Ethanol: Difference between revisions
No edit summary |
Diogenes2000 (talk | contribs) |
||
| Line 141: | Line 141: | ||
'''Alcohol and metronidazole''' |
'''Alcohol and metronidazole''' |
||
One of the most important drug/food interactions that should be noted is between alcohol and metronidazole. |
One of the most important drug/food interactions that should be noted is between alcohol and [[metronidazole]]. |
||
Metronidazole is an antibacterial agent that kills bacteria by damaging cellular DNA and hence cellular function.<ref name="cps">Repchinsky C (ed.) (2012). Compendium of pharmaceuticals and specialties, Ottawa: Canadian Pharmacists Association.{{full citation needed|date=February 2014}}</ref> Metronidazole is usually given to people who have diarrhea caused by ''[[Clostridium difficile]]'' bacteria. ''C. difficile'' is one of the most common microorganisms that cause diarrhea and can lead to complications such as colon inflammation and even more severely, death. |
Metronidazole is an antibacterial agent that kills bacteria by damaging cellular DNA and hence cellular function.<ref name="cps">Repchinsky C (ed.) (2012). Compendium of pharmaceuticals and specialties, Ottawa: Canadian Pharmacists Association.{{full citation needed|date=February 2014}}</ref> Metronidazole is usually given to people who have diarrhea caused by ''[[Clostridium difficile]]'' bacteria. ''C. difficile'' is one of the most common microorganisms that cause diarrhea and can lead to complications such as colon inflammation and even more severely, death. |
||
Revision as of 20:01, 14 November 2014
| |||
| |||
| Names | |||
|---|---|---|---|
| Systematic IUPAC name
Ethanol[1] | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ECHA InfoCard | 100.000.526 | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C2H6O | |||
| Molar mass | 46.069 g·mol−1 | ||
| Density | 0.789 g/cm3 (at 25°C) | ||
| Melting point | −114 °C (−173 °F; 159 K) | ||
| Boiling point | 78.37 °C (173.07 °F; 351.52 K) | ||
| Acidity (pKa) | 15.9 (H2O), 29.8 (DMSO)[2][3] | ||
| Hazards | |||
| Flash point | 16 °C (61 °F; 289 K) | ||
| 365 °C (689 °F; 638 K) | |||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose)
|
7060 mg/kg (oral, rat)[4] | ||
| Related compounds | |||
| Supplementary data page | |||
| Ethanol (data page) | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Commonly referred to simply as alcohol or spirits, ethanol /ˈɛθənɒl/ is also called ethyl alcohol, and drinking alcohol. It is the principal type of alcohol found in alcoholic beverages, produced by the fermentation of sugars by yeasts. It is a neurotoxic[6][7] psychoactive drug and one of the oldest recreational drugs used by humans. It can cause alcohol intoxication when consumed in sufficient quantity. Ethanol is used as a solvent, an antiseptic, a fuel and the active fluid in modern (post-mercury) thermometers. It is a volatile, flammable, colorless liquid with the structural formula CH
3CH
2OH, often abbreviated as C
2H
5OH or C
2H
6O.
Etymology
Ethanol is the systematic name defined by the International Union of Pure and Applied Chemistry (IUPAC) for a molecule with two carbon atoms (prefix "eth-"), having a single bond between them (suffix "-ane"), and an attached functional group-OH group (suffix "-ol").[1]
The prefix ethyl was coined in 1834 by the German chemist Justus Liebig.[8] Ethyl is a contraction of the French word ether (any substance that evaporated or sublimated readily at room temperature) and the Greek word ύλη (hyle, substance).[9]
The name ethanol was coined as a result of a resolution that was adopted at the International Conference on Chemical Nomenclature that was held in April 1892 in Geneva, Switzerland.[10]
The term "alcohol" now refers to a wider class of substances in chemistry nomenclature, but in common parlance it remains the name of ethanol. Ultimately a medieval loan from Arabic al-kuḥl,[11] use of alcohol in this sense is modern, introduced in the mid 18th century. Before that time, Middle Latin alcohol referred to "powdered ore of antimony; powdered cosmetic", by the later 17th century "any sublimated substance; distilled spirit" use for "the spirit of wine" (shortened from a full expression alcohol of wine) recorded 1753. The systematic use in chemistry dates to 1850.
Chemical formula
Ethanol is a 2-carbon alcohol. Its molecular formula is CH3CH2OH. An alternative notation is CH3–CH2–OH, which indicates that the carbon of a methyl group (CH3–) is attached to the carbon of a methylene group (–CH2–), which is attached to the oxygen of a hydroxyl group (–OH). It is a constitutional isomer of dimethyl ether. Ethanol is sometimes abbreviated as EtOH, using the common organic chemistry notation of representing the ethyl group (C2H5) with Et.
Natural occurrence
Ethanol is a byproduct of the metabolic process of yeast. As such, ethanol will be present in any yeast habitat. Ethanol can commonly be found in overripe fruit.[12] Ethanol produced by symbiotic yeast can be found in Bertam Palm blossoms. Although some animal species such as the Pentailed Treeshrew exhibit ethanol-seeking behaviors, most show no interest or avoidance of food sources containing ethanol.[13] Ethanol is also produced during the germination of many plants as a result of natural anerobiosis.[14] Ethanol has been detected in outer space, forming an icy coating around dust grains in interstellar clouds.[15] Minute quantity amounts (244 ppb) of endogenous ethanol and acetaldehyde were found in the exhaled breath of healthy volunteers.[16]
Pharmacology
Activity profile
Ethanol is known to possess the following direct pharmacodynamic actions:[17]
- GABAA receptor positive allosteric modulator (primarily of δ subunit-containing receptors)
- NMDA receptor negative allosteric modulator
- Glycine receptor positive and negative allosteric modulator
- 5-HT3 receptor positive allosteric modulator
- nACh receptor positive and negative allosteric modulator
- L-type calcium channel blocker
- GIRK channel opener
- Glycine reuptake inhibitor[18]
- Adenosine reuptake inhibitor[19]
Some of its actions on ligand-gated ion channels, specifically the nACh receptors and the glycine receptor, are dose-dependent, with potentiation or inhibition occurring dependent on ethanol concentration. This is because ethanol's effects on these channels are a summation of positive and negative allosteric modulatory actions.[17]
Properties
The removal of ethanol from the human body, through oxidation by alcohol dehydrogenase in the liver, is limited. Hence, the removal of a large concentration of alcohol from blood may follow zero-order kinetics. This means that alcohol leaves the body at a constant rate, rather than having an elimination half-life.[20]
The rate-limiting steps for one substance may be in common with other substances. As a result, the blood alcohol concentration can be used to modify the rate of metabolism of methanol and ethylene glycol. Methanol itself is not highly toxic, but its metabolites formaldehyde and formic acid are; therefore, to reduce the rate of production and concentration of these harmful metabolites, ethanol can be ingested.[citation needed] Ethylene glycol poisoning can be treated in the same way.
Pure ethanol will irritate the skin and eyes.[21] Nausea, vomiting and intoxication are symptoms of ingestion. Long-term use by ingestion can result in serious liver damage.[22] Atmospheric concentrations above one in a thousand are above the European Union Occupational exposure limits.[22]
Short-term
| BAC (g/L) | BAC (% v/v) |
Symptoms[23] |
|---|---|---|
| 0.5 | 0.05% | Euphoria, talkativeness, relaxation |
| 1 | 0.1 % | Central nervous system depression, nausea, possible vomiting, impaired motor and sensory function, impaired cognition |
| >1.4 | >0.14% | Decreased blood flow to brain |
| 3 | 0.3% | Stupefaction, possible unconsciousness |
| 4 | 0.4% | Possible death |
| >5.5 | >0.55% | Death |
Effects on the central nervous system
Ethanol is a central nervous system depressant and has significant psychoactive effects in sublethal doses; for specifics, see "Effects of alcohol on the body by dose". Based on its abilities to change the human consciousness, ethanol is considered a psychoactive drug.[24] Death from ethanol consumption is possible when blood alcohol level reaches 0.4%. A blood level of 0.5% or more is commonly fatal. Levels of even less than 0.1% can cause intoxication, with unconsciousness often occurring at 0.3–0.4%.[25]
The amount of ethanol in the body is typically quantified by blood alcohol content (BAC), which is here taken as weight of ethanol per unit volume of blood. The table at the right summarizes the symptoms of ethanol consumption. Small doses of ethanol, in general, produce euphoria and relaxation; people experiencing these symptoms tend to become talkative and less inhibited, and may exhibit poor judgment. At higher dosages (BAC > 1 g/L), ethanol acts as a central nervous system depressant, producing at progressively higher dosages, impaired sensory and motor function, slowed cognition, stupefaction, unconsciousness, and possible death.
Ethanol acts in the central nervous system primarily by binding to the GABAA receptor, increasing the effects of the inhibitory neurotransmitter GABA (i.e., it is a positive allosteric modulator).[26]
Prolonged heavy consumption of alcohol can cause significant permanent damage to the brain and other organs. See Alcohol consumption and health.
According to the US National Highway Traffic Safety Administration, in 2002 about "41% of people fatally injured in traffic crashes were in alcohol related crashes".[27] The risk of a fatal car accident increases exponentially with the level of alcohol in the driver's blood.[28] Most drunk driving laws governing the acceptable levels in the blood while driving or operating heavy machinery set typical upper limits of blood alcohol content (BAC) between 0.02% and 0.08%.[citation needed]
Discontinuing consumption of alcohol after several years of heavy drinking can also be fatal. Alcohol withdrawal can cause anxiety, autonomic dysfunction, seizures, and hallucinations. Delirium tremens is a condition that requires people with a long history of heavy drinking to undertake an alcohol detoxification regimen.
The reinforcing effects of alcohol consumption are also mediated by acetaldehyde generated by catalase and other oxidizing enzymes such as cytochrome P-4502E1 in the brain.[29] Although acetaldehyde has been associated with some of the adverse and toxic effects of ethanol, it appears to play a central role in the activation of the mesolimbic dopamine system.[30]
Effects on metabolism
Ethanol within the human body is converted into acetaldehyde by alcohol dehydrogenase and then into the acetyl in acetyl CoA by acetaldehyde dehydrogenase. Acetyl CoA is the final product of both carbohydrate and fat metabolism, where the acetyl can be further used to produce energy or for biosynthesis. As such, ethanol can be compared to an energy-bearing macronutrient, yielding approximately 7 kcal per gram consumed.[31] However, the product of the first step of this breakdown, acetaldehyde,[32] is more toxic than ethanol. Acetaldehyde is linked to most of the clinical effects of alcohol. It has been shown to increase the risk of developing cirrhosis of the liver[33] and multiple forms of cancer.
During the metabolism of alcohol via the respective dehydrogenases, NAD (Nicotinamide adenine dinucleotide) is converted into reduced NAD. Normally, NAD is used to metabolise fats in the liver, and as such alcohol competes with these fats for the use of NAD. Prolonged exposure to alcohol means that fats accumulate in the liver, leading to the term 'fatty liver'. Continued consumption (such as in alcoholism) then leads to cell death in the hepatocytes as the fat stores reduce the function of the cell to the point of death. These cells are then replaced with scar tissue, leading to the condition called cirrhosis.
Drug interactions
Ethanol can intensify the sedation caused by other central nervous system depressant drugs such as barbiturates, benzodiazepines, opioids, non-benzodiazepines (such as Zolpidem and Zopiclone), antipsychotics, sedative antihistamines, and antidepressants.[25] It interacts with cocaine in vivo to produce cocaethylene, another psychoactive substance.[34] Ethanol enhances the bioavailability of methylphenidate (elevated plasma d-MPH).[35] In combination with cannabis, ethanol increases plasma THC levels, which suggets that ethanol may increase the absorption of THC.[36]
Alcohol and metronidazole
One of the most important drug/food interactions that should be noted is between alcohol and metronidazole.
Metronidazole is an antibacterial agent that kills bacteria by damaging cellular DNA and hence cellular function.[37] Metronidazole is usually given to people who have diarrhea caused by Clostridium difficile bacteria. C. difficile is one of the most common microorganisms that cause diarrhea and can lead to complications such as colon inflammation and even more severely, death.
Patients who are taking metronidazole are strongly advised to avoid alcohol, even after 1 hour after the last dose. The reason is that alcohol and metronidazole can lead to side effects such as flushing, headache, nausea, vomiting, abdominal cramps, and sweating.[38][39][39] These symptoms are often called the disulfiram-like reaction. The proposed mechanism of action for this interaction is that metronidazole can bind to an enzyme that normally metabolizes alcohol. Binding to this enzyme may impair the liver's ability to process alcohol for proper excretion.[40]
Alcohol and digestion
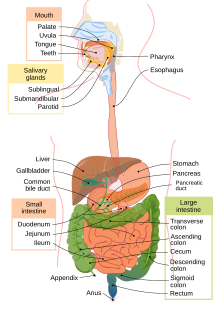
A part of ethyl alcohol is hydrophobic. This hydrophobic or lipophilic end can diffuse across cells that line the stomach wall. In fact, alcohol is one of the rare substances that can be absorbed in the stomach. Most food substances are absorbed in the small intestine. However, even though alcohol can be absorbed in the stomach, it is mostly absorbed in the small intestine because the small intestine has a large surface area that promotes absorption. Once alcohol is absorbed in the small intestine, it delays the release of stomach contents from emptying into the small intestine. Thus, alcohol can delay the rate of absorption of nutrients.[41] After absorption, alcohol reaches the liver where it is metabolized.
Breathalyzers
Alcohol that is not processed by the liver goes to the heart. The liver can process only a certain amount of alcohol per unit time. Thus, when a person drinks too much alcohol, more alcohol can reach the heart. In the heart, alcohol reduces the force of heart contractions. Consequently, the heart will pump less blood, lowering overall body blood pressure.[42] Also, blood that reaches the heart goes to the lungs to replenish blood's oxygen concentration. It is at this stage that a person can breathe out traces of alcohol.[42] This is the underlying principle of the alcohol breath testing (or breathalyzers) to determine if a driver has been drinking and driving.[43]
From the lungs, blood returns to the heart and will be distributed throughout the body. Interestingly, alcohol increases levels of high-density lipoproteins(HDLs), which carry cholesterol.[42] Alcohol is known to make blood less likely to clot, reducing risk of heart attack and stroke. This could be the reason why alcohol could produce health benefits when consumed in moderate amounts.[44] Also, alcohol dilates blood vessels. Consequently, a person will feel warmer, and their face turns flush and pink.[42]
Loss of balance
When alcohol reaches the brain, it has the ability to delay signals that are sent between nerve cells that control balance, thinking and movement.[42]
Frequent urination
Moreover, alcohol can affect the brain's ability to produce antidiuretic hormones. These hormones are responsible for controlling the amount of urine that is produced. Alcohol prevents the body from reabsorbing water, and consequently a person who recently drank alcohol will urinate frequently.[42]
Alcohol and gastrointestinal diseases

Alcohol stimulates gastric juice production, even when food is not present. In other words, when a person drinks alcohol, the alcohol will stimulate stomach's acidic secretions that are intended to digest protein molecules. Consequently, the acidity has potential to harm the inner lining of the stomach. Normally, the stomach lining is protected by a mucus layer that prevents any acids from reaching the stomach cells.
However, in patients who have a peptic ulcer disease (PUD), this mucus layer is broken down. PUD is commonly associated with a bacteria H. pylori. H. pylori secretes a toxin that weakens the mucosal wall. As a result, acid and protein enzymes penetrate the weakened barrier. Because alcohol stimulates a person's stomach to secrete acid, a person with PUD should avoid drinking alcohol on an empty stomach. Drinking alcohol would cause more acid release to damage the weakened stomach wall.[45] Complications of this disease could include a burning pain in the abdomen, bloating and in severe cases, the presence of dark black stools indicate internal bleeding.[46] A person who drinks alcohol regularly is strongly advised to reduce their intake to prevent PUD aggravation.[46]
Magnitude of effects
Some individuals have less effective forms of one or both of the metabolizing enzymes, and can experience more severe symptoms from ethanol consumption than others. However, those having acquired alcohol tolerance have a greater quantity of these enzymes, and metabolize ethanol more rapidly.[47]
Long-term
Birth defects
Ethanol is classified as a teratogen. See fetal alcohol syndrome and fetal alcohol spectrum disorder.
Cancer
IARC list ethanol in alcoholic beverages as Group 1 carcinogens and arguments "There is sufficient evidence for the carcinogenicity of acetaldehyde (the major metabolite of ethanol) in experimental animals.".[48]
Other effects
Frequent drinking of alcoholic beverages has been shown to be a major contributing factor in cases of elevated blood levels of triglycerides.[49]
Ethanol is also widely used, clinically and over the counter, as an antitussive agent.[50]
History
The fermentation of sugar into ethanol is one of the earliest biotechnologies employed by humans. The intoxicating effects of ethanol consumption have been known since ancient times. Ethanol has been used by humans since prehistory as the intoxicating ingredient of alcoholic beverages. Dried residue on 9,000-year-old pottery found in China suggests that Neolithic people consumed alcoholic beverages.[51]
Although distillation was well known by the early Greeks and Arabs, the first recorded production of alcohol from distilled wine was by the School of Salerno alchemists in the 12th century.[52] The first to mention absolute alcohol, in contrast with alcohol-water mixtures, was Raymond Lull.[52]
In 1796, German-Russian chemist Johann Tobias Lowitz obtained pure ethanol by mixing partially purified ethanol (the alcohol-water azeotrope) with an excess of anhydrous alkali and then distilling the mixture over low heat.[53] French chemist Antoine Lavoisier described ethanol as a compound of carbon, hydrogen, and oxygen, and in 1807 Nicolas-Théodore de Saussure determined ethanol's chemical formula.[54][55] Fifty years later, Archibald Scott Couper published the structural formula of ethanol. It was one of the first structural formulas determined.[56]
Ethanol was first prepared synthetically in 1825 by Michael Faraday. He found that sulfuric acid could absorb large volumes of coal gas.[57] He gave the resulting solution to Henry Hennell, a British chemist, who found in 1826 that it contained "sulphovinic acid" (ethyl hydrogen sulfate).[58] In 1828, Hennell and the French chemist Georges-Simon Sérullas independently discovered that sulphovinic acid could be decomposed into ethanol.[59][60] Thus, in 1825 Faraday had unwittingly discovered that ethanol could be produced from ethylene (a component of coal gas) by acid-catalyzed hydration, a process similar to current industrial ethanol synthesis.[61]
Ethanol was used as lamp fuel in the United States as early as 1840, but a tax levied on industrial alcohol during the Civil War made this use uneconomical. The tax was repealed in 1906.[62] Use as an automotive fuel dates back to 1908, with the Ford Model T able to run on petrol(gasoline) or ethanol.[63] It remains a common fuel for spirit lamps.
Ethanol intended for industrial use is often produced from ethylene.[64] Ethanol has widespread use as a solvent of substances intended for human contact or consumption, including scents, flavorings, colorings, and medicines. In chemistry, it is both a solvent and a feedstock for the synthesis of other products. It has a long history as a fuel for heat and light, and more recently as a fuel for internal combustion engines.
Properties
Physical properties

Ethanol is a volatile, colorless liquid that has a slight odor. It burns with a smokeless blue flame that is not always visible in normal light.
The physical properties of ethanol stem primarily from the presence of its hydroxyl group and the shortness of its carbon chain. Ethanol's hydroxyl group is able to participate in hydrogen bonding, rendering it more viscous and less volatile than less polar organic compounds of similar molecular weight, such as propane.
Ethanol is slightly more refractive than water, having a refractive index of 1.36242 (at λ=589.3 nm and 18.35 °C or 65.03 °F).[65]
The triple point for ethanol is 150 K at a pressure of 4.3 × 10−4 Pa.[66]
Solvent properties
Ethanol is a versatile solvent, miscible with water and with many organic solvents, including acetic acid, acetone, benzene, carbon tetrachloride, chloroform, diethyl ether, ethylene glycol, glycerol, nitromethane, pyridine, and toluene.[65][67] It is also miscible with light aliphatic hydrocarbons, such as pentane and hexane, and with aliphatic chlorides such as trichloroethane and tetrachloroethylene.[67]
Ethanol's miscibility with water contrasts with the immiscibility of longer-chain alcohols (five or more carbon atoms), whose water miscibility decreases sharply as the number of carbons increases.[68] The miscibility of ethanol with alkanes is limited to alkanes up to undecane: mixtures with dodecane and higher alkanes show a miscibility gap below a certain temperature (about 13 °C for dodecane[69]). The miscibility gap tends to get wider with higher alkanes and the temperature for complete miscibility increases.
Ethanol-water mixtures have less volume than the sum of their individual components at the given fractions. Mixing equal volumes of ethanol and water results in only 1.92 volumes of mixture.[65][70] Mixing ethanol and water is exothermic, with up to 777 J/mol[71] being released at 298 K.
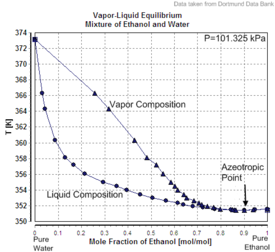
Mixtures of ethanol and water form an azeotrope at about 89 mole-% ethanol and 11 mole-% water[72] or a mixture of about 96 volume percent ethanol and 4% water at normal pressure and T = 351 K. This azeotropic composition is strongly temperature- and pressure-dependent and vanishes at temperatures below 303 K.[73]

Hydrogen bonding causes pure ethanol to be hygroscopic to the extent that it readily absorbs water from the air. The polar nature of the hydroxyl group causes ethanol to dissolve many ionic compounds, notably sodium and potassium hydroxides, magnesium chloride, calcium chloride, ammonium chloride, ammonium bromide, and sodium bromide.[67] Sodium and potassium chlorides are slightly soluble in ethanol.[67] Because the ethanol molecule also has a nonpolar end, it will also dissolve nonpolar substances, including most essential oils[74] and numerous flavoring, coloring, and medicinal agents.
The addition of even a few percent of ethanol to water sharply reduces the surface tension of water. This property partially explains the "tears of wine" phenomenon. When wine is swirled in a glass, ethanol evaporates quickly from the thin film of wine on the wall of the glass. As the wine's ethanol content decreases, its surface tension increases and the thin film "beads up" and runs down the glass in channels rather than as a smooth sheet.
Flammability
An ethanol-water solution that contains 40% ABV (alcohol by volume) will catch fire if heated to about 26 °C (79 °F) and if an ignition source is applied to it. This is called its flash point.[75] The flash point of pure ethanol is 16.60 °C (61.88 °F), less than average room temperature.[citation needed]
The flash points of ethanol concentrations from 10% ABV to 96% ABV are shown below:[76]
- 10% — 49 °C (120 °F)
- 20% — 36 °C (97 °F)
- 30% — 29 °C (84 °F)
- 40% — 26 °C (79 °F)
- 50% — 24 °C (75 °F)
- 60% — 22 °C (72 °F)
- 70% — 21 °C (70 °F)
- 80% — 20 °C (68 °F)
- 90% — 17 °C (63 °F)
- 96% — 17 °C (63 °F)
Alcoholic beverages that have a low concentration of ethanol will burn if sufficiently heated and an ignition source (such as an electric spark or a match) is applied to them. For example, the flash point of ordinary wine containing 12.5% ethanol is about 52 °C (126 °F).[77]
Dishes using burning alcohol for culinary effects are called Flambé.
Production

Ethanol is produced both as a petrochemical, through the hydration of ethylene and, via biological processes, by fermenting sugars with yeast.[78] Which process is more economical depends on prevailing prices of petroleum and grain feed stocks.
Ethylene hydration
Ethanol for use as an industrial feedstock or solvent (sometimes referred to as synthetic ethanol) is made from petrochemical feed stocks, primarily by the acid-catalyzed hydration of ethylene, represented by the chemical equation
- C
2H
4 + H
2O → CH
3CH
2OH
The catalyst is most commonly phosphoric acid,[79][80] adsorbed onto a porous support such as silica gel or diatomaceous earth. This catalyst was first used for large-scale ethanol production by the Shell Oil Company in 1947.[81] The reaction is carried out with an excess of high pressure steam at 300 °C (572 °F). In the U.S., this process was used on an industrial scale by Union Carbide Corporation and others, but now only LyondellBasell uses it commercially.
In an older process, first practiced on the industrial scale in 1930 by Union Carbide,[82] but now almost entirely obsolete, ethylene was hydrated indirectly by reacting it with concentrated sulfuric acid to produce ethyl sulfate, which was hydrolyzed to yield ethanol and regenerate the sulfuric acid:[83]
- C
2H
4 + H
2SO
4 → CH
3CH
2SO
4H - CH
3CH
2SO
4H + H
2O → CH
3CH
2OH + H
2SO
4
Fermentation
Ethanol for use in alcoholic beverages, and the vast majority of ethanol for use as fuel,[84] is produced by fermentation. When certain species of yeast (e.g., Saccharomyces cerevisiae) metabolize sugar in reduced-oxygen conditions they produce ethanol and carbon dioxide. The chemical equations below summarize the conversion:
Fermentation is the process of culturing yeast under favorable thermal conditions to produce alcohol. This process is carried out at around 35–40 °C (95–104 °F). Toxicity of ethanol to yeast limits the ethanol concentration obtainable by brewing; higher concentrations, therefore, are usually obtained by fortification or distillation. The most ethanol-tolerant strains of yeast can survive up to approximately 18% ethanol by volume (Red Star Pasteur Champagne wine yeast, Lalvin EC-1118 wine yeast) and 20% or greater using "Turbo Yeast" as sold for spirit and fuel distillation.
To produce ethanol from starchy materials such as cereal grains, the starch must first be converted into sugars. In brewing beer, this has traditionally been accomplished by allowing the grain to germinate, or malt, which produces the enzyme amylase. When the malted grain is mashed, the amylase converts the remaining starches into sugars. For fuel ethanol, the hydrolysis of starch into glucose can be accomplished more rapidly by treatment with dilute sulfuric acid, fungally produced amylase, or some combination of the two.[85]
Cellulose
Sugars for ethanol fermentation can be obtained from cellulose.[86][87] Until recently, however, the cost of the cellulase enzymes capable of hydrolyzing cellulose has been prohibitive. The Canadian firm Iogen brought the first cellulose-based ethanol plant on-stream in 2004.[88] Its primary consumer so far has been the Canadian government, which, along with the United States Department of Energy, has invested heavily in the commercialization of cellulosic ethanol. Deployment of this technology could turn a number of cellulose-containing agricultural by-products, such as corncobs, straw, and sawdust, into renewable energy resources. Other enzyme companies are developing genetically engineered fungi that produce large volumes of cellulase, xylanase, and hemicellulase enzymes. These would convert agricultural residues such as corn stover, wheat straw, and sugar cane bagasse and energy crops such as switchgrass into fermentable sugars.[89]
Cellulose-bearing materials typically contain other polysaccharides, including hemicellulose. Hydrolysis of hemicellulose gives mostly five-carbon sugars such as xylose. S. cerevisiae, the yeast most commonly used for ethanol production, cannot metabolize xylose. Other yeasts and bacteria are under investigation to ferment xylose and other pentoses into ethanol.
Hydrocarbon
A process developed and marketed by Celanese Corporation under the name TCX Technology uses hydrocarbons such as natural gas or coal for ethanol production rather than using fermented crops such as corn or sugarcane.[90]
Testing


Breweries and biofuel plants employ two methods for measuring ethanol concentration. Infrared ethanol sensors measure the vibrational frequency of dissolved ethanol using the CH band at 2900 cm−1. This method uses a relatively inexpensive solid state sensor that compares the CH band with a reference band to calculate the ethanol content. The calculation makes use of the Beer-Lambert law. Alternatively, by measuring the density of the starting material and the density of the product, using a hydrometer, the change in specific gravity during fermentation indicates the alcohol content. This inexpensive and indirect method has a long history in the beer brewing industry.
Research
Research on ethanol production includes working from alternative source materials, novel catalysts and new chemical processes.
Purification
Distillation
Ethylene hydration or brewing produces an ethanol–water mixture. For most industrial and fuel uses, the ethanol must be purified. Fractional distillation can concentrate ethanol to 95.6% by volume (89.5 mole%). This mixture is an azeotrope with a boiling point of 78.1 °C (172.6 °F), and cannot be further purified by distillation. Addition of an entraining agent, such as benzene, cyclohexane, or heptane, allows a new ternary azeotrope comprising the ethanol, water, and the entraining agent to be formed. This lower-boiling ternary azeotrope is removed preferentially, leading to water-free ethanol.[80]
At pressures less than atmospheric pressure, the composition of the ethanol-water azeotrope shifts to more ethanol-rich mixtures, and at pressures less than 70 torr (9.333 kPa), there is no azeotrope, and it is possible to distill absolute ethanol from an ethanol-water mixture. While vacuum distillation of ethanol is not presently economical, pressure-swing distillation is a topic of current research. In this technique, a reduced-pressure distillation first yields an ethanol-water mixture of more than 95.6% ethanol. Then, fractional distillation of this mixture at atmospheric pressure distills off the 95.6% azeotrope, leaving anhydrous ethanol at the bottom.[citation needed]
Molecular sieves and desiccants
Molecular sieves can be used to selectively absorb the water from the 95.6% ethanol solution. Synthetic zeolite in pellet form can be used, as well as a variety of plant-derived absorbents, including cornmeal, straw, and sawdust. The zeolite bed can be regenerated essentially an unlimited number of times by drying it with a blast of hot carbon dioxide. Cornmeal and other plant-derived absorbents cannot readily be regenerated, but where ethanol is made from grain, they are often available at low cost. Absolute ethanol produced this way has no residual benzene, and can be used to fortify port and sherry in traditional winery operations.
Apart from distillation, ethanol may be dried by addition of a desiccant, such as molecular sieves, cellulose, and cornmeal. The desiccants can be dried and reused.[80]
Membranes and reverse osmosis
Membranes can also be used to separate ethanol and water. Membrane-based separations are not subject to the limitations water-ethanol azeotrope because separation is not based on vapor-liquid equilibria. Membranes are often used in the so-called hybrid membrane distillation process. This process uses a pre-concentration distillation column as first separating step. The further separation is then accomplished with a membrane operated either in vapor permeation or pervaporation mode. Vapor permeation uses a vapor membrane feed and pervaporation uses a liquid membrane feed.
Other techniques
A variety of other techniques have been discussed, including the following:[80]
- Liquid-liquid extraction of ethanol from an aqueous solution;
- Extraction of ethanol from grain mash by supercritical carbon dioxide;
- Pervaporation;
- Pressure swing adsorption.[91]
Grades of ethanol
Ethanol is available in a range of purities that result from its production or, in the case of denatured alcohol, are introduced intentionally.
Denatured alcohol
Pure ethanol and alcoholic beverages are heavily taxed as psychoactive drugs, but ethanol has many uses that do not involve consumption by humans. To relieve the tax burden on these uses, most jurisdictions waive the tax when an agent has been added to the ethanol to render it unfit to drink. These include bittering agents such as denatonium benzoate and toxins such as methanol, naphtha, and pyridine. Products of this kind are called denatured alcohol.[92][93]
Absolute alcohol
Absolute or anhydrous alcohol refers to ethanol with a low water content. There are various grades with maximum water contents ranging from 1% to a few parts per million (ppm) levels. Absolute alcohol is not intended for human consumption. If azeotropic distillation is used to remove water, it will contain trace amounts of the material separation agent (e.g. benzene).[94] Absolute ethanol is used as a solvent for laboratory and industrial applications, where water will react with other chemicals, and as fuel alcohol. Spectroscopic ethanol is an absolute ethanol with a low absorbance in ultraviolet and visible light, fit for use as a solvent in ultraviolet-visible spectroscopy.[95]
Pure ethanol is classed as 200 proof in the U.S., equivalent to 175 degrees proof in the UK system.[96]
Rectified spirits
Rectified spirit, an azeotropic composition of 96% ethanol containing 4% water, is used instead of anhydrous ethanol for various purposes. Wine spirits are about 94% ethanol (188 proof). The impurities are different from those in 95% (190 proof) laboratory ethanol.[97]
Reactions
Ethanol is classified as a primary alcohol, meaning that the carbon its hydroxyl group attaches to has at least two hydrogen atoms attached to it as well. Many ethanol reactions occur at its hydroxyl group.
Ester formation
In the presence of acid catalysts, ethanol reacts with carboxylic acids to produce ethyl esters and water:
- RCOOH + HOCH2CH3 → RCOOCH2CH3 + H2O
This reaction, which is conducted on large scale industrially, requires the removal of the water from the reaction mixture as it is formed. Esters react in the presence of an acid or base to give back the alcohol and a salt. This reaction is known as saponification because it is used in the preparation of soap. Ethanol can also form esters with inorganic acids. Diethyl sulfate and triethyl phosphate are prepared by treating ethanol with sulfur trioxide and phosphorus pentoxide respectively. Diethyl sulfate is a useful ethylating agent in organic synthesis. Ethyl nitrite, prepared from the reaction of ethanol with sodium nitrite and sulfuric acid, was formerly used as a diuretic.
Dehydration
Strong acid desiccants cause the partial dehydration of ethanol to form diethyl ether and other byproducts. If the dehydration temperature exceeds around 160 °C (320 °F), full dehydration will occur and ethylene will be the main product.
- 2 CH3CH2OH → CH3CH2OCH2CH3 + H2O (ca. 120 °C)
- CH3CH2OH → H2C=CH2 + H2O (above 160 °C)
Combustion
Complete combustion of ethanol forms carbon dioxide and water:
- C2H5OH (l) + 3 O2 (g) → 2 CO2 (g) + 3 H2O (liq); −ΔHc = 1371 kJ/mol[98] = 29.8 kJ/g = 327 kcal/mol = 7.1 kcal/g
- C2H5OH (l) + 3 O2 (g) → 2 CO2 (g) + 3 H2O (g); −ΔHc = 1236 kJ/mol = 26.8 kJ/g = 295.4 kcal/mol = 6.41 kcal/g[99]
Specific heat = 2.44 kJ/(kg·K)
Acid-base chemistry
Ethanol is a neutral molecule and the pH of a solution of ethanol in water is nearly 7.00. Ethanol can be quantitatively converted to its conjugate base, the ethoxide ion (CH3CH2O−), by reaction with an alkali metal such as sodium:[68]
- 2 CH3CH2OH + 2 Na → 2 CH3CH2ONa + H2
or a very strong base such as sodium hydride:
- CH3CH2OH + NaH → CH3CH2ONa + H2
The acidity of water and ethanol are nearly the same, as indicated by their pKa of 15.7 and 16 respectively. Thus, sodium ethoxide and sodium hydroxide exist in an equilbrium that is closely balanced:
- CH3CH2OH + NaOH ⇌ CH3CH2ONa + H2O
Halogenation
Ethanol is not used industrially as a precursor to ethyl halides, but the reactions are illustrative. Ethanol reacts with hydrogen halides to produce ethyl halides such as ethyl chloride and ethyl bromide via an SN2 reaction:
- CH3CH2OH + HCl → CH3CH2Cl + H2O
These reactions require a catalyst such as zinc chloride.[83] HBr requires refluxing with a sulfuric acid catalyst.[83] Ethyl halides can, in principle, also be produced by treating ethanol with more specialized halogenating agents, such as thionyl chloride or phosphorus tribromide.[68][83]
- CH3CH2OH + SOCl2 → CH3CH2Cl + SO2 + HCl
Upon treatment with halogens in the presence of base, ethanol gives the corresponding haloform (CHX3, where X = Cl, Br, I). This conversion is called the haloform reaction.[100] " An intermediate in the reaction with chlorine is the aldehyde called chloral:
- 4 Cl2 + CH3CH2OH → CCl3CHO + 5 HCl
Oxidation
Ethanol can be oxidized to acetaldehyde and further oxidized to acetic acid, depending on the reagents and conditions.[83] This oxidation is of no importance industrially, but in the human body, these oxidation reactions are catalyzed by the enzyme liver alcohol dehydrogenase. The oxidation product of ethanol, acetic acid, is a nutrient for humans, being a precursor to acetyl CoA, where the acetyl group can be spent as energy or used for biosynthesis.
Other uses
Motor fuel
| Energy content of some fuels compared with ethanol:[101] | |||
|---|---|---|---|
| Fuel type | MJ/L | MJ/kg | Research octane number |
| Dry wood (20% moisture) | ~19.5 | ||
| Methanol | 17.9 | 19.9 | 108.7[102] |
| Ethanol | 21.2[103] | 26.8[103] | 108.6[102] |
| E85 (85% ethanol, 15% gasoline) |
25.2 | 33.2 | 105 |
| Liquefied natural gas | 25.3 | ~55 | |
| Autogas (LPG) (60% propane + 40% butane) |
26.8 | 50. | |
| Aviation gasoline (high-octane gasoline, not jet fuel) |
33.5 | 46.8 | 100/130 (lean/rich) |
| Gasohol (90% gasoline + 10% ethanol) |
33.7 | 47.1 | 93/94 |
| Regular gasoline/petrol | 34.8 | 44.4[104] | min. 91 |
| Premium gasoline/petrol | max. 104 | ||
| Diesel | 38.6 | 45.4 | 25 |
| Charcoal, extruded | 50 | 23 | |
The largest single use of ethanol is as a motor fuel and fuel additive. More than any other major country, Brazil relies on ethanol as a motor fuel. Gasoline sold in Brazil contains at least 25% anhydrous ethanol. Hydrous ethanol (about 95% ethanol and 5% water) can be used as fuel in more than 90% of new cars sold in the country. Brazilian ethanol is produced from sugar cane and noted for high carbon sequestration.[105] The US uses Gasohol (max 10% ethanol) and E85 (85% ethanol) ethanol/gasoline mixtures.

Ethanol may also be utilized as a rocket fuel, and is currently in lightweight rocket-powered racing aircraft.[106]
Australian law limits the use of pure ethanol sourced from sugarcane waste to up to 10% in automobiles. It has been recommended that older cars (and vintage cars designed to use a slower burning fuel) have their valves upgraded or replaced.[107]
According to an industry advocacy group for promoting ethanol called the American Coalition for Ethanol, ethanol as a fuel reduces harmful tailpipe emissions of carbon monoxide, particulate matter, oxides of nitrogen, and other ozone-forming pollutants.[108] Argonne National Laboratory analyzed the greenhouse gas emissions of many different engine and fuel combinations. Comparing ethanol blends with gasoline alone, they showed reductions of 8% with the biodiesel/petrodiesel blend known as B20, 17% with the conventional E85 ethanol blend, and that using cellulosic ethanol lowers emissions 64%.[109]
Ethanol combustion in an internal combustion engine yields many of the products of incomplete combustion produced by gasoline and significantly larger amounts of formaldehyde and related species such as acetaldehyde.[110] This leads to a significantly larger photochemical reactivity that generates much more ground level ozone.[111] These data have been assembled into The Clean Fuels Report comparison of fuel emissions[112] and show that ethanol exhaust generates 2.14 times as much ozone as does gasoline exhaust.[citation needed] When this is added into the custom Localised Pollution Index (LPI) of The Clean Fuels Report the local pollution (pollution that contributes to smog) is 1.7 on a scale where gasoline is 1.0 and higher numbers signify greater pollution.[citation needed] The California Air Resources Board formalized this issue in 2008 by recognizing control standards for formaldehydes as an emissions control group, much like the conventional NOx and Reactive Organic Gases (ROGs).[113]

World production of ethanol in 2006 was 51 gigalitres (1.3×1010 US gal), with 69% of the world supply coming from Brazil and the United States.[114] More than 20% of Brazilian cars are able to use 100% ethanol as fuel, which includes ethanol-only engines and flex-fuel engines.[115] Flex-fuel engines in Brazil are able to work with all ethanol, all gasoline or any mixture of both. In the US flex-fuel vehicles can run on 0% to 85% ethanol (15% gasoline) since higher ethanol blends are not yet allowed or efficient. Brazil supports this population of ethanol-burning automobiles with large national infrastructure that produces ethanol from domestically grown sugar cane. Sugar cane not only has a greater concentration of sucrose than corn (by about 30%), but is also much easier to extract. The bagasse generated by the process is not wasted, but is used in power plants to produce electricity.[citation needed]

The United States fuel ethanol industry is based largely on corn. According to the Renewable Fuels Association, as of October 30, 2007, 131 grain ethanol bio-refineries in the United States have the capacity to produce 7.0 billion US gallons (26,000,000 m3) of ethanol per year. An additional 72 construction projects underway (in the U.S.) can add 6.4 billion US gallons (24,000,000 m3) of new capacity in the next 18 months. Over time, it is believed that a material portion of the ≈150-billion-US-gallon (570,000,000 m3) per year market for gasoline will begin to be replaced with fuel ethanol.[116]

Sweet sorghum is a potential source of ethanol, which is suitable for growing in dryland conditions. It is being investigated by the International Crops Research Institute for the Semi-Arid Tropics (ICRISAT) for its potential to provide fuel, along with food and animal feed, in arid parts of Asia and Africa.[117] The water requirement of sweet sorghum is one-third that of sugarcane on a comparable time scale. Also, sweet sorghum requires about 22% less water than corn also known as maize. The world’s first sweet sorghum-based ethanol production distillery began commercial ethanol production in 2007 in Andhra Pradesh, India.[118]
Ethanol's high miscibility with water means that it cannot be shipped through modern pipelines like liquid hydrocarbons.[119] Mechanics have seen increased cases of damage to small engines, in particular, the carburetor, attributable to the increased water retention by ethanol in fuel.[120]
Household heating

Ethanol fuels flue-less, real flame fireplaces. Ethanol is kept in a burner containing a wick such as glass wool, a safety shield to reduce the chances of accidents and an extinguisher such as a plate or shutter to cut off oxygen.
It provides almost the same visual benefits of a real flame log or coal fire without the need to vent the fumes via a flue as ethanol produces very little hazardous carbon monoxide, and little or no noticeable scent. It does emit carbon dioxide and requires oxygen. Therefore, external ventilation of the room containing the fire is needed to ensure safe operation.
An additional benefit is that, unlike a flue based fireplace, 100% of the heat energy produced enters the room. This serves to offset some of the heat loss from an external air vent, as well as offset the relatively high cost of the fuel compared to other forms of heating.
Feedstock
Ethanol is an important industrial ingredient and has widespread use as a base chemical for other organic compounds. These include ethyl halides, ethyl esters, diethyl ether, acetic acid, ethyl amines, and, to a lesser extent, butadiene.
Antiseptic
Ethanol is used in medical wipes and in most common antibacterial hand sanitizer gels at a concentration of about 62% v/v as an antiseptic. Ethanol kills organisms by denaturing their proteins and dissolving their lipids and is effective against most bacteria and fungi, and many viruses, but is ineffective against bacterial spores.[121]
Solvent
Ethanol is miscible with water and is a good general purpose solvent. It is found in paints, tinctures, markers, and personal care products such as perfumes and deodorants.
Charts

|

|
|
| Excess volume of the mixture of ethanol and water (volume contraction) | Heat of mixing of the mixture of ethanol and water | Vapor-liquid equilibrium of the mixture of ethanol and water (including azeotrope) |
|

|
|
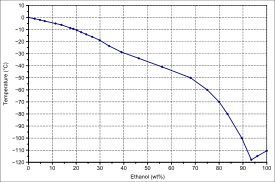
| Solid-liquid equilibrium of the mixture of ethanol and water (including eutecticum) | Miscibility gap in the mixture of dodecane and ethanol |
History
Ethanol was commonly used as fuel in early bipropellant rocket (liquid propelled) vehicles, in conjunction with an oxidizer such as liquid oxygen. The German V-2 rocket of World War II, credited with beginning the space age, used ethanol, mixed with 25% of water to reduce the combustion chamber temperature.[122][123] The V-2's design team helped develop U.S. rockets following World War II, including the ethanol-fueled Redstone rocket which launched the first U.S. satellite.[124] Alcohols fell into general disuse as more efficient rocket fuels were developed.[123]
See also
References
- ^ a b "Ethanol – Compound Summary". The PubChem Project. USA: National Center for Biotechnology Information.
- ^ Ballinger, P., Long, F.A. (1960). "Acid Ionization Constants of Alcohols. II. Acidities of Some Substituted Methanols and Related Compounds1,2". Journal of the American Chemical Society. 82 (4): 795. doi:10.1021/ja01489a008.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Arnett, E.M., Venkatasubramaniam, K.G. (1983). "Thermochemical acidities in three superbase systems". J. Org. Chem. 48 (10): 1569. doi:10.1021/jo00158a001.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ "ChemIDplus - 64-17-5 - LFQSCWFLJHTTHZ-UHFFFAOYSA-N - Alcohol [USP] - Similar structures search, synonyms, formulas, resource links, and other chemical information". Retrieved 13 October 2014.
- ^ "Ethanol Material Safety Data Sheet – Europe". Distill.com. Retrieved 2014-01-18.
- ^ "10th Special Report to the U.S. Congress on Alcohol and Health: Highlights from Current Research" (PDF). National Institute of Health. National Institute on Alcohol Abuse and Alcoholism. June 2000. p. 134. Retrieved 21 October 2014.
The brain is a major target for the actions of alcohol, and heavy alcohol consumption has long been associated with brain damage. Studies clearly indicate that alcohol is neurotoxic, with direct effects on nerve cells. Chronic alcohol abusers are at additional risk for brain injury from related causes, such as poor nutrition, liver disease, and head trauma.
- ^ Brust, John C.M. (4 April 2010). "Ethanol and Cognition: Indirect Effects, Neurotoxicity and Neuroprotection: A Review". International Journal of Environmental Research and Public Health. Int. J. Environ. Res. Public Health. pp. 1540–1557. doi:10.3390/ijerph7041540. Retrieved 21 October 2014.
Ethanol affects cognition in a number of ways. Indirect effects include intoxication, withdrawal, brain trauma, central nervous system infection, hypoglycemia, hepatic failure, and Marchiafava-Bignami disease. Nutritional deficiency can cause pellagra and Wernicke-Korsakoff disorder. Additionally, ethanol is a direct neurotoxin and in sufficient dosage can cause lasting dementia. However, ethanol also has neuroprotectant properties and in low-to-moderate dosage reduces the risk of dementia, including Alzheimer type.
{{cite web}}: CS1 maint: unflagged free DOI (link) - ^ Liebig, Justus (1834) "Ueber die Constitution des Aethers und seiner Verbindungen" (On the constitution of ether and its compounds), Annalen der Pharmacie, 9 : 1–39. From page 18: "Bezeichnen wir die Kohlenwasserstoffverbindung 4C + 10H als das Radikal des Aethers mit E2 und nennen es Ethyl, …" (Let us designate the hydrocarbon compound 4C + 10H as the radical of ether with E2 and name it ethyl …).
- ^ Harper, Douglas. "ethyl". Online Etymology Dictionary.
- ^ For a report on the 1892 International Conference on Chemical Nomenclature, see:
- Armstrong, Henry (1892). "The International Conference on Chemical Nomenclature". Nature. 46 (1177): 56–59. doi:10.1038/046056c0.
- Armstrong's report is reprinted with the resolutions in English in: Armstrong, Henry (1892). "The International Conference on Chemical Nomenclature". The Journal of Analytical and Applied Chemistry. 6: 390–400 (398).
The alcohols and the phenols will be called after the name of the hydrocarbon from which they are derived, terminated with the suffix ol (ex. pentanol, pentenol, etc.).
- ^ OED; etymonline.com
- ^ Dudley, Robert (2004). "Ethanol, Fruit Ripening, and the Historical Origins of Human Alcoholism in Primate Frugivory". Integrative Comparative Biology. 44 (4): 315–323. doi:10.1093/icb/44.4.315. PMID 21676715.
- ^ Graber, Cynthia (2008). "Fact or Fiction?: Animals Like to Get Drunk". Scientific American. Retrieved 2010-07-23.
- ^ Leblová, Sylva; Sinecká, Eva and Vaníčková, Věra (1974). "Pyruvate metabolism in germinating seeds during natural anaerobiosis". Biologia Plantarum. 16 (6): 406–411. doi:10.1007/BF02922229.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Schriver, A.; Schriver-Mazzuoli, L.; Ehrenfreund, P. and d’Hendecourt, L. (2007). "One possible origin of ethanol in interstellar medium: Photochemistry of mixed CO2–C2H6 films at 11 K. A FTIR study". Chemical Physics. 334 (1–3): 128–137. Bibcode:2007CP....334..128S. doi:10.1016/j.chemphys.2007.02.018.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ "A longitudinal study of ethanol a... [Rapid Commun Mass Spectrom. 2006] - PubMed - NCBI". Retrieved 13 October 2014.
- ^ a b Spanagel R (April 2009). "Alcoholism: a systems approach from molecular physiology to addictive behavior". Physiol. Rev. 89 (2): 649–705. doi:10.1152/physrev.00013.2008. PMID 19342616.
- ^ Harald Sitte; Michael Freissmuth (2 August 2006). Neurotransmitter Transporters. Springer Science & Business Media. pp. 472–. ISBN 978-3-540-29784-0.
- ^ Allen-Gipson DS, Jarrell JC, Bailey KL, Robinson JE, Kharbanda KK, Sisson JH, Wyatt TA. (2009). "Ethanol Blocks Adenosine Uptake via Inhibiting the Nucleoside Transport System in Bronchial Epithelial Cells". Alcohol Clin Exp Res. 33 (5): 791–8. doi:10.1111/j.1530-0277.2009.00897.x. PMC 2940831. PMID 19298329.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Becker, CE (2013-08-12). "The Clinical Pharmacology of Alcohol". California Medicine. 113 (3). Ncbi.nlm.nih.gov: 37–45. PMC 1501558. PMID 5457514.
- ^ Minutes of Meeting. Technical Committee on Classification and Properties of Hazardous Chemical Data (January 12–13, 2010).
- ^ a b "Safety data for ethyl alcohol". Msds.chem.ox.ac.uk. 2008-05-09. Retrieved 2011-01-03.
- ^ Pohorecky LA, Brick J (1988). "Pharmacology of ethanol". Pharmacol. Ther. 36 (2–3): 335–427. doi:10.1016/0163-7258(88)90109-X. PMID 3279433.
- ^ Alcohol use and safe drinking. US National Institutes of Health .
- ^ a b Yost, David A. (2002). "Acute care for alcohol intoxication" (PDF). 112 (6). Postgraduate Medicine Online. Archived from the original (PDF) on 2010-12-14. Retrieved 2007-09-29.
{{cite journal}}: Cite journal requires|journal=(help) - ^ Santhakumar V, Wallner M, Otis TS (2007). "Ethanol acts directly on extrasynaptic subtypes of GABAA receptors to increase tonic inhibition". Alcohol. 41 (3): 211–21. doi:10.1016/j.alcohol.2007.04.011. PMC 2040048. PMID 17591544.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Hingson R, Winter M (2003). "Epidemiology and consequences of drinking and driving". Alcohol research & health : the journal of the National Institute on Alcohol Abuse and Alcoholism. 27 (1): 63–78. PMID 15301401.
- ^ Naranjo CA, Bremner KE (1993). "Behavioural correlates of alcohol intoxication". Addiction. 88 (1): 25–35. doi:10.1111/j.1360-0443.1993.tb02761.x. PMID 8448514.
- ^ Attention: This template ({{cite pmid}}) is deprecated. To cite the publication identified by PMID 21332529, please use {{cite journal}} with
|pmid=21332529instead. - ^ Attention: This template ({{cite pmid}}) is deprecated. To cite the publication identified by PMID 18001279, please use {{cite journal}} with
|pmid=18001279instead. - ^ Nutrition Coordinating Center, University of Minnesota. "Primary Energy Sources". Regents of the University of Minnesota. Retrieved July 15, 2014.
- ^ Boggan, Bill. "Metabolism of Ethyl Alcohol in the Body". Chemases.com. Retrieved 2007-09-29.
- ^ Boggan, Bill. "Effects of Ethyl Alcohol on Organ Function". Chemases.com. Retrieved 2007-09-29.
- ^ Attention: This template ({{cite pmid}}) is deprecated. To cite the publication identified by PMID 12485948, please use {{cite journal}} with
|pmid=12485948instead. - ^ Attention: This template ({{cite pmid}}) is deprecated. To cite the publication identified by PMID 3188424, please use {{cite journal}} with
|pmid=3188424instead. - ^ Lukas, Scott E.; Orozco, Sara (2001). "Ethanol increases plasma Δ9-tetrahydrocannabinol (THC) levels and subjective effects after marihuana smoking in human volunteers". Drug and Alcohol Dependence. 64 (2): 143–9. doi:10.1016/S0376-8716(01)00118-1. PMID 11543984.
- ^ Repchinsky C (ed.) (2012). Compendium of pharmaceuticals and specialties, Ottawa: Canadian Pharmacists Association.[full citation needed]
- ^ Attention: This template ({{cite pmid}}) is deprecated. To cite the publication identified by PMID 2952478, please use {{cite journal}} with
|pmid=2952478instead. - ^ a b SCS Pharmaceuticals. Flagyl® IV and Flagyl® I.V. RTU® (metronidazole hydrochloride) prescribing information (dated April 16, 1997). In: Physicians’ desk reference. 48th ed. Montvale, NJ: Medical Economics Company Inc; 1998:2563-5.
- ^ "Ethanol/metronidazole", p. 335 in Tatro DS, Olin BR, eds. Drug interaction facts. St. Louis: JB Lippincott Co, 1988, ISBN 0932686478.
- ^ Sherwood, Lauralee; Kell, Robert and Ward, Christopher (2010). Human Physiology: From Cells to Systems. Cengage Learning. ISBN 978-0-495-39184-5.
{{cite book}}: CS1 maint: multiple names: authors list (link)[page needed] - ^ a b c d e f How Your Body Processes Alcohol. Dummies.com. Retrieved on 2013-04-27.[unreliable source?]
- ^ How Breathalyzers work. Electronics.howstuffworks.com
- ^ "Alcohol effects on the digestive system". Alcoholrehab.com
- ^ Overview of Peptic Ulcer Disease: Etiology and Pathophysiology. Medscape.com. Retrieved on 2013-04-27.
- ^ a b Peptic Ulcer Disease (Stomach Ulcers) Cause, Symptoms, Treatments. Webmd.com. Retrieved on 2013-04-27.
- ^ Agarwal DP, Goedde HW (1992). "Pharmacogenetics of alcohol metabolism and alcoholism". Pharmacogenetics. 2 (2): 48–62. doi:10.1097/00008571-199204000-00002. PMID 1302043.
- ^ http://monographs.iarc.fr/ENG/Classification/ClassificationsGroupOrder.pdf
- ^ "Triglycerides". American Heart Association. Archived from the original on 2007-08-27. Retrieved 2007-09-04.
- ^ Calesnick, B.; Vernick, H. (1971). "Antitussive activity of ethanol". Q J Stud Alcohol. 32 (2): 434–441. PMID 4932255.
- ^ Roach, J. (July 18, 2005). "9,000-Year-Old Beer Re-Created From Chinese Recipe". National Geographic News. Retrieved 2007-09-03.
- ^ a b Forbes, Robert James (1948) A short history of the art of distillation, Brill, p. 89, ISBN 9004006176.
- ^ Lowitz, T. (1796) "Anzeige eines, zur volkommen Entwasserung des Weingeistes nothwendig zu beobachtenden, Handgriffs" (Report of a task that must be done for the complete dehydration of wine spirits [i.e., alcohol-water azeotrope]), (Crell's) Chemische Annalen …, vol. 1, pp. 195–204. See pp. 197–198: Lowitz dehydrated the azeotrope by mixing it with a 2:1 excess of anhydrous alkali and then distilling the mixture over low heat.
- ^ "Alcohol". 1911 Encyclopædia Britannica. LoveToKnow. Archived from the original on 2006-08-21.
- ^ de Saussure, Théodore (1807) "Mémoire sur la composition de l'alcohol et de l'éther sulfurique," Journal de physique, de chimie, d'histoire naturelle et des arts, vol. 64, pp. 316–354. In his 1807 paper, Saussure determined ethanol's composition only roughly; a more accurate analysis of ethanol appears on page 300 of his 1814 paper: de Saussure, Théodore (1814) "Nouvelles observations sur la composition de l'alcool et de l'éther sulfurique," Annales de Chimie, 89: 273–305.
- ^ Couper AS (1858). "On a new chemical theory" (online reprint). Philosophical magazine. 16 (104–16). Retrieved 2007-09-03.
- ^ Faraday, M. (1825) "On new compounds of carbon and hydrogen, and on certain other products obtained during the decomposition of oil by heat," Philosophical Transactions of the Royal Society of London 115: 440–466. In a footnote on page 448, Faraday notes the action of sulfuric acid on coal gas and coal-gas distillate; specifically, "The [sulfuric] acid combines directly with carbon and hydrogen; and I find when [the resulting compound is] united with bases [it] forms a peculiar class of salts, somewhat resembling the sulphovinates [i.e., ethyl sulfates], but still different from them."
- ^ Hennell, H. (1826) "On the mutual action of sulphuric acid and alcohol, with observations on the composition and properties of the resulting compound," Philsophical Transactions of the Royal Society, vol. 116, pages 240–249. On page 248, Hennell mentions that Faraday gave him some sulfuric acid in which coal gas had dissolved and that he (Hennell) found that it contained "sulphovinic acid" (ethyl hydrogen sulfate).
- ^ Hennell, H. (1828). "On the mutual action of sulfuric acid and alcohol, and on the nature of the process by which ether is formed". Philosophical Transactions of the Royal Society of London. 118: 365–371. doi:10.1098/rstl.1828.0021. On page 368, Hennell produces ethanol from "sulfovinic acid" (ethyl hydrogen sulfate).
- ^ Sérullas, Georges-Simon (1828) "De l'action de l'acide sulfurique sur l'alcool, et des produits qui en résultent" (On the action of sulfuric acid on alcohol, and products that result from it), Annales de Chimie et de Physique, vol 39, pages 152–186. On page 158, Sérullas mentions the production of alcohol from "sulfate acid d'hydrogène carboné" (hydrocarbon acid sulfate).
- ^ In 1855, the French chemist Marcellin Berthelot confirmed Faraday's discovery by preparing ethanol from pure ethylene. Marcellin Berthelot (1855) "Sur la formation de l'alcool au moyen du bicarbure d'hydrogène" (On the formation of alcohol by means of ethylene), Annales de chimie et de physique, series 3, vol. 43, pp. 385–405. (Note: The chemical formulas in Berthelot's paper are wrong because chemists at that time used the wrong atomic masses for the elements; e.g., carbon (6 instead of 12), oxygen (8 instead of 16), etc.)
- ^ Siegel, Robert (2007-02-15). "Ethanol, Once Bypassed, Now Surging Ahead". NPR. Retrieved 2007-09-22.
- ^ DiPardo, Joseph. "Outlook for Biomass Ethanol Production and Demand" (PDF). United States Department of Energy. Retrieved 2007-09-22.
- ^ Myers, Richard L.; Myers, Rusty L. (2007). The 100 most important chemical compounds: a reference guide. Westport, Conn.: Greenwood Press. p. 122. ISBN 0-313-33758-6.
{{cite book}}: CS1 maint: multiple names: authors list (link) - ^ a b c Lide, D. R., ed. (2000). CRC Handbook of Chemistry and Physics 81st edition. CRC press. ISBN 0-8493-0481-4.
- ^ "What is the triple point of alcohol?". Webanswers.com. 2010-12-31.[scientific citation needed]
- ^ a b c d Windholz, Martha (1976). The Merck index: an encyclopedia of chemicals and drugs (9th ed.). Rahway, N.J., U.S.A: Merck. ISBN 0-911910-26-3.[page needed]
- ^ a b c Morrison, Robert Thornton; Boyd, Robert Neilson (1972). Organic Chemistry (2nd ed.). Allyn and Bacon, inc. ISBN 0-205-08452-4.
{{cite book}}: CS1 maint: multiple names: authors list (link)[page needed] - ^ Dahlmann U, Schneider GM (1989). "(Liquid + liquid) phase equilibria and critical curves of (ethanol + dodecane or tetradecane or hexadecane or 2,2,4,4,6,8,8-heptamethylnonane) from 0.1 MPa to 120.0 MPa". J Chem Thermodyn. 21 (9): 997. doi:10.1016/0021-9614(89)90160-2.
- ^ "Ethanol". Encyclopedia of chemical technology. Vol. 9. 1991. p. 813.
- ^ Costigan MJ, Hodges LJ, Marsh KN, Stokes RH, Tuxford CW (1980). "The Isothermal Displacement Calorimeter: Design Modifications for Measuring Exothermic Enthalpies of Mixing". Aust. J. Chem. 33 (10): 2103. doi:10.1071/CH9802103.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Lei Z, Wang H, Zhou R, Duan Z (2002). "Influence of salt added to solvent on extractive distillation". Chem Eng J. 87 (2): 149. doi:10.1016/S1385-8947(01)00211-X.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Pemberton RC, Mash CJ (1978). "Thermodynamic properties of aqueous non-electrolyte mixtures II. Vapour pressures and excess Gibbs energies for water + ethanol at 303.15 to 363.15 K determined by an accurate static method". J Chem Thermodyn. 10 (9): 867. doi:10.1016/0021-9614(78)90160-X.
- ^ Merck Index of Chemicals and Drugs, 9th ed.; monographs 6575 through 6669
- ^ "Flash Point and Fire Point". Nttworldwide.com.
- ^ "Flash points of ethanol-based water solutions". Engineeringtoolbox.com. Retrieved June 23, 2011.
- ^ Wolke, Robert L. (5 July 2006). "Combustible Combination". Washington Post. Retrieved 27 January 2011.
- ^ Mills GA, Ecklund EE (1987). "Alcohols as Components of Transportation Fuels". Annual Review of Energy. 12: 47. doi:10.1146/annurev.eg.12.110187.000403.
- ^ Roberts, John D.; Caserio, Marjorie C. (1977). Basic Principles of Organic Chemistry. W. A. Benjamin, Inc. ISBN 0-8053-8329-8.[page needed]
- ^ a b c d Naim Kosaric, Zdravko Duvnjak, Adalbert Farkas, Hermann Sahm, Stephanie Bringer-Meyer, Otto Goebel and Dieter Mayer in "Ethanol" Ullmann's Encyclopedia of Industrial Chemistry, 2011, Wiley-VCH, Weinheim. doi:10.1002/14356007.a09_587.pub2
- ^ "Ethanol". Encyclopedia of chemical technology. Vol. 9. 1991. p. 82.
- ^ Lodgsdon, J.E (1991). "Ethanol". In Howe-Grant, Mary; Kirk, Raymond E.; Othmer, Donald F.; Kroschwitz, Jacqueline I. (ed.). Encyclopedia of chemical technology. Vol. 9 (4th ed.). New York: Wiley. p. 817. ISBN 0-471-52669-X.
{{cite book}}: CS1 maint: multiple names: editors list (link) - ^ a b c d e Streitweiser, Andrew Jr.; Heathcock, Clayton H. (1976). Introduction to Organic Chemistry. MacMillan. ISBN 0-02-418010-6.
{{cite book}}: CS1 maint: multiple names: authors list (link) - ^ Medium and long-term opportunities and risks of the biotechnological production of bulk chemicals from renewable resources – the potential of white biotechnology. The BREW project final report, Utrecht, September 2006. p. 4.
- ^ Badger, P.C. "Ethanol From Cellulose: A General Review." p. 17–21. In: J. Janick and A. Whipkey (eds.), Trends in new crops and new uses. ASHS Press, 2002, Alexandria, VA. Retrieved on September 2, 2007.
- ^ Taherzadeh MJ, Karimi K (2007). "Acid-based hydrolysis processes for ethanol from lignocellulosic materials: A review" (PDF). BioResources. 2: 472.
- ^ Taherzadeh MJ, Karimi K (2007). "Enzymatic-based hydrolysis processes for ethanol from lignocellulosic materials: A review" (PDF). BioResources. 2: 707.
- ^ Ritter SK (2004). "Biomass or Bust". Chemical & Engineering News. 82 (22): 31. doi:10.1021/cen-v082n048.p031.
- ^ Clines, Tom (July 2006). "Brew Better Ethanol". Popular Science Online. Archived from the original on 2007-11-03.
- ^ Market access will save US from $4 gasoline, says FuelChoiceNow BiofuelsDigest, Sep. 21, 2011. Retrieved Apr. 6, 2012.
- ^ Jeong, Jun-Seong; Jeon, Hyungjin; Ko, Kyung-mo; Chung, Bongwoo; Choi, Gi-Wook (2012). "Production of anhydrous ethanol using various PSA (Pressure Swing Adsorption) processes in pilot plant". Renewable Energy. 42: 41. doi:10.1016/j.renene.2011.09.027.
- ^ "U-M Program to Reduce the Consumption of Tax-free Alcohol; Denatured Alcohol a Safer, Less Expensive Alternative" (PDF). University of Michigan. Retrieved 2007-09-29.
- ^ Great Britain (2005). The Denatured Alcohol Regulations 2005. Statutory Instrument 2005 No. 1524.
- ^ Bernthsen, August; Bansal, Raj K. (2003) A textbook of organic chemistry, p. 402, ISBN 81-224-1459-1.
- ^ Christian, Gary D. (2003) Analytical chemistry, Vol. 1, Wiley, ISBN 0-471-21472-8[page needed]
- ^ Textbook Of Food & Bevrge Mgmt, Tata McGraw-Hill, 2007 ISBN 0-07-065573-1 p. 268
- ^ Kunkee, Ralph E. and Amerine, Maynard A. (1968). "Sugar and Alcohol Stabilization of Yeast in Sweet Wine". Appl Microbiol. 16 (7): 1067–75. PMC 547590. PMID 5664123.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Rossini, Frederick D. (1937). "Heats of Formation of Simple Organic Molecules". Ind. Eng. Chem. 29 (12): 1424. doi:10.1021/ie50336a024.
- ^ Calculated from heats of formation from CRC Handbook of Chemistry and Physics, 49th Edition, 1968–1969.
- ^ Chakrabartty, in Trahanovsky, Oxidation in Organic Chemistry, pp 343–370, Academic Press, New York, 1978
- ^ Appendix B, Transportation Energy Data Book from the Center for Transportation Analysis of the Oak Ridge National Laboratory
- ^ a b Eyidogan, Muharrem; Ozsezen, Ahmet Necati; Canakci, Mustafa; Turkcan, Ali (2010). "Impact of alcohol–gasoline fuel blends on the performance and combustion characteristics of an SI engine". Fuel. 89 (10): 2713–2720. doi:10.1016/j.fuel.2010.01.032.
- ^ a b Thomas, George: Template:PDF. Livermore, CA. Sandia National Laboratories. 2000.
- ^ Thomas, George (2000). "Overview of Storage Development DOE Hydrogen Program" (PDF). Sandia National Laboratories. Retrieved 2009-08-01.
- ^ Reel, M. (August 19, 2006) "Brazil's Road to Energy Independence", Washington Post.
- ^ Rocket Racing League Unveils New Flying Hot Rod, by Denise Chow, Space.com, 2010-04-26. Retrieved 2010-04-27.
- ^ Green, Ray. "Model T Ford Club Australia (Inc.)". Retrieved 24 June 2011.
- ^ Ethanol 101. American Coalition for Ethanol.
- ^ The Biofuels FAQs, The Biofuels Source Book, Energy Future Coalition, United Nations Foundation.
- ^ California Air Resources Board, Definition of a Low Emission Motor Vehicle in Compliance with the Mandates of Health and Safety Code Section 39037.05, second release, October 1989
- ^ Lowi, A. and Carter, W.P.L.; A Method for Evaluating the Atmospheric Ozone Impact of Actual Vehicle emissions, S.A.E. Technical Paper, Warrendale, PA; March 1990
- ^ Jones, T.T.M. (2008) The Clean Fuels Report: A Quantitative Comparison Of Motor Fuels, Related Pollution and Technologies. researchandmarkets.com
- ^ "Adoption of the Airborne Toxic Control Measure to Reduce Formaldehyde Emissions from Composite Wood Products". USA: Window and Door Manufacturers Association. July 30, 2008. Archived from the original on 2010-03-09.
- ^ "2008 World Fuel Ethanol Production". U.S.: Renewable Fuels Association.
- ^ "Tecnologia flex atrai estrangeiros". Agência Estado.
- ^ "First Commercial U.S. Cellulosic Ethanol Biorefinery Announced". Renewable Fuels Association. 2006-11-20. Retrieved May 31, 2011.
- ^ Sweet sorghum for food, feed and fuel New Agriculturalist, January 2008.
- ^ Developing a sweet sorghum ethanol value chain ICRISAT, 2013
- ^ Horn, W. and Krupp, F. (2008) Earth: The Sequel: The Race to Reinvent Energy and Stop Global Warming, W. W. Norton, p. 85, ISBN 0393068102.
- ^ Mechanics see ethanol damaging small engines, Msnbc.com, 8 January 2008
- ^ McDonnell G, Russell AD (1999). "Antiseptics and disinfectants: activity, action, and resistance". Clin. Microbiol. Rev. 12 (1): 147–79. PMC 88911. PMID 9880479.
- ^ Darling, David. "The Internet Encyclopedia of Science: V-2".
- ^ a b Braeunig, Robert A. "Rocket Propellants." (Website). Rocket & Space Technology, 2006. Retrieved on 2007-08-23.
- ^ "A Brief History of Rocketry." NASA Historical Archive, via science.ksc.nasa.gov.
Further reading
- The National Institute on Alcohol Abuse and Alcoholism maintains a database of alcohol-related health effects. ETOH Archival Database (1972–2003) Alcohol and Alcohol Problems Science Database.
- Boyce, John M., and Pittet Didier. (2003). "Hand Hygiene in Healthcare Settings". Centers for Disease Control, Atlanta, Georgia, United States.
- Onuki, Shinnosuke; Koziel, Jacek A.; van Leeuwen, Johannes; Jenks, William S.; Grewell, David; Cai, Lingshuang (June 2008). Ethanol production, purification, and analysis techniques: a review. 2008 ASABE Annual International Meeting. Providence, RI. Retrieved February 16, 2013.
- Sci-toys website explanation of US denatured alcohol designations
- Smith, M.G., and M. Snyder. (2005). "Ethanol-induced virulence of Acinetobacter baumannii". American Society for Microbiology meeting. Volume 1 June 5 – June 9. Atlanta.
External links
- Alcohol (Ethanol) at The Periodic Table of Videos (University of Nottingham)
- International Labour Organization ethanol safety information
- National Pollutant Inventory – Ethanol Fact Sheet
- CDC – NIOSH Pocket Guide to Chemical Hazards – Ethyl Alcohol
- National Institute of Standards and Technology chemical data on ethanol
- ChEBI – biology related
- Chicago Board of Trade news and market data on ethanol futures
- Calculation of vapor pressure, liquid density, dynamic liquid viscosity, surface tension of ethanol
- Ethanol History A look into the history of ethanol
- ChemSub Online: Ethyl alcohol
- Industrial ethanol production process flow diagram using ethylene and sulphuric acid
- Alcohol solvents
- Anatomical preservation
- Anxiolytics
- Disinfectants
- Ethanol
- 5-HT3 agonists
- GABAA receptor positive allosteric modulators
- Glycine receptor agonists
- Household chemicals
- Human metabolites
- NMDA receptor antagonists
- Oxygenates
- Primary alcohols
- Rocket fuels
- Sedatives
- Teratogens
- IARC Group 1 carcinogens
- Commodity chemicals
- Cholinergics