pharmaceuticals S.A. (Pty) Ltd.[3]
In Argentina it is sold as a mouthwash under the name Ernex, manufactured by Casasco.
Indications
Odontostomatology: gingivitis, stomatitis, glossitis, aphthous ulcers, dental surgery and oral ulceration due to radiation therapy.
Otorhinolaryngology: pharyngitis, tonsillitis, post-tonsillectomy, radiation or intubation mucositis.
Contraindications
There are no contraindications to the use of Benzydamine except for known hypersensitivity.
Side effects
Benzydamine is well tolerated. Occasionally oral tissue numbness or stinging sensations may occur. Benzydamine may be abused recreationally.[4] In oral dosages of 500 mg to 3000 mg it is a deliriant and CNS stimulant (a cough drop has 3 mg dose), popular in Poland, Brazil and Romania. In Brazil it is very popular and widely used for recreational purposes, particularly among teenagers and as a club drug.[5] A person in a benzydamine trip may experience (because of large dopamine release) a feeling of well-being, euphoria and, in higher doses, hallucinations, paranoia, dry mouth and convulsions. The trip can last up to 8 hours, after that the user becomes tired and quiet, but sleeping is almost impossible. Unlike other NSAIDs, it does not inhibit cyclooxygenase or lipooxygenase, and is not ulcerogenic.[4]
Chemistry
Lisciani, R (1968). "Researches on the topical activity of benzydamine". European Journal of Pharmacology. 3: 157. doi:10.1016/0014-2999(68)90069-1.
References
- ^ Turnbull RS. Benzydamine Hydrochloride (Tantum) in the management of oral inflammatory conditions. Journal of the Canadian Dental Association. 1995 Feb;61(2):127-34.
- ^ Material Safety Data Sheets for Difflam varieties sold in Australia
- ^ 3M Pharmaceuticals S.A. (Pty) Ltd
- ^ a b Anand JS, Glebocka ML, Korolkiewicz RP. Recreational abuse with benzydamine hydrochloride (tantum rosa). Clinical Toxicology (Philadelphia, Pa). 2007;45(2):198-9.
- ^ Alves L (n.d.). "Benflogin (cloridrato de benzidamina)" (in Portuguese). Brasil Escola. Retrieved on November 5, 2008.
External links
Stomatological preparations (A01) | |
|---|---|
| Caries prophylaxis | |
| Infection and antiseptics |
|
| Corticosteroids (Glucocorticoids) | |
| Other | |
| pyrazolones / pyrazolidines | |
|---|---|
| salicylates | |
| acetic acid derivatives and related substances | |
| oxicams | |
| propionic acid derivatives (profens) |
|
| n-arylanthranilic acids (fenamates) | |
| COX-2 inhibitors (coxibs) | |
| other | |
| NSAID combinations | |
Key: underline indicates initially developed first-in-class compound of specific group; #WHO-Essential Medicines; †withdrawn drugs; ‡veterinary use. | |
| pyrazolones / pyrazolidines | |
|---|---|
| salicylates | |
| acetic acid derivatives and related substances | |
| oxicams | |
| propionic acid derivatives (profens) |
|
| n-arylanthranilic acids (fenamates) | |
| COX-2 inhibitors (coxibs) | |
| other | |
| NSAID combinations | |
Key: underline indicates initially developed first-in-class compound of specific group; #WHO-Essential Medicines; †withdrawn drugs; ‡veterinary use. | |
| Anti-inflammatory preparations, non-steroids |
| ||||||
|---|---|---|---|---|---|---|---|
| Capsaicin derivatives | |||||||
| Other | |||||||
| |||||||||||||||||||||||||
| Prolactin inhibitors | |
|---|---|
| Anti-inflammatory products for vaginal administration | |
- CS1 Portuguese-language sources (pt)
- Drugs with non-standard legal status
- ECHA InfoCard ID from Wikidata
- Chem-molar-mass both hardcoded and calculated
- Infobox-drug molecular-weight unexpected-character
- Pages using infobox drug with unknown parameters
- Articles without EBI source
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without InChI source
- Articles without UNII source
- Articles with missing files
