Cannabidiol
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Epidiolex |
| AHFS/Drugs.com | International Drug Names |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 13-19% (oral),[1] 11-45% (mean 31%; inhaled)[2] |
| Elimination half-life | 9 h[1] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.215.986 |
| Chemical and physical data | |
| Formula | C21H30O2 |
| Molar mass | 314.4636 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 66 °C (151 °F) |
| Boiling point | 180 °C (356 °F) (range: 160–180 °C)[3] |
| |
| |
| (verify) | |
| Part of a series on |
| Cannabis |
|---|
 |
Cannabidiol (CBD) is one of at least 85 active cannabinoids identified in cannabis.[4] It is a major phytocannabinoid, accounting for up to 40% of the plant's extract.[5] CBD is considered to have a wider scope of medical applications than tetrahydrocannabinol (THC). An orally-administered liquid containing CBD has received orphan drug status in the US, for use as a treatment for Dravet syndrome, under the brand name Epidiolex.[6]
Clinical applications

Neurological effects
Cannabidiol has been seen to be anticonvulsant in animals, but controlled studies in humans are lacking.[7] Randomized trials in treatment-resistant epilepsy syndromes are planned in 2015.
A 2010 study found that strains of cannabis containing higher concentrations of cannabidiol did not produce short-term memory impairment vs. strains with similar concentrations of THC, but lower CBD. The researchers attributed these attenuated memory effects to CBD's role as a CB1 antagonist.[8] Transdermal CBD is neuroprotective in animals.[9]
Cannabidiol's strong antioxidant properties have been shown to play a role in the compound's neuroprotective and anti-ischemic effects.[10]
Dravet syndrome
Dravet syndrome is a rare form of epilepsy that is difficult to treat. It is a catastrophic form of intractable epilepsy that begins in infancy. Initial seizures are most often prolonged events and in the second year of life other seizure types begin to emerge.[11] While high profile and anecdotal reports of results from high-CBD/low-THC preparations have sparked interest in treatment with cannabinoids,[12] there is insufficient medical evidence to draw conclusions about their safety or efficacy.[12][13]
Psychotropic effect
CBD has anti-psychotic effects and may counteract the potential psychotomimetic effects of THC on individuals with latent schizophrenia;[5] some reports show it to be an alternative treatment for schizophrenia that is safe and well-tolerated.[14] Studies have shown CBD may reduce schizophrenic symptoms due to its apparent ability to stabilize disrupted or disabled NMDA receptor pathways in the brain, which are shared and sometimes contested by norepinephrine and GABA.[14][15] Leweke et al. performed a double blind, 4 week, explorative controlled clinical trial to compare the effects of purified cannabidiol and the atypical antipsychotic amisulpride on improving the symptoms of schizophrenia in 42 patients with acute paranoid schizophrenia. Both treatments were associated with a significant decrease of psychotic symptoms after 2 and 4 weeks as assessed by Brief Psychiatric Rating Scale and Positive and Negative Syndrome Scale. While there was no statistical difference between the two treatment groups, cannabidiol induced significantly fewer side effects (extrapyramidal symptoms, increase in prolactin, weight gain) when compared to amisulpride.[16] A phase 2 study of 88 schizophenic patients who failed to respond to a first-line anti-psychotic had cannabidol(or placebo) added on.[17] Cannabidiol improved positive and cognitive symptoms over six weeks with no severe adverse effects.
Studies have shown cannabidiol decreases activity of the limbic system [18] and decreases social isolation induced by THC.[19] Cannabidiol has also been shown to reduce anxiety in social anxiety disorder.[20][21]
Chronic cannabidiol administration in rats was found to produce reactions suggesting anxiety, indicating that prolonged treatment with cannabidiol might lead to anxiety.[22] Those results have been contested by Gururajan,[23] and contradict Réus,[24] whose experimentation cover the same duration.
Cannabidiol has demonstrated antidepressant-like effects in animal models of depression.[25][26][27]
CBD-enhanced cannabis
Selective breeding by growers in the USA dramatically lowered the CBD content of cannabis; their customers preferred varietals that were more mind-altering due to a higher THC, lower CBD content.[28] To meet the demands of medical cannabis patients, growers are currently developing more CBD-rich strains.[29]
Charlotte's Web, a cannabis plant, was developed in 2011 by crossbreeding a strain of marijuana with industrial hemp. This process created a variety with less tetrahydrocannabinol (THC) and more cannabidiol (CBD) than typical varieties of cannabis the US (see Charlotte's Web in the section above).
In November 2012, Tikun Olam, an Israeli medical cannabis facility announced a new strain of the plant which has only cannabidiol as an active ingredient, and virtually no THC, providing some of the medicinal benefits of cannabis without the euphoria.[30][31] The researchers said the cannabis plant, enriched with CBD, "can be used for treating diseases like rheumatoid arthritis, colitis, liver inflammation, heart disease and diabetes". Research on CBD-enhanced cannabis began in 2009, resulting in Avidekel, a cannabis strain that contains 15.8% CBD and less than 1% THC. Raphael Mechoulam, a cannabinoid researcher, said "...Avidekel is thought to be the first CBD-enriched cannabis plant with no THC to have been developed in Israel".[32] In February 2014, a patent application was filed for a cannabis plant named 'avidekel'.[33]
Industrial hemp
Several industrial hemp varieties can be legally cultivated in western Europe. A variety such as "Fedora 17" has a cannabinoid profile consistently around 1% cannabidiol (CBD) with THC less than 0.1%.[34]
Extraction can be done with olive oil, ethanol, or CO2, and other nonpolar to semipolar solvents.[citation needed]
Pharmacology
Pharmacodynamics
Cannabidiol has a very low affinity for CB1 and CB2 receptors but acts as an indirect antagonist of their agonists.[10][35] While one would assume that this would cause cannabidiol to reduce the effects of THC, it may potentiate THC's effects by increasing CB1 receptor density or through another CB1-related mechanism.[36] It may also extend the duration of the effects of THC via inhibition of the cytochrome P-450-3A and 2C enzymes.[37]
Recently, it was found to be an antagonist at the putative new cannabinoid receptor, GPR55, a GPCR expressed in the caudate nucleus and putamen.[38] Cannabidiol has also been shown to act as a 5-HT1A receptor agonist,[39] an action which may be involved in its antidepressant,[25][40] anxiolytic,[40][41] and neuroprotective[42][43] effects. Cannabidiol is an allosteric modulator of μ and δ-opioid receptors.[44] Cannabidiol's pharmacological effects have also been attributed to PPAR-γ receptor agonism and intracellular calcium release.[5]
Research suggests that CBD may exert some of its pharmacological action through its inhibition of FAAH, which may in turn increase the levels of endocannabinoids, such as anandamide, produced by the body.[45]
Pharmacokinetic interactions
There is some preclinical evidence to suggest that cannabidiol may reduce THC clearance, modestly increasing THC's plasma concentrations resulting in a greater amount of THC available to receptors, increasing the effect of THC in a dose-dependent manner.[46][47] Despite this the available evidence in humans suggests no significant effect of CBD on THC plasma levels.[48]
Pharmaceutical preparations
Nabiximols (USAN, trade name Sativex) is an aerosolized mist for oral administration containing a near 1:1 ratio of CBD and THC. The drug was approved by Canadian authorities in 2005 to alleviate pain associated with multiple sclerosis.[49][50][51]
Isomerism
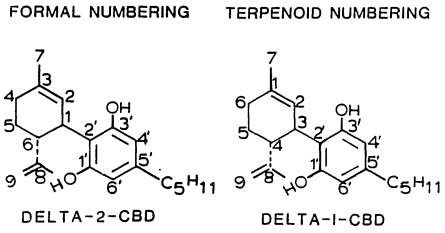
| 7 double bond isomers and their 30 stereoisomers | ||||||||
|---|---|---|---|---|---|---|---|---|
| Formal numbering | Terpenoid numbering | Number of stereoisomers | Natural occurrence | Convention on Psychotropic Substances Schedule | Structure | |||
| Short name | Chiral centers | Full name | Short name | Chiral centers | ||||
| Δ5-cannabidiol | 1 and 3 | 2-(6-isopropenyl-3-methyl-5-cyclohexen-1-yl)-5-pentyl-1,3-benzenediol | Δ4-cannabidiol | 1 and 3 | 4 | No | unscheduled | 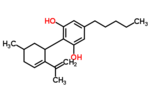
|
| Δ4-cannabidiol | 1, 3 and 6 | 2-(6-isopropenyl-3-methyl-4-cyclohexen-1-yl)-5-pentyl-1,3-benzenediol | Δ5-cannabidiol | 1, 3 and 4 | 8 | No | unscheduled | 
|
| Δ3-cannabidiol | 1 and 6 | 2-(6-isopropenyl-3-methyl-3-cyclohexen-1-yl)-5-pentyl-1,3-benzenediol | Δ6-cannabidiol | 3 and 4 | 4 | ? | unscheduled | 
|
| Δ3,7-cannabidiol | 1 and 6 | 2-(6-isopropenyl-3-methylenecyclohex-1-yl)-5-pentyl-1,3-benzenediol | Δ1,7-cannabidiol | 3 and 4 | 4 | No | unscheduled | 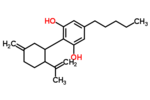
|
| Δ2-cannabidiol | 1 and 6 | 2-(6-isopropenyl-3-methyl-2-cyclohexen-1-yl)-5-pentyl-1,3-benzenediol | Δ1-cannabidiol | 3 and 4 | 4 | Yes | unscheduled | 
|
| Δ1-cannabidiol | 3 and 6 | 2-(6-isopropenyl-3-methyl-1-cyclohexen-1-yl)-5-pentyl-1,3-benzenediol | Δ2-cannabidiol | 1 and 4 | 4 | No | unscheduled | 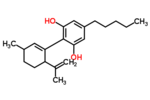
|
| Δ6-cannabidiol | 3 | 2-(6-isopropenyl-3-methyl-6-cyclohexen-1-yl)-5-pentyl-1,3-benzenediol | Δ3-cannabidiol | 1 | 2 | No | unscheduled | 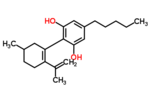
|
See also: Tetrahydrocannabinol#Isomerism, Abnormal cannabidiol.
Chemistry
Cannabidiol is insoluble in water but soluble in organic solvents such as pentane. At room temperature, it is a colorless crystalline solid.[52] In strongly basic media and the presence of air, it is oxidized to a quinone.[53] Under acidic conditions it cyclizes to THC.[54] The synthesis of cannabidiol has been accomplished by several research groups.[55][56][57]
Biosynthesis
Cannabis produces CBD-carboxylic acid through the same metabolic pathway as THC, until the last step, where CBDA synthase performs catalysis instead of THCA synthase.[58]
Legal status
Cannabidiol is not scheduled by the Convention on Psychotropic Substances.
Legal Status in Canada
Cannabidiol is a Schedule II drug in Canada.[59]
Legal Status in the United States
The legal status of Cannabidiol in the United States at the federal level is not immediately clear. The Controlled Substances Act (CSA) does not specifically list cannabidiol in Schedule I[60] nor in any of the other schedules,[61] however it appears that the Drug Enforcement Administration (DEA) presumes to assert authority to regulate cannabidiol as a Schedule I controlled substance.[62][63] The legal basis for that presumed authority is unclear.
The drug Schedules list "Tetrahydrocannabinols" and "marihuana" both as Schedule I drugs under the Controlled Substances Act,[60] however cannabidiol is unlikely to be considered as a Schedule I drug on the basis of being covered by the listing of "Marihuana" or by the listing of "Tetrahydrocannabinols" under Schedule I of the CSA.
- "Marijuana" has a DEA Drug Code of 7360 (distinct from cannabidiol's Drug Code of 7372) and is defined by the CSA as "all parts of the plant Cannabis sativa L., whether growing or not; the seeds thereof; the resin extracted from any part of such plant; and every compound, manufacture, salt, derivative, mixture, or preparation of such plant, its seeds or resin." Exempted from regulation under the definition are "the mature stalks of such plant, fiber produced from such stalks, oil or cake made from the seeds of such plant, any other compound, manufacture, salt, derivative, mixture, or preparation of such mature stalks (except the resin extracted therefrom), fiber, oil, or cake, or the sterilized seed of such plant which is incapable of germination."[64] A DEA Interpretive Rule published in 2001 states that the "definition of marijuana was intended to include those parts of marijuana which contain THC and to exclude those parts which do not. ... The legislative history is absolutely clear that Congress meant to outlaw all plants popularly known as marijuana to the extent those plants possessed THC."Cite error: The
<ref>tag has too many names (see the help page). Cannabidiol isolated by extraction from marijuana sources does not contain THC, and synthetically produced cannabidiol does not contain THC either. It therefore stands to reason that cannabidiol is not covered under the prohibition on marijuana.
- "Tetrahydrocannabinols" listed under Schedule I of the CSA are unlikely to include cannabidiol. Tetrahydrocannabinols are defined as follows:
Unless specifically excepted or unless listed in another schedule, any material, compound, mixture, or preparation, which contains any quantity of the following hallucinogenic substances, or which contains any of its salts, isomers, and salts of isomers whenever the existence of such salts, isomers, and salts of isomers is possible within the specific chemical designation (for purposes of this paragraph only, the term "isomer" includes the optical, position and geometric isomers):
- (31) Tetrahydrocannabinols (DEA Drug Code: 7370)
- Meaning tetrahydrocannabinols naturally contained in a plant of the genus Cannabis (cannabis plant), as well as synthetic equivalents of the substances contained in the cannabis plant, or in the resinous extractives of such plant, and/or synthetic substances, derivatives, and their isomers with similar chemical structure and pharmacological activity to those substances contained in the plant, such as the following:
- 1 cis or trans tetrahydrocannabinol, and their optical isomers
- 6 cis or trans tetrahydrocannabinol, and their optical isomers
- 3,4 cis or trans tetrahydrocannabinol, and its optical isomers
- (Since nomenclature of these substances is not internationally standardized, compounds of these structures, regardless of numerical designation of atomic positions covered.)
Since cannabidiol is chemically not a tetrahydrocannabinol (nor indeed a "cannabinol" of any kind) and cannabidiol has a DEA Drug Code of 7372 (distinct from Tetrahydrocannabinols' designated Drug Code of 7370),[62] it stands to reason that cannabidiol is not considered one of the drugs placed into Schedule I under the listing of "Tetrahydrocannabinols" in the CSA.
Furthermore, cannabidiol was not placed into Schedule I when The Controlled Substances Act was amended in July 2012 with the US Congress' passing of the Synthetic Drug Abuse Prevention Act of 2012 (SDAPA) (which came into effect on January 4, 2013[65])[66] to ban various cannabinoids, cathinones, and phenethylamines.[60] The part adding to Schedule I various "cannabimimetic agents" which include molecules more closely resembling so-called "classically" structured cannabinoids reads as follows:
(d)
(1) Unless specifically exempted or unless listed in another schedule, any material, compound, mixture, or preparation which contains any quantity of cannabimimetic agents, or which contains their salts, isomers, and salts of isomers whenever the existence of such salts, isomers, and salts of isomers is possible within the specific chemical designation.
- (2) In paragraph (1):
- (A) The term “cannabimimetic agents” means any substance that is a cannabinoid receptor type 1 (CB1 receptor) agonist as demonstrated by binding studies and functional assays within any of the following structural classes:
- (i) 2-(3-hydroxycyclohexyl)phenol with substitution at the 5-position of the phenolic ring by alkyl or alkenyl, whether or not substituted on the cyclohexyl ring to any extent.
Cannabidiol, while being a more "classically structured" cannabinoid (not like the much more recently discovered cannabinoid receport agonists with indole rings such as many of the JWH- and AM- named series), was not on the list of specifically newly banned cannabinoids (even among those with a more so-called "classic structure"),[65][60] and it does not fall into the category of unlisted cannabinoids which are caught by the definition above for several reasons. Primarily, CBD is not a CB1 agonist; it is a CB1 antagonist.[67][8] Also, unlike CP 47,497's homologues and similar synthetic "classical structured cannabinoids" which the above definition was written carefully to include, the cannabidiol molecule has a cyclohexene ring where the amended law requires a cyclohexane ring, and further cannabidiol does not have the required 3-hydroxyl moiety bonded to its cyclohexenyl functional group where the law requires a hydroxyl moiety bonded to the 3- position of a cyclohexyl functional group.
Extracts and concentrates of hemp products which are high in cannabidiol content are very likely legal under US federal law as long as they meet certain requirements. Marihuana is defined by 21 U.S.C. §802(16), which is part of the Controlled Substances Act, and it has a DEA Number / Drug Code of 7360. Exempted from regulation under the definition of marihana is "the mature stalks of such plant, fiber produced from such stalks, oil or cake made from the seeds of such plant, any other compound, manufacture, salt, derivative, mixture, or preparation of such mature stalks (except the resin extracted therefrom), fiber, oil, or cake, or the sterilized seed of any such plant which is incapable of germination."[64][68][69] Under this exception, what are known as industrial hemp-finished products are legally imported into the United States each year.[70] Hemp finished products, including hemp oil and extracts of hemp products which are high in cannabidiol, are legal in the United States for this reason.
US 507 Patent
In October 2003, U.S. patent U.S. patent 6,630,507 entitled "Cannabinoids as antioxidants and neuroprotectants" was assigned to "The United States Of America As Represented By The Department Of Health And Human Services." The patent was filed in April 1999 and listed as the inventors: Aidan J. Hampson, Julius Axelrod, and Maurizio Grimaldi, who all held positions at the National Institute of Mental Health (NIMH) in Bethesda, MD, which is part of the National Institutes of Health (NIH), an agency of the United States Department of Health and Human Services (HHS). The patent mentions cannabidiol's ability as an antiepileptic, to lower intraocular pressure in the treatment of glaucoma, lack of toxicity or serious side effects in large acute doses, its neuroprotectant properties, its ability to prevent neurotoxicity mediated by NMDA, AMPA, or kainate receptors; its ability to attenuate glutamate toxicity, its ability to protect against cellular damage, its ability to protect brains from ischemic damage, its anxiolytic effect, and its superior antioxidant activity which can be used in the prophylaxis and treatment of oxidation associated diseases.[71]
"Oxidative associated diseases include, without limitation, free radical associated diseases, such as ischemia, ischemic reperfusion injury, inflammatory diseases, systemic lupus erythematosus, myocardial ischemia or infarction, cerebrovascular accidents (such as a thromboembolic or hemorrhagic stroke) that can lead to ischemia or an infarct in the brain, operative ischemia, traumatic hemorrhage (for example a hypovolemic stroke that can lead to CNS hypoxia or anoxia), spinal cord trauma, Down's syndrome, Crohn's disease, autoimmune diseases (e.g. rheumatoid arthritis or diabetes), cataract formation, uveitis, emphysema, gastric ulcers, oxygen toxicity, neoplasia, undesired cellular apoptosis, radiation sickness, and others. The present invention is believed to be particularly beneficial in the treatment of oxidative associated diseases of the CNS, because of the ability of the cannabinoids to cross the blood brain barrier and exert their antioxidant effects in the brain. In particular embodiments, the pharmaceutical composition of the present invention is used for preventing, arresting, or treating neurological damage in Parkinson's disease, Alzheimer's disease and HIV dementia; autoimmune neurodegeneration of the type that can occur in encephalitis, and hypoxic or anoxic neuronal damage that can result from apnea, respiratory arrest or cardiac arrest, and anoxia caused by drowning, brain surgery or trauma (such as concussion or spinal cord shock)."[71]
On November 17, 2011, the Federal Register published that the National Institutes of Health of the United States Department of Health and Human Services was "contemplating the grant of an exclusive patent license to practice the invention embodied in U.S. Patent 6,630,507" to the company KannaLife based in New York, for the development and sale of cannabinoid and cannabidiol based therapeutics for the treatment of hepatic encephalopathy in humans.[72][73][74]
On July 9, 2012 — KannaLife Sciences, Inc. (“KannaLife”) Signed an Exclusive License Agreement With National Institutes of Health – Office of Technology Transfer (“NIH-OTT”) aka the United States Federal Government for the Commercialization of U.S. Patent 6,630,507, “Cannabinoids as Antioxidants and Neuroprotectants” (the “’507 Patent”).[71]
References
- ^ a b Mechoulam R, Parker LA, Gallily R (November 2002). "Cannabidiol: an overview of some pharmacological aspects". J Clin Pharmacol (Review). 42 (11 Suppl): 11S–19S. doi:10.1177/0091270002238789. PMID 12412831.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Scuderi C, Filippis DD, Iuvone T, Blasio A, Steardo A, Esposito G (May 2009). "Cannabidiol in medicine: a review of its therapeutic potential in CNS disorders". Phytother Res (Review). 23 (5): 597–602. doi:10.1002/ptr.2625. PMID 18844286.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ [unreliable medical source?] McPartland JM, Russo EB (2001). "Cannabis and cannabis extracts: greater than the sum of their parts?" (PDF). Journal of Cannabis Therapeutics. 1 (3/4): 103–132. doi:10.1300/J175v01n03_08.
- ^ Borgelt LM, Franson KL, Nussbaum AM, Wang GS (February 2013). "The pharmacologic and clinical effects of medical cannabis". Pharmacotherapy (Review). 33 (2): 195–209. doi:10.1002/phar.1187. PMID 23386598.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b c Campos AC, Moreira FA, Gomes FV, Del Bel EA, Guimarães FS (December 2012). "Multiple mechanisms involved in the large-spectrum therapeutic potential of cannabidiol in psychiatric disorders". Philos. Trans. R. Soc. Lond., B, Biol. Sci. (Review). 367 (1607): 3364–78. doi:10.1098/rstb.2011.0389. PMC 3481531. PMID 23108553.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Wilner, AN (25 March 2014). "Marijuana for Epilepsy: Weighing the Evidence". Medscape Neurology. WebMD. Retrieved 2 April 2014.
- ^ Devinsky, Orrin; Cilio, Maria Roberta; Cross, Helen; Fernandez-Ruiz, Javier; French, Jacqueline; Hill, Charlotte; Katz, Russell; Di Marzo, Vincenzo; Jutras-Aswad, Didier (2014-06-01). "Cannabidiol: pharmacology and potential therapeutic role in epilepsy and other neuropsychiatric disorders". Epilepsia. 55 (6): 791–802. doi:10.1111/epi.12631. ISSN 1528-1167. PMID 24854329.
- ^ a b Morgan, C. J. (October 2010). "Impact of cannabidiol on the acute memory and psychotomimetic effects of smoked cannabis: naturalistic study: naturalistic study [corrected]". British Journal of Psychiatry. 197 (4): 285–90. doi:10.1192/bjp.bp.110.077503. PMID 20884951.
- ^ Liput DJ, Hammell DC, Stinchcomb AL, Nixon K (2013). "Transdermal delivery of cannabidiol attenuates binge alcohol-induced neurodegeneration in a rodent model of an alcohol use disorder". Pharmacology Biochemistry and Behavior. 111: 120–7. doi:10.1016/j.pbb.2013.08.013. PMID 24012796.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b Mechoulam R, Peters M, Murillo-Rodriguez E, Hanus LO (August 2007). "Cannabidiol--recent advances". Chem. Biodivers. (Review). 4 (8): 1678–92. doi:10.1002/cbdv.200790147. PMID 17712814.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ http://www.dravetfoundation.org/dravet-syndrome/what-is-dravet-syndrome#sthash.jAC0bZ89.dpuf What is Dravet Syndrome?
- ^ a b Melville, Nancy A. (14 Aug 2013), Seizure Disorders Enter Medical Marijuana Debate, Medscape Medical News., retrieved 2014-01-14
{{citation}}: Italic or bold markup not allowed in:|publisher=(help) - ^ Gloss D, Vickrey B (13 June 2012). "Cannabinoids for epilepsy". Cochrane Database Syst Rev (Review). 6: CD009270. doi:10.1002/14651858.CD009270.pub2. PMID 22696383.
- ^ a b Zuardi AW, Crippa JA, Hallak JE, Moreira FA, Guimarães FS (April 2006). "Cannabidiol, a Cannabis sativa constituent, as an antipsychotic drug". Braz. J. Med. Biol. Res. (Review). 39 (4): 421–9. doi:10.1590/S0100-879X2006000400001. PMID 16612464.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Long LE, Malone DT, Taylor DA (2005). "Cannabidiol Reverses MK-801-Induced Disruption of Prepulse Inhibition in Mice". Neuropsychopharmacology. 31 (4): 795–803. doi:10.1038/sj.npp.1300838. PMID 16052245.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Leweke FM, Piomelli D, Pahlisch F, Muhl D, Gerth CW, Hoyer C, Klosterkötter J, Hellmich M, Koethe D (2012). "Cannabidiol enhances anandamide signaling and alleviates psychotic symptoms of schizophrenia". Translational Psychiatry. 2 (3): e94–. doi:10.1038/tp.2012.15. PMC 3316151. PMID 22832859.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ plc, GW Pharmaceuticals. "GW Pharmaceuticals Announces Positive Proof of Concept Data in Schizophrenia". Retrieved 2015-09-15.
- ^ Crippa JA, Zuardi AW, Garrido GE, Wichert-Ana L, Guarnieri R, Ferrari L, Azevedo-Marques PM, Hallak JE, McGuire PK, Filho Busatto G (October 2003). "Effects of Cannabidiol (CBD) on Regional Cerebral Blood Flow". Neuropsychopharmacology. 29 (2): 417–426. doi:10.1038/sj.npp.1300340. PMID 14583744.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Malone DT, Jongejan D, Taylor DA (August 2009). "Cannabidiol reverses the reduction in social interaction produced by low dose Δ9-tetrahydrocannabinol in rats". Pharmacology Biochemistry and Behavior. 93 (2): 91–96. doi:10.1016/j.pbb.2009.04.010. PMID 19393686.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Bergamaschi MM, Queiroz RH, Chagas MH, de Oliveira DC, De Martinis BS, Kapczinski F, Quevedo J, Roesler R, Schröder N, Nardi AE, Martín-Santos R, Hallak JE, Zuardi AW, Crippa JA (May 2011). "Cannabidiol Reduces the Anxiety Induced by Simulated Public Speaking in Treatment-Naïve Social Phobia Patients". Neuropsychopharmacology. 36 (6): 1219–1226. doi:10.1038/npp.2011.6. PMC 3079847. PMID 21307846.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Crippa JA, Derenusson GN, Ferrari TB, Wichert-Ana L, Duran FL, Martin-Santos R, Simões MV, Bhattacharyya S, Fusar-Poli P, Atakan Z, Santos Filho A, Freitas-Ferrari MC, McGuire PK, Zuardi AW, Busatto GF, Hallak JE (January 2011). "Neural basis of anxiolytic effects of cannabidiol (CBD) in generalized social anxiety disorder: a preliminary report". J Psychopharmacol. 25 (1): 121–130. doi:10.1177/0269881110379283. PMID 20829306.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ ElBatsh MM, Assareh N, Marsden CA, Kendall DA (May 2012). "Anxiogenic-like effects of chronic cannabidiol administration in rats". Psychopharmacology. 221 (2): 239–247. doi:10.1007/s00213-011-2566-z. PMID 22083592.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Gururajan A (2012). "Comment on: "Anxiogenic-like effects of chronic cannabidiol administration in rats" (Elbatsh MM, Assareh N, Marsden CA, Kendall DA, Psychopharmacology 2012)". Psychopharmacology. 222 (4): 725–6, author reply 727. doi:10.1007/s00213-012-2780-3. PMID 22760485.
- ^ Réus GZ, Stringari RB, Ribeiro KF, Luft T, Abelaira HM, Fries GR, Aguiar BW, Kapczinski F, Hallak JE, Zuardi AW, Crippa JA, Quevedo J (2011). "Administration of cannabidiol and imipramine induces antidepressant-like effects in the forced swimming test and increases brain-derived neurotrophic factor levels in the rat amygdala". Acta Neuropsychiatrica. 23 (5): 241–248. doi:10.1111/j.1601-5215.2011.00579.x. PMID 25379896.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b Zanelati TV, Biojone C, Moreira FA, Guimarães FS, Joca SR (January 2010). "Antidepressant-like effects of cannabidiol in mice: possible involvement of 5-HT1A receptors". British Journal of Pharmacology. 159 (1): 122–8. doi:10.1111/j.1476-5381.2009.00521.x. PMC 2823358. PMID 20002102.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Réus, GZ; Stringari, RB; Ribeiro, KF; Luft, T; Abelaira, HM; Fries, GR; Aguiar, BW; Kapczinski, F; Hallak, JE; Zuardi, AW; Crippa JA; Quevedo, J (October 2011). "Administration of cannabidiol and imipramine induces antidepressant-like effects in the forced swimming test and increases brain-derived neurotrophic factor levels in the rat amygdala". Acta Neuropsychiatrica. 23 (5): 241–248. doi:10.1111/j.1601-5215.2011.00579.x. PMID 25379896.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ El-Alfy AT, Ivey K, Robinson K, Ahmed S, Radwan M, Slade D, Khan I, ElSohly M, Ross S (June 2010). "Antidepressant-like effect of Δ9-tetrahydrocannabinol and other cannabinoids isolated from Cannabis sativa L". Pharmacology Biochemistry and Behavior. 95 (4): 434–442. doi:10.1016/j.pbb.2010.03.004. PMC 2866040. PMID 20332000.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Romney, Lee (13 September 2012). "On the frontier of medical pot to treat boy's epilepsy". Los Angeles Times.
- ^ Good, Alastair (26 October 2010). "Growing marijuana that won't get you high". The Daily Telegraph. London.
- ^ Sidner, Sara (8 November 2012). "Medical marijuana without the high" (video). CNN.
An Israeli company has cultivated a new type of medical marijuana.
{{cite journal}}: Cite journal requires|journal=(help) - ^ Solon, Olivia (5 July 2012). "Medical Marijuana Without the High". Wired.comTemplate:Inconsistent citations
{{cite news}}: CS1 maint: postscript (link) - ^ Lubell, Maayan (3 July 2012). "What a drag, Israeli firm grows 'highless' marijuana". Reuters. Retrieved 31 Jan 2014.
- ^ Cohen, Ytzchak (11 September 2014). "Cannabis plant named 'avidekel'". USPTO. Retrieved 7 October 2014.
- ^ Fournier, G.; Beherec, O.; Bertucelli, S. (2003). "Intérêt du rapport Δ-9-THC / CBD dans le contrôle des cultures de chanvre industriel". Annales de Toxicologie Analytique. 15 (4): 250–259. doi:10.1051/ata/2003003.
- ^ Pertwee RG (2008). "The diverse CB1 and CB2 receptor pharmacology of three plant cannabinoids: Δ9-tetrahydrocannabinol, cannabidiol and Δ9-tetrahydrocannabivarin". British Journal of Pharmacology. 153 (2): 199–215. doi:10.1038/sj.bjp.0707442. PMC 2219532. PMID 17828291.
- ^ Hayakawa K, Mishima K, Hazekawa M, Sano K, Irie K, Orito K, Egawa T, Kitamura Y, Uchida N, Nishimura R, Egashira N, Iwasaki K, Fujiwara M (2008). "Cannabidiol potentiates pharmacological effects of Δ9-tetrahydrocannabinol via CB1 receptor-dependent mechanism". Brain Research. 1188: 157–164. doi:10.1016/j.brainres.2007.09.090. PMID 18021759.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Alchimia Blog, Cannabinoids and their medicinal properties
- ^ Ryberg E, Larsson N, Sjögren S, Hjorth S, Hermansson NO, Leonova J, Elebring T, Nilsson K, Drmota T, Greasley PJ (2007). "The orphan receptor GPR55 is a novel cannabinoid receptor". British Journal of Pharmacology. 152 (7): 1092–101. doi:10.1038/sj.bjp.0707460. PMC 2095107. PMID 17876302.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Russo EB, Burnett A, Hall B, Parker KK (August 2005). "Agonistic properties of cannabidiol at 5-HT1a receptors". Neurochemical Research. 30 (8): 1037–43. doi:10.1007/s11064-005-6978-1. PMID 16258853.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b Resstel LB, Tavares RF, Lisboa SF, Joca SR, Corrêa FM, Guimarães FS (January 2009). "5-HT1A receptors are involved in the cannabidiol-induced attenuation of behavioural and cardiovascular responses to acute restraint stress in rats". British Journal of Pharmacology. 156 (1): 181–8. doi:10.1111/j.1476-5381.2008.00046.x. PMC 2697769. PMID 19133999.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Campos AC, Guimarães FS (August 2008). "Involvement of 5HT1A receptors in the anxiolytic-like effects of cannabidiol injected into the dorsolateral periaqueductal gray of rats". Psychopharmacology. 199 (2): 223–30. doi:10.1007/s00213-008-1168-x. PMID 18446323.
- ^ Mishima K, Hayakawa K, Abe K, Ikeda T, Egashira N, Iwasaki K, Fujiwara M (May 2005). "Cannabidiol prevents cerebral infarction via a serotonergic 5-hydroxytryptamine1A receptor-dependent mechanism". Stroke; a Journal of Cerebral Circulation. 36 (5): 1077–82. doi:10.1161/01.STR.0000163083.59201.34. PMID 15845890.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Hayakawa K, Mishima K, Nozako M, Ogata A, Hazekawa M, Liu AX, Fujioka M, Abe K, Hasebe N, Egashira N, Iwasaki K, Fujiwara M (March 2007). "Repeated treatment with cannabidiol but not Delta9-tetrahydrocannabinol has a neuroprotective effect without the development of tolerance". Neuropharmacology. 52 (4): 1079–87. doi:10.1016/j.neuropharm.2006.11.005. PMID 17320118.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Kathmann M, Flau K, Redmer A, Tränkle C, Schlicker E (2006). "Cannabidiol is an allosteric modulator at mu- and delta-opioid receptors". Naunyn-Schmiedeberg's Archives of Pharmacology. 372 (5): 354–361. doi:10.1007/s00210-006-0033-x. PMID 16489449.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Campos, A. C.; Moreira, F. A.; Gomes, F. V.; Del Bel, E. A.; Guimaraes, F. S. (2012). "Multiple mechanisms involved in the large-spectrum therapeutic potential of cannabidiol in psychiatric disorders". Philosophical Transactions of the Royal Society B: Biological Sciences. 367 (1607): 3364–3378. doi:10.1098/rstb.2011.0389. ISSN 0962-8436.
- ^ Bornheim LM, Kim KY, Li J, Perotti BY, Benet LZ (August 1995). "Effect of cannabidiol pretreatment on the kinetics of tetrahydrocannabinol metabolites in mouse brain". Drug Metabolism and Disposition. 23 (8): 825–831. PMID 7493549.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Klein C, Karanges E, Spiro A, Wong A, Spencer J, Huynh T, Gunasekaran N, Karl T, Long LE, Huang XF, Liu K, Arnold JC, McGregor IS (November 2011). "Cannabidiol potentiates Δ⁹-tetrahydrocannabinol (THC) behavioural effects and alters THC pharmacokinetics during acute and chronic treatment in adolescent rats". Psychopharmacology. 218 (2): 443–457. doi:10.1007/s00213-011-2342-0. PMID 21667074.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Hunt CA, Jones RT, Herning RI, Bachman J (June 1981). "Evidence that Cannabidiol Does Not Significantly Alter the Pharmacokinetics of Tetrahydrocannabinol in Man". Journal of Pharmacokinetics and Biopharmaceutics. 9 (3): 245–260. doi:10.1007/BF01059266. PMID 6270295.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ United States Adopted Names Council: Statement on a nonproprietary name
- ^ "Fact Sheet — Sativex". Health Canada. Retrieved 16 May 2013.
- ^ GWPharma- Welcome
- ^ Jones PG, Falvello L, Kennard O, Sheldrick GM Mechoulam R (1977). "Cannabidiol". Acta Cryst. B33 (10): 3211–3214. doi:10.1107/S0567740877010577.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Mechoulam R, Ben-Zvi Z, Gaoni Y (1968). "Hashish—XIII On the nature of the beam test". Tetrahedron. 24 (16): 5615–5624. doi:10.1016/0040-4020(68)88159-1. PMID 5732891.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Gaoni Y, Mechoulam R (1966). "Hashish—VII The isomerization of cannabidiol to tetrahydrocannabinols". Tetrahedron. 22 (4): 1481–1488. doi:10.1016/S0040-4020(01)99446-3.
- ^ Petrzilka T, Haefliger W, Sikemeier C, Ohloff G, Eschenmoser A (1967). "Synthese und Chiralität des (−)-Cannabidiols". Helv. Chim. Acta. 50 (2): 719–723. doi:10.1002/hlca.19670500235. PMID 5587099.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Gaoni Y, Mechoulam R (1985). "Boron trifluoride etherate on alumuna — a modified Lewis acid reagent. An improved synthesis of cannabidiol". Tetrahedron Letters. 26 (8): 1083–1086. doi:10.1016/S0040-4039(00)98518-6.
- ^ Kobayashi Y, Takeuchi A, Wang YG (2006). "Synthesis of cannabidiols via alkenylation of cyclohexenyl monoacetate". Org. Lett. 8 (13): 2699–2702. doi:10.1021/ol060692h. PMID 16774235.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Marks MD, Tian L, Wenger JP, Omburo SN, Soto-Fuentes W, He J, Gang DR, Weiblen GD, Dixon RA (2009). "Identification of candidate genes affecting Δ9-tetrahydrocannabinol biosynthesis in Cannabis sativa". Journal of Experimental Botany. 60 (13): 3715–3726. doi:10.1093/jxb/erp210. PMC 2736886. PMID 19581347.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Controlled Drugs and Substances Act - Schedule II
- ^ a b c d §1308.11 Schedule I.
- ^ Controlled Substances by CSA Schedule - September 2014
- ^ a b [1], SCHEDULE AND DRUG CODES
- ^ Scheduling process at DEA - the example of cannabidiol
- ^ a b Title 21 US Code Controlled Substances Act, §802. Definitions.
- ^ a b Rules - 2013 > Establishment of Drug Codes for 26 Substances
- ^ Synthetic Drug Abuse Prevention Act of 2012 - GovTrack.us
- ^ Russo EB (August 2011). "Taming THC: potential cannabis synergy and phytocannabinoid-terpenoid entourage effects". Br. J. Pharmacol. (Review). 163 (7): 1344–64. doi:10.1111/j.1476-5381.2011.01238.x. PMC 3165946. PMID 21749363.
- ^ Definition of marijuana under the Controlled Substances Act.
- ^ Hemp Industries Assn., v. Drug Enforcement Admin., 9th Circuit Court of Appeals case involving industrial hemp.
- ^ Hemp, Many definitions of common terms associated with hemp, including the history of hemp use.
- ^ a b c US patent 6630507, Hampson, Aidan J.; Axelrod, Julius; Grimaldi, Maurizio, "Cannabinoids as antioxidants and neuroprotectants", issued 2003-10-07
- ^ "Federal Register | Prospective Grant of Exclusive License: Development of Cannabinoid(s) and Cannabidiol(s) Based Therapeutics To Treat Hepatic Encephalopathy in Humans". Federalregister.gov. November 17, 2011. Retrieved August 13, 2013.
- ^ "KannaLife Sciences, Inc. Signs Exclusive License Agreement With National Institutes Of Health Office Of Technology Transfer (NIH-OTT)". thestreet.com. Retrieved 2012-07-09.
- ^ "KannaLife in R&D Collaboration for Cannabinoid-Based Drugs". Genengnews.com. Retrieved 2013-04-04.
External links
- Project CBD Non-profit educational service dedicated to promoting and publicizing research into the medical utility of cannabidiol.
